I contracted Covid-19 in March 2000. The medics had to beat on my chest to bring me back to life. I was in Lenox-Hill Hospital for a month with a lung tube. The disease left one of my fingers paralized making it hard to type. So I did some research which came down to this: Peter Cottonton Daszak deliberately and intentionally caused a pandemic by putting all the players in place then ignoring warning signs that would be apparent to anyone with his credentials. It's all here, all referenced with live links to my sources. 3/7/2025
With its new focus, Accountability Journalism Institute has expanded its board of directors, bringing in respected scientists, human rights advocates, and journalists. Among the new board members is renowned zoologist Dr. Peter Daszak, the former president of EcoHealth Alliance, who has been the target of politically motivated attacks based on unsupported narratives about the origins of COVID-19. "I joined the Board because I've seen first hand the damage that politically-driven misinformation can do through five years of awful attacks against me, my family and my work," Daszak said. "These often involved the same methods, organizations and people as those lined up against climate change, GMO and vaccine scientists. The AJI will shine a light on who's behind anti-science misinformation, and who pays for it, so we can all better distinguish fact from fiction." House Oversight Committee Report points to Daszak as cause of Covid-19 Pandemic
FINDING: Dr. Peter Daszak Provided False Statements to Congress in Violation of 18 U.S.C. 1001 and 18 U.S.C. 1621. In comparing Dr. Daszaks testimonyboth during his transcribed interview and public hearingto available documents, it is likely Dr. Daszak provided false statements to the Select Subcommittee in violation of 18 U.S.C. 1001 and 18 U.S.C. 1621.
a) Except as otherwise provided in this section, whoever, in any matter within the jurisdiction of the executive, legislative, or judicial branch of the Government of the United States, knowingly and willfully
(1) falsifies, conceals, or covers up by any trick, scheme, or device a material fact;
(2) makes any materially false, fictitious, or fraudulent statement or representation; or
(3) makes or uses any false writing or document knowing the same to contain any materially false, fictitious, or fraudulent statement or entry;
shall be fined under this title, imprisoned not more than 5 years or, if the offense involves international or domestic terrorism (as defined in section 2331), imprisoned not more than 8 years, or both. If the matter relates to an offense under chapter 109A, 109B, 110, or 117, or section 1591, then the term of imprisonment imposed under this section shall be not more than 8 years.
1) The possibility that COVID-19 emerged because of a laboratory or research related accident is not a conspiracy theory.
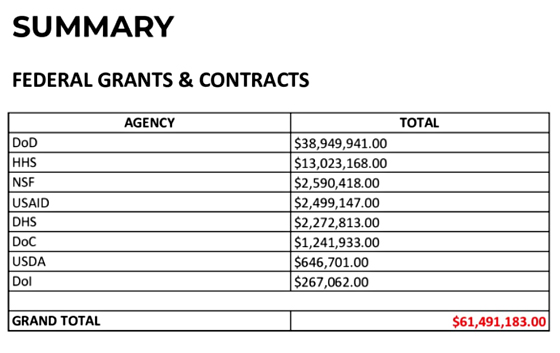
2) EcoHealth Alliance, Inc. and Dr. Peter Daszak should never again receive U.S. taxpayer dollars.
3) The U.S. National Institutes of Health funded gain-of-function research at the Wuhan Institute of Virology.
2) The Chinese government, agencies within the U.S. Government, and some members of the international scientific community sought to cover-up facts concerning the origins of the pandemic.

From: Peter Daszak Sent: Tuesday April 28, 2020 11:30 AM TO: Honging Li Tammie O'Rourke CC: Goldstein,Tracy Alekseia Chmura Christine Kreuder Johnson Subject: Re: China Genbank Sequences Importance: High All- It's extremely important that we don't have these sequences as part of our PREDICT release to Genbank at this point. As you probably heard this was part of a grant terminated by the N.I.H. https://politico.com/news2020/04/27/trump-cuts-research-bat-human-virus-china-213076 Having them as part of PREDICT will bring very unwanted attention to UC Davis, PREDICT and USAID. Cheers, Peter




Dr. Daszak: "This summary contains the information for the 2014 and 2017 NIH and NIAID grants to the Ecohealth Alliance that funded the WIV research on bat conronaviruses. As the grant description shows, this research included gain-of-function / gain-of-threat research to make coronaviruses viruses more pathogenic using techniques including genetic engineering, cell culture, and animal experimentation." Source of document: Covid-references-International Center for Technology Assessment
Dr. Fauci told Rand Paul: Senator, with all due respect, you are entirely and completely incorrect, that the N.I.H. has not ever and does not now fund gain-of-function research in the Wuhan Institute. The N.I.H. used Daszak as its surrogate to do this. To hold Daszak responsible means holding Fauci responsible which the Democrats and Corporate Media will not do.
https://www.nytimes.com/2021/06/20/science/covid-lab-leak-wuhan.html
This entry (above) refers to https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1006698f DATED NOVEMBER 30, 2017: Daszak claimed this experiment was funded by USAID, NIH etc. This is a lie. It was funded by the ChiComs. Ben Hu, Lei-Ping Zeng, Xing-Lou Yang, Xing-Yi Ge, Wei Zhang, Bei Li, Jia-Zheng Xie, Xu-Rui Shen, Yun-Zhi Zhang, Ning Wang, Dong-Sheng Luo, Xiao-Shuang Zheng, Mei-Niang Wang, Peter Daszak, Lin-Fa Wang, Jie Cui, Zheng-Li Shi.

WAP editorial .... original Daszak strain mutated into OMICRON Global tally of new Omicron coronavirus cases has for the first time passed one million per day on average. NYT...... Leftwing Annenberg Foundation and Big Pharma Johnson & Johnson attack lab leak theory China reports on Congressional investigation FOLLOW BILLY BOSTICKSON ON TWITTER. DRASTIC...Scientist who wrote anti-lab-leak-letter on Fauci's pad...Daszak and FBI Intercept Article...Smithsonian Funding Daszak?
"Construction of recombinant viruses: Recombinant viruses with the S gene of the novel bat SARSr-CoVs and the backbone of the infectious clone of SARSr-CoV WIV1 were constructed using the reverse genetic system described previously. The fragments E and F were re-amplified with primer pairs...The fragment Es and Fs were both digested with BglI (NEB) and BsaI (NEB). The Rs4231 S gene was digested with BsmBI. The Rs7327 S gene was digested with BsaI. The other fragments and bacterial artificial chromosome (BAC) were prepared as described previously. Then the two prepared spike DNA fragments were separately inserted into BAC with Es, Fs and other fragments. The correct infectious BAC clones were screened. The chimeric viruses1 were rescued as described previously." (Deborah A. Kimbrell is Associate Research Geneticist Emerita and Lecturer Unit Department of Molecular and Cellular Biology, College of Biological Sciences U of C Davis).
1. A chimeric virus is a genetic chimerism or chimera is a single organism composed of cells with more than one distinct genotype. These experiments were creating novel viruses like the SARS-CoV19N.
Doctor "Daszakstein" wanted to see a pandemic engulf humanity to cut down human population in favor of vegitation and animal life, so he convinced Fauci, USAID, Pentagon and others to fund his PREDICT PROGRAM. This is why Daszak will never embrace the lab-leak theory, because he would be incriminating himself. DASZAK: "The implications of wildlife EIDs (Emerging Infectious Diseases) are twofold: emerging wildlife diseases cause direct and indirect loss of biodiversity and add to the threat of zoonotic disease emergence. Since human environmental changes are largely responsible for their emergence, the threats wildlife EIDs pose to biodiversity and human health represent yet another consequence of anthropogenic influence on ecosystems. More and more people are also living and working closer to wildlife. It isnt about one or two individuals putting people at risk. The risk also comes from clear-cutting rainforests, remote mining, and even widespread suburbanization." Anthropogenic means caused by humans. Daszak tweeted this to IBPES "Most pandemics are linked to wildlife and human-caused environmental change, such as deforestation and wildlife trade." His Zoonotic spill-over from nature cover-story, however, fell apart after "an allied power," (probably France who built the BSL-4 LAB at the WIV), determined several scientists became contaminated with an enhanced SARS virus and brought it, within their bodies, into Wuhan, during its incubation period and after the FBI determined with moderate certainty there was a lab leak.
How could one man cause all this havoc? He is a cunning psychopathic zoologist, although he is sometimes listed as a parasitologist. The lab leak theory has been entwined with politics, rather than science, thanks to Donald Trump who promulgated the theory alongside his bleach drinking remedy, and thanks to Fauci, who has become a target of anti-vaccination types and who must resign or be fired, because he approved Daszak's experiments. Daszak could have blown the whistle on the dangerous conditions at the WIV, cut their funding, and prevented the pandemic, instead he did just the opposite. He didn't do a thing because he is an Eco-Fascist who wanted a pandemic. He must be given a fair trial in USDC/SDNY then face the federal death penalty. There is recedent for charging Daszak: A Brazilian congressional panel recommend President Jair Bolsonaro be charged with crimes against humanity, asserting that he intentionally let the coronavirus rip through the country and kill hundreds of thousands in a failed bid to achieve herd immunity and revive Latin Americas largest economy. He was blamed for 300,000 deaths NYT
Daszak violated the law regarding grant subrecipients that required him to "Monitor the activities of the subrecipient as necessary to ensure that the subaward is used for authorized purposes, in compliance with Federal statutes, regulations, and the terms and conditions of the subaward; and that subaward performance goals are achieved. Pass-through entity monitoring of the subrecipient must include: (1) Providing subrecipients with training and technical assistance on program-related matters; and (2) Performing on-site reviews of the subrecipient's program operations; (3) Arranging for agreed-upon-procedures engagements as described in § 75.425.
https://www.govregs.com/regulations/title45_chapterA_part75_subpartD_subjgrp28_section75.352
In order to receive the grant Daszak had to sign-off on this declaration: "I am aware that any false, fictitious, or fraudulent information, or the omission of any material fact, may subject me to criminal, civil or administrative penalties for fraud, false statements, false claims or otherwise. (U.S. Code Title 18, Section 1001 and Title 31, Sections 3729-3730 and 3801-3812). There is no statute of limitations for embezzelment of federal money. Fauci knew where the money was going - straight to the WIV to conduct these "maligned" experiments that Fauci thought might save mankind. Fauci let this GoF research out-sourcing go down. Program Officer Erik J Stemmy probably didn't approve Daszak's grants without showing them to his boss, Fauci. But it was Peter Daszak who caused the pandemic that setback humanity for decades.

Just as Robert Kennedy Jr., an environmental lawyer, uses anti-vaccination propaganda to increase the human death rate, Daszak uses his "small non-profit," as Dr. Fauci calls it, the Eco-Health Alliance, to do the same. Also see Also see robertkennedyjr.org

The limited experiment described in the final progress report provided by EcoHealth Alliance was testing if spike proteins from naturally occurring bat coronaviruses circulating in China were capable of binding to the human ACE2 receptor in a mouse model. All other aspects of the mice, including the immune system, remained unchanged. In this limited experiment, laboratory mice infected with the SHC014 WIV1 bat coronavirus became sicker than those infected with the WIV1 bat coronavirus. As sometimes occurs in science, this was an unexpected result of the research, as opposed to something that the researchers set out to do. Regardless, the viruses being studied under this grant were genetically very distant from SARS-CoV-2. The research plan was reviewed by NIH in advance of funding, and NIH determined that it did not to fit the definition of research involving enhanced pathogens of pandemic potential (ePPP) because these bat coronaviruses had not been shown to infect humans. As such, the research was not subject to departmental review under the HHS P3CO Framework. However, out of an abundance of caution and as an additional layer of oversight, language was included in the terms and conditions of the grant award to EcoHealth that outlined criteria for a secondary review, such as a requirement that the grantee report immediately a one log increase in growth. These measures would prompt a secondary review to determine whether the research aims should be re- evaluated or new biosafety measures should be enacted. EcoHealth failed to report this finding right away, as was required by the terms of the grant. EcoHealth is being notified that they have five days from today to submit to NIH any and all unpublished data from the experiments and work conducted under this award. Additional compliance efforts continue. Lawrence A. Tabak, D.D.S., Ph.D. Principal Deputy Director
Dr. Tabak is the principal deputy director of the National Institutes of Health (NIH) and the deputy ethics counselor of the Agency. He previously served as acting principal deputy director of NIH (2009), and prior to that as director of the National Institute of Dental and Craniofacial Research from 2000-2010. Prior to joining NIH, Dr. Tabak was the senior associate dean for Research and professor of Dentistry and Biochemistry & Biophysics in the School of Medicine and Dentistry at the University of Rochester in New York. A former NIH MERIT recipient, Dr. Tabak's major research focus has been on the structure, biosynthesis and function of glycoproteins. Also see New York Times Bat Research Group Failed to Submit Virus Studies Promptly, N.I.H. Says
JUNE 6, 2014 "We will use Spike Protein1 sequence data, infectious clone technology,2 in vitro and in vivo3 infection experiments and analysis of receptor binding to test the hypothesis that % divergence thresholds in Spike Protein sequences predict spillover potential. We will combine these data with bat host distribution, viral diversity and phylogeny, human survey of risk behaviors and illness, and serology to identify SARSr-CoV spillover risk hotspots across southern China. Together these data and analyses will be critical for the future development of public health interventions and enhanced surveillance to prevent the re-emergence of SARS or the emergence of a novel SARSr-CoV. Project Start 2014-06-01 / Project End 2020-04-24 / Budget Start 2019-07-24 / Budget End 2020-04-24 / Support Year 6 / Fiscal Year 2019 / Total Cost / Indirect Cost" https://grantome.com/grant/NIH/R01-AI110964-06
1. The spike protein is what gives the Corona Virus its unique name. It is the part of the virus that plugs into the ACE2 receptors in human lungs and allows the nano-particle to infect the human body just like plugging a USB cord into a port on your computer.
2. Infectious clones of viral genomes were initially produced in the late 1970s and early 1980s. The first was made in 1978 by inserting a DNA copy of the RNA genome of the bacteriophage QB, made with reverse transcriptase, into a plasmid vector. Infectious virus was produced when the cloned viral DNA was inserted into E. coli. In 1980, infectious cloned retroviral DNA was produced by inserting the integrated viral DNA from the cellular genome into a plasmid vector. The next year, a DNA copy of the RNA genome of poliovirus was produced by reverse transcription and inserted into a plasmid vector. When the cloned copy of the viral genome was introduced into mammalian cells, infectious virus was produced. The infectious viral DNA clone is a double-edged sword. It enables manipulation of the viral genome at will, allowing unprecedented genetic analysis and the use of viruses as vectors for gene therapy. But nearly any virus can now be recovered from the nucleotide sequence effectively making it impossible to ever truly eradicate a virus from the globe. This was thanks to Baric "reverse genetics."
3. In vitro: The term in vitro refers to a medical study or experiment which is done in the laboratory within the confines of a test tube or laboratory dish. In vivo: The term in vivo refers to a medical test, experiment, or procedure that is done on (or in) a living organism, such as a laboratory animal or human.
1. The proposal is considered to potentially involve GoF/DURC research because they propose to synthesize spike glycoproteins which bind to human cell receptors and insert them into SARSr-CoV backbones to assess whether they can cause SARS-like disease.
2. However the proposal does not mention or assess potential risks of Gain of Function (GoF) research.
3. Nor does the proposal mention or assess Dual Use Research of Concern (DURC) issues, and thus fails to present a DURC risk mitigation plan.
4. The proposal hardly addresses or discusses ethical, legal, and social issues (ELSI).
Atlantic Mag. confirms docs authenticity, but claims it does not bolster the lab leak theory. "The most alarming aspect of the research plan revealed in the documents is a plan to search viral genetic databases for new types of 'furin cleavage sites' which help a virus attack a host. According to the proposal, 'high-risk' versions of these sites, once identified, would then be grafted onto SARS-like bat coronaviruses via genetic engineering. DAILY MAIL....Lancet Panel Disbanded thanks to Daszak...

What is SARSr-CoV that is being used to circumvent gain-of-function concerns? "Herein, we report the findings of our 5-year surveillance of SARSr-CoVs in a cave inhabited by multiple species of horseshoe bats in Yunnan Province, China. The full-length genomes of 11 newly discovered SARSr-CoV strains, together with our previous findings, reveals that the SARSr-CoVs circulating in this single location are highly diverse in the S gene, ORF3 and ORF8. Importantly, strains with high genetic similarity to SARS-CoV in the hypervariable N-terminal domain (NTD) and receptor-binding domain (RBD) of the S1 gene, the ORF3 and ORF8 region, respectively, were all discovered in this cave. In addition, we report the first discovery of bat SARSr-CoVs highly similar to human SARS-CoV in ORF3b and in the split ORF8a and 8b."
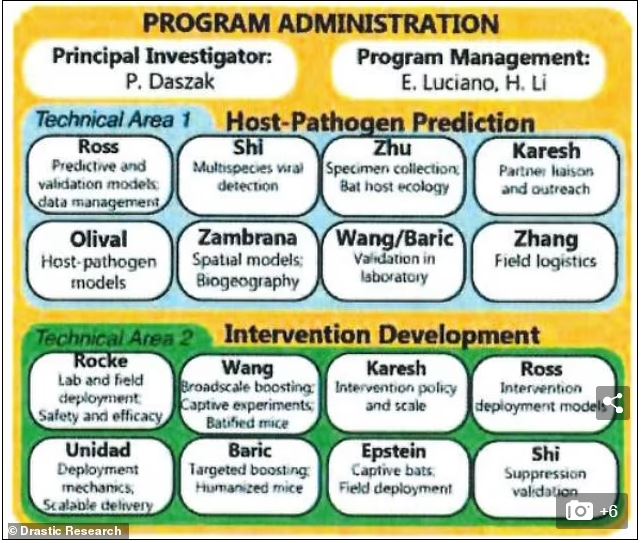
DRASTIC Summary of EcoHealth's DEFUSE Grant Proposal
EcoHealth Alliance Executive Summary of DEFUSE
EcoHealth's full DEFUSE grant proposal to DARPA
"Because Of its (now) known nature, the SARSr-CoV-WIV s illness is readily resolved with early treatment that inhibits the viral replication that spreads the spike protein around the body (which induce a harmful overactive immune response as the body tries to clear the spikes from the ACE-2 receptors.) Many of the early treatment protocols ignored by the authorities because they inhibit viral replication or modulate the immune response to the spike proteins which makes sense within the context of what Ec-Health was creating. Some of these treatment protocols also inhibit the action of the engineered spike protein. For instance Ivermectin (Identified a curative in April 2020) is identified in the proposal as a SARS-Cov inhibitor, as is interferon (Identified May 2020 as a curative.)"

Most of the recombinant experiences were carried out by Baric and Shi financed by the NIH and Daszak's re-purposing of Eco-Health grants. NOVEMBER 21, 2019 Daszak tweeted: "Weve made great progress with bat SARS-related CoVs, IDing 50 novel strains, sequencing Spike Protein genes, IDing ones that bind to human cells, using recombinant viruses, humanized mice to see SARS-like signs, and showing some dont respond to MAbs, vaccines."
Using the reverse genetics technique we previously developed for WIV1, we constructed a group of infectious bacterial artificial chromosome (BAC) clones with the backbone of WIV1 and variants of S genes from 8 different bat SARSr-CoVs. [Ben Hu ,Lei-Ping Zeng , Xing-Lou Yang , Xing-Yi Ge, Wei Zhang, Bei Li, Jia-Zheng Xie, Xu-Rui Shen, Yun-Zhi Zhang, Ning Wang, Dong-Sheng Luo, Xiao-Shuang Zheng, Mei-Niang Wang, Peter Daszak, Lin-Fa Wang, Jie Cui, Zheng-Li Shi] Yuan KY et. al. assayed 13 different species of bats. The results proved that SARS-CoV in bats was the most closely related to that in humans. The SARS-like CoV in Chinese horseshoe bats has 88%-92% sequence homology with human SARS-CoV. Moreover, the S2 m motif in its 3'UTRs and the phylogenetic analyses of four fully characterized genomes of SARS-like CoV indicated that the horseshoe bats have great potential to be one of the natural reservoirs. In MAY 2016 the United States Government issued a REPORT that contained a set of strict rules for determining which DURC GoF/GoT experiments should be funded by the National Institute of Health and other relevant USG entities based on Risk v. Benefit. There was a controversy in the scientific community as to the value of these experiments, however, Daszak was fully committed to performing these proceedures and continued to outsource them to the WIV where no such oversight existed.
Founded in 1956, Wuhan Institute of Virology (WIV), Chinese Academy of Sciences (CAS), was initially named as Wuhan Microbiology Laboratory. It was among the earliest national institutions established after the founding of the New China. In 1961, it became the South China Institute of Microbiology of CAS, and was redesignated as Wuhan Microbiology Institute CAS in 1962. In 1970, under the administration of the Hubei Commission of Science & Technology, it was renamed as Microbiology Institute of Hubei Province. In June 1978, it was returned to the administration of CAS, and adopted its current designation.
For the Shi Zheng Li complete Question and Answer document where she admits to this click HERE
"Wuhan Institute of Virology, Wuhan, China (Zhengli Shi and Xingyi Ge) The Shi laboratory includes 4 rooms totaling (b) (4 , one equipped with two C02 incubators for tissue culture, one equipped facilities including with high speed centrifuge, 2 -20°C, 3 -80°C freezers, 2 PCR machines, 1 ELISA plate reader, one for molecule diagnosis equipped with two biosafety cabinets, and one normal laboratory equipped various small equipment items (mini-centrifuges, gel electrophoresis units, circulating adjustable water baths, and heat blocks). Also available to Dr. Shi's group is a fully equipped biosafety level 3 laboratory, a newly opened BLS-4 laboratory (the first in China) and Institute-supported facility center, which houses full-time staff and equipment for electronic microscopy, ultracentrifugation, confocal microcopy, and sequencing machine. Experiment conducted in BSL-2 Lab The FBI is the one agency whose views were identified in the Biden Report and it stated their was an accidental leak. Yet Carl Zimmer of the NYT continues to put one computer scientist's theory that the virus came from nature on the front page. Daszak is the number one proponent of the "it came from nature" theory to protect himself. Afterall, how could it be co-incidence that of all the places to first break-out, it was in the same city Daszak's experiments were being conducted on Corona viruses? WSJ on the Lab Leak theory
[The FBI] assesses with moderate confidence that the first human infection with SARS-CoV-2 most likely was the result of a laboratory-associated incident, probably involving experimentation, animal handling, or sampling by the Wuhan Institute of Virology. These analysts give weight to the inherently risky nature of work on coronaviruses...For these hypotheses, IC analysts consider an exposure that occurs during animal sampling activity that supports biological research to be a laboratory- associated incident and not natural contact....They also see the potential that a laboratory worker inadvertently was infected while collecting unknown animal specimens...[The FBI] assesses with moderate confidence that COVID-19 most likely resulted from a laboratory- associated incident involving WIV or other researcherseither through exposure to the virus during experiments or through sampling. Some analysts at elements that are unable to coalesce around either explanation also assess a laboratory origin with low confidence. These analysts place emphasis on academic articles authored by WIV employees indicating that WIV scientists conducted research on other coronaviruses under what these analysts consider to be inadequate biosafety conditions that could have led to opportunities for a laboratory-associated incident. These analysts also take into account SARS-CoV-2s genetic epidemiology and that the initial recorded COVID-19 clusters occurred only in Wuhanand that WIV researchers who conducted sampling activity throughout China provided a node for the virus to enter the city....[The FBI] finds the laboratory-associated origin theory most likely assess that WIV researchers inherently risky work with coronaviruses provided numerous opportunities for them to unwittingly become infected with SARS-CoV-2. Although the IC has no indications that WIV research involved SARS-CoV-2 or a close progenitor virus, [the FBI] notes that it is plausible that researchers may have unwittingly exposed themselves to the virus without sequencing it during experiments or sampling activities, possibly resulting in asymptomatic or mild infection. Academic literature indicates that WIV researchers conducted research with bat coronaviruses or collected samples from species that are known to carry close relatives of SARS-CoV-2. Based on currently available information, the closest known relatives to SARS-CoV-2 in bats have been identified in Yunnan Province, and researchers bringing samples to laboratories provide a plausible link between these habitats and the city. [The FBI] analysts that assess COVID-19 most likely originated from a laboratory-associated incident also place emphasis on information suggesting researchers in China used biosafety practices that increased the risk of exposure to viruses. Academic publications suggest that WIV researchers did not use adequate biosafety precautions at least some of the time, increasing the risk of a laboratory-associated incident.
[The FBI] analysts that find the laboratory-associated origin theory most likely assess that WIV researchers inherently risky work with coronaviruses provided numerous opportunities for them to unwittingly become infected with SARS-CoV-2. Although the IC has no indicationsthat WIV research involved SARS-CoV-2 or a close progenitor virus, [the FBI] analysts note that it is plausible that researchers may have unwittingly exposed themselves to the virus without sequencing it during experiments or sampling activities, possibly resulting in asymptomatic or mild infection. Academic literature indicates that WIV researchers conducted research with bat coronaviruses or collected samples from species that are known to carry close relatives of SARS-CoV-2.
Based on currently available information, the closest known relatives to SARS-CoV-2 in bats have been identified in Yunnan Province, and researchers bringing samples to laboratories provide a plausible link between these habitats and the city.
[The FBI] analysts also note that Chinas investigations into the pandemics origin might not uncover evidence of a laboratory-associated incident if it involved only a small number of researchers who did not acknowledge or have knowledge of a potential infection. FBI IC VIEWS REVEALED Q "What did you learn in the course of your research about the origins of COVID?
A. I learned that we're not going to answer this question absent one of two things happening, either we find the intermediate host, the animal that spread COVID, or there's a whistleblower inside China, or someone close to this who knows that this came out of a lab, comes forward, defects, goes overseas, or we intercept some communication that we shouldn't have had access to. Absent something like that, we're not going to be able to answer this question. This is going to be a battle of competing narratives. I think over time, the side of the ledger that that says that this might have come out of a lab has grown more robust and the side of the ledger that this came out of a natural species has not really moved. And if anything, you can argue that side of the ledger has been diminished by a couple of facts. Number one, we've looked for the intermediate host and we haven't found it. And number two, the idea that the market, this initial market, was the source of the initial spread has been firmly debunked, even the Chinese acknowledged that. And that was a big part of the thesis around the zoonotic origin, the animal origin, because the idea was that the animal was in that market. We now know that market wasn't the source of the spread. It was a path along the spread. Beijing has even acknowledged that-- Beijing had acknowledged it, right.
Q. But when you say intermediate host, you mean what happened between the bat and the human, who came in between.
A. There's an animal, maybe a pangolin, there's some other animal in between and it could have been-- it wasn't necessarily a bat and it could have been a bat to a human, but we haven't found the virus in nature. And there's been a pretty exhaustive search. The Chinese have mounted an exhaustive search, and I don't think that the fact that they've been looking for it proves that they know it didn't come out of a lab. I think that they would be looking for it even if they knew it came out of a lab. And the reality is if it came out of a lab, the number of people who actually are aware of that could be a very small subset of people.
Q. When you say came out of a lab, you were saying through a lab accident, not a construct?
A. Yeah. No, I don't think anyone, and the administration has said this in the intelligence report they put out that they don't believe-- they've sort of firmly debunked the idea that this was something that could have deliberately come out of a lab, or was deliberately engineered. But the possibility is that you've had labs doing research on novel coronaviruses, you had novel coronaviruses being brought to those labs, particularly the Wuhan Institute of Virology. We know that they were doing that research in what's called BSL-2 labs, lower security labs, where tight precautions aren't taken. We also know that the Wuhan Institute of Virology [BSL-4] was a sloppy lab. At the time that it opened, there was an article in the journal Science raising questions about the integrity of that lab where scientists at the time that it opened in 2017 [2018] said, we're worried about the procedures in this lab. We're worried about the training, we're worried about the way it was built. We know that the lab was conducting high-end research with the Chinese military. The French were in that lab and were eventually kicked out. And at the time that they were kicked out, they were aware that the Chinese military had moved into that lab. So there were circumstances created around that facility, and there were operating procedures around that facility that created a lot of risk. That doesn't mean it came out of the lab, but it certainly makes that a suspect location. And there was another lab. The CDC, the Chinese CDC maintained a lab literally blocks from the wet market that was first implicated in this virus that was also conducting coronavirus research in BSL-2 labs.
Q. So other than the whodunit factor, why does it matter? Why do we need to know who patient 0 is? And how will that affect the Biden administration's response?
A. Yeah, it matters a lot because if we determine-- first of all, if we determine that this came out of a lab or we even assess that there's a high probability that this came out of a lab, I think it changes how we try to govern research internationally. We're going to need something like the International Atomic Energy Agency for BSL 4 labs. We're going to have to look much more carefully about who's creating BSL 4 labs, the kind of research going on in those labs.
Q. On the global response, you wrote, COVID crushed the global order of public health cooperation. Are you saying WHO is just not up to the task?
A. Well, it's not just the WHO. I mean, past epidemics crushed the global order of cooperation. We just didn't learn the lesson.
Q. Was there quashing of information by the WHO?
A. I think the WHO, for too long--
Q. Did they cover up for China, is the allegation.
A. They weren't willing to confront China, right? They put out statements extolling China's behavior and how forthcoming China was. I mean, it's part of the record now. Tedros, it's in his Twitter feed, it's in the Twitter feed of the head of the WHO giving props to China for how forthcoming they were. Now, was some of that an attempt to try to give them public praise because privately you were giving them a harder sell? I think some of that was going on in the US with some of the tweets and the statements that we were putting out. I don't know that that was happening at the WHO level because I was talking to some WHO officials at the time and have talked to a number since. And I think the WHO really did believe China was behaving in sort of an appropriate way and was providing cover for them as they were getting criticized by other parts of the world. Clearly, they weren't. And I think that that was knowable at the time. I mean, China didn't share the source strains. Tedros of WHO didn't want to push China on sharing the source strains publicly because he said, well, they have no commitment to do it. And he's right. Under the International Health Regulations, there was no requirement that you had to share the samples. But there were requirements that you had to share samples of novel emerging respiratory pathogens and other settings. So clearly, the spirit of the International Health Regulations was that this should be shared. But because it wasn't the letter of the law, the WHO didn't want to push China publicly to do it, even though that would have been very helpful for other countries.
Q. You're talking about actual viral samples not the sequence--
A. Right. Actual samples of the virus because the sequence alone isn't good enough if you want to develop a vaccine or a diagnostic. You actually at some point need the live virus. Now, we eventually got the live virus because we had spread here in the United States but, you know, at least publicly we didn't have the virus until there was a case in Seattle. It's possible we got the virus earlier from one of China's neighbors where there was some earlier spread.
Q. That part of the equation being dealt with now?
A. No, I don't think the WHO has taken any steps to reform how it operates in a global crisis like this. Whether or not they're able to-- whether or not the organization has a self-awareness to do that and is able to self-organize to actually implement meaningful reform, I'm not sure. I think the Biden administration lost an opportunity to compel different behavior at the WHO by simply rejoining the WHO. I was critical at the time that President Trump made the decision to pull out of the WHO, I had actually spoken with him directly, urging him not to do it, suggested that there were other ways he could send a strong message to China short of pulling out of the WHO. But once we pulled out of the WHO, and we were going to re-enter, we should have used the reentry to extract some kind of agreement about the WHO engaging in some reform process. And as best I know, and as best we all know based on what's public, that wasn't done. The leverage was given up.
SEARCHABLE NIH / NIAID DOCS SEARCHABLE NIH / NIAID DOCS 2
A group of virologists, many of whom receive funding from Fauci's NIH, or as we say in the venacular, were on Fauci's pad, wrote: "The suspicion that SARS-CoV-2 might have a laboratory origin stems from the coincidence that it was first detected in a city that houses a major virological laboratory that studies coronaviruses." They claim it started in the Wet Market there. A large city in China typically has a few hundred wet markets. Why this particular one near the WIV? If the disease came from rural China, as Daszak suggests, why didn't anyone get infected along the way? But because Trump the Hump said the pandemic was caused by a lab leak, it became a partisan issue.
Claims it has nothing to do with his approval of Daszak's experiments! Ha! Ha! The Human Genome Project, which James Watson helped initiate in 1988, assembled a composite 'reference' genome of DNA from many individuals. Francis Collins was NIH director part of the time the study was conducted and did not participate in the actual research. Watson, who discovered DNAs double helix structure alongside Francis Crick and Rosalind Franklin in the 1950s, said that genes cause a difference on IQ tests between blacks and whites. African-Americans represent just 1.5 percent of grant applications to the N.I.H. which Collins headed, but none of these grant applications involved Gain-of-Threat research, so what race are stupid, evil ones from? Scientists and public health experts have criticized [Collins] efforts at studying Covid-19 treatments, saying some that ultimately did not show strong results such as convalescent plasma and hydroxychloroquine were prioritized over more rigorous research. NYT
GUPTA: These exchanges seem to assert that U.S. taxpayer dollars, via the NIH through your organization went to the Wuhan Institute of Virology and funded gain of function research.
DASZAK: Yes.
GUPTA: Which may, not definitively by any means, but may have been a source of this pandemic. What do you say?
DASZAK: That's just plain not true. Dr. Fauci testified publicly under oath that that's not true and he's right. We didn't do work that run against any of the rules.
Daszak knew they were being conducted in a leaky BSL-2 lab because he had been there many times in person. Was Daszak just negligent, or is there something sinister going on here? The answer lies in Entire EcoFascist Manifesto he co-authored along with his close associate Aleksei Chmura.
If the fallen angels [in the BRUEGEL painting] represent the evil mirror image of St. Michael and his cohort, so they also represent the mirror image of our own genetic kindpathogenic organisms which are otherwise just like us, but have fallen from grace through an evolutionary (not spiritual) pathway that takes them to a netherworld where they can feed only on our genes, our cells, our flesh...In Bruegels curious chimeras, we find yet another analogyfor the genetic recombination, mutation and evolution that negative-stranded RNA viruses in particular undergo as they shift, morph and adapt to their changing habitatanimal cells... Perhaps he reminds us also that it is the nature of our interactions with wildlifehere represented as an evil entity for humanity to vanquishwhich cause new zoonoses to emerge in the first place. Perhaps the EcoHealth view is that if we tread carefully, we might avoid those nasty little pincers waiting to nip! There was no COVID conspiracy: Daszak manipulated several unwitting key figures and a SARS virus found in bats was genetically enginered to be a novel Covid virus or a natural virus, that shouldn't have been in the BSL-2 lab, escaped. The original SARS already had caused a minor pandemic: SARS surfaced in the fall of 2002 in Guangdong Province, China. The original SARS-CoV spread worldwide within a few months, though it was quickly contained. SARS-CoV was shown to be transmitted from civet cats to humans. In 2015 Baric and Shi Zheng Li discovered the civets contracted the virus from bats, then authored a scientific peer-reviewed paper that stated that virologists better keep a close watch on the bat viruses in China because they were poised to spill over and cause a pandemic. Daszak is not listed in this paper which was credible. The Chinese China built the BSL-4 the Wuhan Institute of Virology (WIV) in 1956 but didn't give them the BSL-4 Lab, the highest bio-security lab, until 2018 because of dual use biowarfare concerns. The BSL-4 Lab is for diseases for which there is no known vaccine, cure or therapeutic. The gain-of-threat experiments should have been conducted there. This aspect brought the MIT Tech Review to it's senses about "conspiracy theory" being an excuse for a lab leak:
The CDC recognizes four levels of biosafety and recommends which pathogens should be studied at which level. Biosafety level 1 is for nonhazardous organisms and requires virtually no precautions: wear a lab coat and gloves as needed. BSL-2 is for moderately hazardous pathogens that are already endemic in the area, and relatively mild interventions are indicated: close the door, wear eye protection, dispose of waste materials in an autoclave. BSL-3 is where things get serious. Its for pathogens that can cause serious disease through respiratory transmission, such as influenza and SARS, and the associated protocols include multiple barriers to escape. Labs are walled off by two sets of self-closing, locking doors; air is filtered; personnel use full PPE and N95 masks and are under medical surveillance. BSL-4 is for the baddest of the baddies, such as Ebola and Marburg: full moon suits and dedicated air systems are added to the arsenal.
There are no enforceable standards of what you should and shouldnt do. Its up to the individual countries, institutions, and scientists. Dr. Daszakstein's unwitting assistant was Ralph "Igor" Baric, a former college swim team champ, a micro-biologist who specializes in Gain-of-Threat experiments. Just as Igor erred and stole the brain of a criminal rather than a normal person's brain for Dr. Frankenstein's experiments Baric erred by teaching the Bat Lady recombinant experiments that may have led to the creation of a highly contagious virus, not a vaccine, assuming that a natural bat virus didn't escape the WIV.
Ralph Steven Baric, (born 1954) Daszak's former unwitting accomplise, who believed he was working to develop a vaccine, Dr. Ralph Baric, stated: "The experiments that they did under the conditions that they did them were risky. They're working on bats SARS like viruses that have never been shown to cause human infection. Well, there are some scientists speaking up, that can't rule out the possibility of a lab leak. Their papers indicate that they did much of their work with these bat viruses under biological safety two conditions. There are many more laboratory accidents or laboratory acquired infections BSL-2 as compared to BSL-3. We do all the research in our lab on bat related coronaviruses under biological safety three enhanced conditions. We wear portable air breathing apparatuses with tied back suits so the workers are protected from anything that might be in the laboratory." Baric was able to increase the virulence of viruses by cutting their RNA into strips and altering each individual piece of genetic material. Baric was also known for his senseless experiments such as the creation of "no-see-um" viruses, through passaging1, that showed no traces of genetic manipulation. This technique, which was taught to Shi Zheng Li thanks to Daszak, was instrumental in making it difficult to determine the lab origin SARS-CoV-19, since virus was passaged to make it infect new hosts in an effort to determine which virus will spillover to humans. Baric also developed a method to bring viruses of the past, such as polio, back, through reverse genetic engineering. So a virus could never be wiped off the planet as long as its nucleic code existed. Nucleic acid is a complex organic substance present in living cells, especially DNA or RNA, whose molecules consist of many nucleotides linked in a long chain. Baric once gave a lecture on how to profit from a pandemic. Baric erred and caused a minor SARS outbreak in North Carolina 2003. Look at the document. Later, one of his SARS-infected lab rats bit a student's hand. Baric applied for patents for his inventions and had a large staff at the University of North Carolina. Baric visited the WIV and the Bat Lady visited his lab - thanks to Daszak bringing them together. It should be noted that "although GoF experiments can make a virus more deadly and transmittable many gain-of-function experiments were worthwhile. In 1937, researchers found that when they passed the yellow fever virus through chicken cells, it lost the ability to cause disease in humans a discovery that led to a vaccine for yellow fever." But Baric's creations were often pointless. Baric's base salary is $190,300.00 a year.
At a regular basis, Dr. Baric will report the results of the teams research to Dr. Daszak and Dr. Shi and together, they will use this information to identify additional research priorities and design downstream studies. Ors. Daszak, Shi and Barie have published together in the past and participated on research project applications. Dr. Barie recently spent several days in Wuhan, China, where he discussedresearch strategies and collaborations with Dr. Daszak and Dr. Shi. He will work closely with Dr. Sims and Mr. Yount to prepare timely reports, share research and discuss future research directions with the group. Baric Sub-award Baric's base salary is $190,300.00 a year.
1. Passaging is the placing of a live virus into an animal or cell culture to which it is not adapted and then, before the virus dies out, transferring it to another animal or cell of the same type. Passaging is often done iteratively. The theory is that the virus will rapidly evolve (since viruses have high mutation rates) and become adapted to the new animal or cell type. Passaging a virus, by allowing it to become adapted to its new situation, creates a new pathogen. SCIMEX
They're working on bats SARS like viruses that have never been shown to cause human infection. This means they are enhancing them to do so - this is known as "Gain-of-Threat."
MIT TECHNOLOGY REVIEW puts a lot of blame on Ralph Baric. However Baric's father was not a Nazi death camp executioner, like Daszak's father Bohdan, who justified his crimes to his son by inculcating him with Nazi ideology. Evil runs in Daszak's blood. Daszak's father, already a Ukrainian Nazi, voluneered to become a death camp executioner.
Ralph Baric, although at least one of his experiments seemed pointless, like developing a method that made it impossible to discern a artifical virus from a natural one, never funneled any of his USG grant money to the WIV like Daszak did but he collaborated with it. Baric did give a lecture titled "How to Profit from the Next Pandemic." Baric was a cowboy and out of control. His lab was known as "The Wild West." His experiments may have caused a minor outbreak of SARS CoV-1 in Chapel Hill, North Carolina in 1983. His experiments must be reviewed to determine if they were a threat to humanity. For 20 years, beneath the radar, Baric had been playing with millions of people's lives. In Wuhan, China and Chapel Hill, North Carolina, Daszak, Dr. Shi Zheng Li collaborated with Dr. Ralph Baric, and routinely created viruses more dangerous than those that exist in nature.
Dr. Baric had developed, and taught Dr. Shi a general method for engineering bat coronaviruses to attack other species. What was worse, Baric had developed a method of genetic engineering wherein it was impossible to determine if the virus had been altered, which he called the "no-see-um" technique after the pesky little, almost invisible, flies found at various beaches in the South. The point of doing this escapes me? The specific targets were human cells grown in cultures and humanized mice. These laboratory mice are genetically engineered to carry the human version of a protein called ACE2 that studs the surface of cells that line the lungs. The "Bat Lady" flew back to her lab at the Wuhan Institute of Virology (WIV) and resumed the work she had started on genetically engineering coronaviruses to attack human cells using this technique that she learned from Dr. Baric. Her genetically altered virus, enhanced to attack human beings, escaped. It wasn't the first time a genetically altered SARS virus leaked. Evidence suggests that in 2003 a less virulent SARS virus escaped from Baric's North Carolina lab causing several infections.
When you are dealing Baric remember one thing. The University of North Carolina School of Medicine, ranked 17th in the country in total National Institutes of Health (NIH) funding for federal fiscal year 2020 and 5th among peer public universities, according the Blue Ridge Institute for Medical Research. The total dollar amount is $345,729,473, a jump of more than $29 million from FY 2019. This amount does not include research and development contracts with the NIH. The UNC Gillings School of Global Public Health, which is part of UNC, where Ralph Baric works, is ranked 3rd in total NIH funding, 1st among public universities. Fauci has been his boss for decades. Baric signed on to the email calling for new Covid-19 investigation and didn't sign Daszak's email supporting the Zoonotic theory to disguise his part in the pandemic.
2002 Dr. Baric announced that he found a way to create a full-length infectious clone of the entire mouse-hepatitis genome. His infectious construct replicated itself just like the real thing, he wrote. Not only that, but hed figured out how to perform their assembly seamlessly, without any signs of human handiwork. Nobody would know if the virus had been fabricated in a laboratory or grown in nature. Baric called this the no-seem method, and he asserted that it had broad and largely unappreciated molecular biology applications. A. Professional Development/Invited Presentations
Invited Speaker: Engineering the Genomes of Microorganisms. DARPA Meeting on
Synthetic Biology, Menlo Park, California. March 2003.
Invited Speaker: Coronavirus Vaccines. NIAID. SARS: Developing a Research Response, May 30, 2003
Coronavirus Reverse Genetics. Layola University School of Medicine, March 2003.
Grant Review 2003
1. Genetics study section FEBRUARY and OCTOBER, 2003. Ad hoc.
2. Experimental Virology, FEBRUARY, 2003. Ad hoc
3. NIH ad hoc review, Poxvirus vaccine program project. SEPTEMBER 2003. The Baric laboratory has pioneered reverse genetic analyses of CoVs and DENVs. Several CoV infectious cDNA clones are available in the lab, including SARS-CoV, MERS-CoV, conventional human and model CoVs, and several bat Co Vs with pandemic potential. The availability of these genetic platforms allows for detailed studies into the role of viral genes in pathogenesis, innate immune antiviral immunity, vaccine performance and design, virus-receptor interactions, entry and virus evolution. The CDC reported: "DECEMBER 31, 2003 In the United States, EIGHT SARS infections were documented by laboratory testing and an additional 19 probable SARS infections were reported. The North Carolina Department of Health and Human Services (DHHS) March 19, 2003: Two possible SARS cases reported in N.C., one in WAKE COUNTY [where Baric's UNC lab is located] and one in ORANGE COUNTY. Both cases subsequently tested negative. April 1, 2003: Third and fourth cases of suspected SARS reported, (one in WAKE COUNTY and one in Mecklenburg County). Both cases subsequently tested negative. April 8, 2003: Fifth case of suspected SARS reported (Iredell County). Case subsequently tested negative. April 9, 2003: Sixth suspected SARS case reported (WAKE COUNTY). Case subsequently tested negative. April 21, 2003: Seventh suspected SARS case reported (WAKE COUNTY). Case subsequently tested negative. April 28, 2003: EIGHTH suspected SARS case (WAKE COUNTY). Case tested negative. June 3, 2003: North Carolina reported its ninth case of suspected SARS, an ORANGE COUNTY man, to the Centers for Disease Control and Prevention (CDC). He was reported as probable on June 6, 2003, and then as a confirmed case of SARS on June 9, 2003, when CDC laboratory tests were positive. By the time North Carolina reported its confirmed case in June 2003, that case was the eighth laboratory-confirmed case of SARS in the United States. No further confirmed cases were reported in the U.S., and the transmission of SARS-CoV was declared contained in July 2003. APRIL 28, 2003: "North Carolina has reported its EIGHTH case of suspected SARS to the Centers for Disease Control and Prevention. The patient is a WAKE COUNTY woman who developed a fever and respiratory symptoms after returning to WAKE COUNTY from a trip to TORONTO, CANADA. The patient is recovering and is under voluntary isolation at home. THE OTHER SEVEN SUSPECTED CASES OF SARS IN NORTH CAROLINA HAVE ALL NOW RECOVERED FROM THEIR SARS-RELATED SYMPTOMS and may return to their normal activities. June 3, 2003: North Carolina reported its ninth case of suspected SARS to the Centers for Disease Control and Prevention (CDC). The patient is an ORANGE COUNTY man who developed symptoms after returning to North Carolina from a trip to TORONTO, CANADA. He is being treated as an outpatient and is recovering at home under isolation. SARS-CoV infected around 8000 individuals worldwide with an overall mortality of 10% during the 20022003 pandemic. Two of the North Carolina cases came from the Toronto outbreak cased by a woman who returned from Hong Kong but what about the others? As of May 7, 2003 a total of 328 SARS cases in the United States were reported from 38 states, of which 265 (81%) were classified as suspect SARS, and 63 (19%) were classified as probable SARS (more severe illnesses). There seems to be a disproportionate number of cases in North Carolina but due to medical privacy concerns we will never know if they were in or around Chapel Hill? (one case was reported there) NOVEMBER 2015 "Utilizing the SARS-CoV infectious clone, we generated and characterized a chimeric virus expressing the spike of bat coronavirus SHC014 in a mouse adapted SARS-CoV backbone. The results indicate that group 2b viruses encoding the SHC014 spike in a wild type backbone can efficiently utilize multiple ACE2 receptor orthologs1 , replicate efficiently in primary human airway cells, and achieve in vitro titers2 equivalent to epidemic strains of SARS-CoV. Additionally, in vivo experiments demonstrate replication of the chimeric virus in mouse lung with notable pathogenesis. Evaluation of available SARS-based immune-therapeutic and prophylactic modalities revealed poor efficacy; both monoclonal antibody and vaccine approaches failed to neutralize and protect from CoVs utilizing the novel spike protein. Importantly, based on these findings, we synthetically rederived an infectious full length SHC014 recombinant virus and demonstrate robust viral replication both in vitro and in vivo. Together, the work highlights a continued risk of SARS-CoV reemergence from viruses currently circulating in bat populations."....While offering preparation against future emerging viruses, this approach must be considered in the context of the US government-mandated pause on GoF/GoT studies. Based on previous models of emergence the creation of chimeric viruses like SHC014-MA15 was not expected to increase pathogenicity. However, while SHC014-MA15 is attenuated relative to parental mouse adapted, equivalent studies examining the wild-type Urbani spike within the MA15 backbone produced no weight loss and replication attenuation. As such, relative to the Urbani Spike-MA15 CoV, SHC014 MA15 constitutes a gain in pathogenesis. Based on these findings, review panels may deem similar studies too risky to pursue as increased pathogenicity in mammalian models cannot be excluded. Coupled with restrictions on mouse adapted strains and monoclonal antibodies generated against escape mutants, research into CoV emergence and therapeutic efficacy may be severely limited moving forward. Together, these data and restrictions represent a crossroads of GoF/GoT research concerns; the potential to prepare and mitigate future outbreaks must be weighed against the risk of creating more dangerous pathogens. In developing policies moving forward, it is important to consider the value of the data generated by these studies and if they warrant further study or the inherent risks involved.
1. Orthologs are genes in different species that evolved from a common ancestral gene by speciation, and, in general, orthologs retain the same function during the course of evolution.
2. The concentration of an antibody, as determined by finding the highest dilution at which it is still able to cause agglutination (the clumping together in suspension of antigen-bearing cells in the presence of specific antibodies of the antigen).
Reported studies were initiated after the University of North Carolina Institutional Biosafety Committee approved the experimental protocol: PROJECT TITLE: GENERATING INFECTIOUS CLONES OF BAT SARS-LIKE COVS; LAB SAFETY PLAN ID: 20145741; SCHEDULE G ID: 12279. These studies were initiated prior to the U.S. Government Deliberative Process Research Funding Pause on Selected GoF/GoT Research Involving Influenza, MERS, and SARS Viruses and the current manuscript has been reviewed by the funding agency, the National Institutes of Health (NIH). Continuation of these studies have been requested and approved by NIH. [Fauci]
FULL STORY "Researchers from the University of North Carolina at Chapel Hill have discovered a new bat SARS-like virus that can jump directly from its bat hosts to humans without mutation. However, researchers point out that if the SARS-like virus did jump, it is still unclear whether it could spread from human to human. The discovery, reported in the November 9, 2015 issue of Nature Medicine, is notable not only because there is no treatment for this newly discovered virus, but also because it highlights an ongoing debate over the government's decision to suspend all gain-of- function experiments on a variety of select agents earlier this year. The move has put a substantial standstill on the development of vaccines or treatments for these pathogens should there be an outbreak."Studies have predicted the existence of nearly 5,000 coronaviruses in bat populations and some of these have the potential to emerge as human pathogens," said senior author Ralph Baric, a faculty member at the Gillings School of Global Public Health and expert in coronaviruses. "So this is not a situation of if there will be an outbreak of one of these coronaviruses but rather when and how prepared we'll be to address it." SARS first jumped from animals to humans in 2002-2003 and caused a worldwide outbreak, resulting in 8,000 cases, including one case in Chapel Hill. With nearly 800 deaths during that outbreak, SARS-CoV presents much like flu symptoms but then can accelerate, compromise breathing and bring on a deadly form of pneumonia. The outbreak was controlled through public health interventions and the original virus was thought to have been extinct since 2004. Baric and his team demonstrated that the newly-identified SARS-like virus, labeled SHC014-CoV and found in the Chinese horseshoe bats, can jump between bats and humans by showing that the virus can latch onto and use the same human and bat receptor for entry. [ACE2] The virus also replicates as well as SARS-CoV in primary human lung cells, the preferred target for infection. "This virus is highly pathogenic and treatments developed against the original SARS virus in 2002 and the ZMapp drugs used to fight Ebola fail to neutralize and control this particular virus," said Baric. "So building resources, rather than limiting them, to both examine animal populations for new threats and develop therapeutics is key for limiting future outbreaks." Journal Reference: Vineet D Menachery, Boyd L Yount, Kari Debbink, Sudhakar Agnihothram, Lisa E Gralinski, Jessica A Plante, Rachel L Graham, Trevor Scobey, Xing-Yi Ge, Eric F Donaldson, Scott H Randell, Antonio Lanzavecchia, Wayne A Marasco, SHI ZHENGLI-LI Ralph S Baric. A SARS-like cluster of circulating bat coronaviruses shows potential for human emergence. Nature Medicine, 2015; DOI: 10.1038/nm.3985
by Jonathan Latham, PhD
University researchers genetically engineer a human pandemic virus. They inject the new virus into a laboratory mouse. The infected mouse then bites a researcher
..It is a plot worthy of a Hollywood blockbuster about risky coronavirus research. But according to newly obtained minutes of the Institutional Biosafety Committee (IBC) of the University of North Carolina (UNC), Chapel Hill, these exact events need not be imagined. They occurred for real between April 1st and May 6th this year. The identity of the bitten coronavirus researcher has not been revealed except that they were working in a high security BSL-3 virus lab when the accident happened. Other researchers are also calling for restraint. In a paper titled Prudently conduct the engineering and synthesis of the SARS-CoV-2 virus, researchers from China and the US critiqued the synthesis in February of a full length infectious clone (Gao et al., 2020; Thao et al., 2020). And, in concluding, these researchers asked a question that is even more pertinent now than then Once the risks [of a lab escape] become a reality, who or which organization should take responsibility for them? The accident at the University of North Carolina (UNC) is now in the public domain but only thanks to a FOIA request submitted by Hammond (in line with NIH guidelines) and shared with Independent Science News. espite the FOIA request, apart from the fact that UNC classified it as an official Reportable Incident, i.e. that must be reported to National Institutes of Health (NIH) in Washington DC, scarcely any information about the accident is available.In part this is because the minutes of the relevant IBC meeting (May 6th, 2020, p109) are extremely brief. They do not provide any details of the fate of the bitten researcher. Nor do they state, for example, whether the researcher developed an active infection, nor whether they developed symptoms, nor if they transmitted the recombinant virus to anyone else. Neither do they reveal what kind of recombinant virus was being used or the purpose of the experiment. To try to learn more, Independent Science News emailed the lab of Ralph Baric at UNC, which, based on their research history is the most likely coronavirus research group involved (Roberts et al., 2007; Menachery et al., 2015), the University Biosafety Officer, and UNC Media relations. Only the latter replied: The April 2020 incident referred to in the University Institutional Biosafety Committee meeting minutes involved a mouse-adapted SARS-CoV-2 strain used in the development of a mouse model system.
At the dawn of recombinant DNA, at the request of the scientific community itself, following the fabled Asilomar conference, the United States government took the position of not regulating genetic engineering in the lab. The deal that big science struck with the government was that, in return for not being directly regulated, principal investigators would take personal responsibility for lab biosafety, involve the public in decision-making, and accept public accountability for their actions. The NIH Guidelines and Institutional Biosafety Committee system of self-regulation by researchers is founded upon the principal of personal responsibility of PIs and the promise of transparency. The redaction of the researchers identities from IBC meeting minutes, in order to hide the activities of researchers and avoid accountability for accidents, fundamentally contradicts the core principles of the US oversight system and violates the commitments that science made. There is no justification for UNCs redaction of the names of the laboratory heads and the identities of pathogens. UNCs redactions violate conditions UNC agreed to in exchange for NIH funding of UNCs research and, if not corrected, should result in the termination of current NIH funding, and the loss of eligibility for future NIH funding, of UNCs research.
This concern over risks and benefits is shared by Edward Hammond. Using FOIA again he has further discovered that researchers at the University of Pittsburgh (whose identity is redacted) plan to make what Hammond calls Corona-thrax. In short, according to its Institutional Biosafety Committee, Pittsburgh researchers intend put the spike protein of SARS-CoV-2 (which allows the virus to gain entry into human cells) into Bacillus anthracis which is the causative agent of anthrax. The anthrax strain proposed to be used for this experiment is disarmed but, Hammond agrees with Gao et al., (2020) that the balance of risks and benefits appears not to be receiving adequate consideration. This experiment was nevertheless approved by the Institutional Biosafety Committee of the University of Pittsburgh. But by redacting the name of the laboratory from the minutes and also every name of the members of the committee which approved it, the University has supplied a de facto response to the final question posed by Gao et al.: who will take responsibility for risky coronavirus research?
The accident at the University of North Carolina (UNC) is now in the public domain but only thanks to a FOIA request submitted by Hammond (in line with NIH guidelines) and shared with Independent Science News. Despite the FOIA request, apart from the fact that UNC classified it as an official “Reportable Incident”, i.e. that must be reported to National Institutes of Health (NIH) in Washington DC, scarcely any information about the accident is available. In part this is because the minutes are extremely brief. They do not provide any details of the fate of the bitten researcher. Nor do they state, for example, whether the researcher developed an active infection, nor whether they developed symptoms, nor if they transmitted the recombinant virus to anyone else. Neither do they reveal what kind of recombinant virus was being used or the purpose of the experiment.
To try to learn more, Independent Science News emailed the lab of Ralph Baric at UNC, which, based on their research history is the most likely coronavirus research group involved (Roberts et al., 2007; Menachery et al., 2015), the University Biosafety Officer, and UNC Media relations. Only the latter replied:
“The April 2020 incident referred to in the University Institutional Biosafety Committee meeting minutes involved a mouse-adapted SARS-CoV-2 strain used in the development of a mouse model system.” UNC media relations also told Independent Science News that: “The researcher did not develop any symptoms and no infection occurred as a result of the incident.” Our questions in full and the full UNC reply are available here.
The second reason for this lack of information is that the UNC redacted the names of Principal Investigators (PIs) whose research required biosafety scrutiny, along with many of the experimental specifics. Nevertheless, unredacted parts of minutes from IBC meetings held in 2020 contain descriptions of experiments that potentially encompass the accident. They include:
Application 75223:
In all, any one of eight sets of different experiments approved by the UNC Chapel Hill IBC in 2020 proposed infecting mice with live infectious and mutant SARS-CoV-2-like coronaviruses under BSL-3 conditions and therefore could have led to the accident.
MAY 2021 Baric I really believe that the genetic sequence for sars-CoV-2 really points to a natural-origin event from wildlife, Ralph Baric said that sars-CoV-2 was different enough from known viruses that to retrofit it from an ancestral strain would have required a truly unprecedented feat of genetic reëngineering. And of course you dont know what youre engineering, because SARS-CoV-2 would not have existed." Another possibility was that a virus nearly identical to the eventual pathogen, which had been collected in the wild and stored in the virus repository, had somehow escaped containment, but he hadnt seen any specific evidence to support that hypothesis, either. All that is known for sure is that a pathogen that likely originated in bats had transferred species and caused a global pandemic in human beings, perhaps in a way that had only happened a few times in modern medical history or in a waythrough a labthat may never have happened before. Barics preference was for the theory that resembled previous outbreaks." [Of course Baric would say this since he taught Shi Zhengli how to create viruses in the lab that could not be distinguished from those in nature and so he bears responsibility for the pandemic.]
At University of Iowa, scientist Stanley Perlman launched work for the deadly MERS virus without faculty approval, the Des Moines Register reported in December 2014. Worse still, Perlmans team conducted the MERS research in a biosafety level-2 lab, instead of a level-3 facility as is required by federal regulators, it said. GLOBAL TIMES Dr. Shi Zheng Li, "The pandemic is nature's punishment for an unsanitary lifestyle. So shut your stinky mouths. How on earth can I offer up evidence for something where there is no evidence? I dont know how the world has come to this, constantly pouring filth on an innocent scientist. My lab has never conducted or cooperated in conducting GOF experiments that enhance the virulence of viruses. [Just the transmissability] This is no longer a question of science. It is speculation rooted in utter distrust. Its really sad to read this Letter written by these 18 prominent scientists. The hypothesis of a lab leaking is just based on the expertise of a lab which has long been working on bat coronaviruses which are phylogenetically related to SARS-CoV-2. This kind of claim will definitely damage the reputation and enthusiasm of scientists who are dedicated to work on the novel animal viruses which have potential spillover risk to human populations and eventually weaken the ability of humans to prevent the next pandemic...Another piece of evidence that I can give you is that our lab has been doing research for 15 years, and all our work has been published. We also have a library of our own genetic sequences, and we have experimental records of all our work related to the virus, which are accessible for people to check Dr. Zhengli Shi, Senior Virologist. Dr. Shi will oversee the coronavirus screening for all samples collected in China. Novel SARS-like coronaviruses were detected in Rhinolophus bats collected in different regions of Guangdong province. Diverse novel betacoronaviruses related to HKU5 were detected in Pipistrellus bats and laio in Guangdong and in Asel/iscus stoliczkanus in Mengla, Yunnan. Novel coronaviruses related to HKU9 were found in Cynopterus sphinx and Rousettus leschenaulti in Mengla. In addition, sequences significantly divergent to other CoV were obtained from three samples of /a io and Hipposideros bats. "Bats: Depending on the species and size of bat, swabs wil l be taken from the oropharynx, urogenital tract, and rectum. Fresh feces will be collected if available, in which case a rectal swab will not be collected. Blood will be collected from fruit bats either from the cephalic vein or from the radial artery or vein using a 25 gauge needle and 1 cc syringe. Blood will be collected from bats weighing less than 100g according to published techniques. Principal Investigator/Program Director: Daszak, Peter" INTERCEPT DOC page 72, 167, 124
Daszak's dupe at the WIV was Shi Zheng Li, the director of Bio-Safety. In January 2018, the State Department had visited her lab after reports of unsafe conditions. During interactions with scientists at the WIV laboratory, they noted the new lab has a serious shortage of appropriately trained technicians and investigators needed to safely operate this high-containment laboratory, the researchers also showed that various SARS-like coronaviruses can interact with ACE2, the human receptor identified for SARS-coronavirus." Were the unsafe conditions the fact that the corona virus procedures were being carried out in a low level lab? Doctor Daszak knew the young Technicians and scientists were incompetent and sloppy. He knew Shi Zheng Li, "The Bat Lady" was a weirdo. Interestingly enough Shi stated that no experiments were conducted at the WIV to make the virus more deadly but never addressed the experiments that made it more contagious. Daszak put her in touch with the genius Dr. Ralph Baric and, with him, she co-authored a paper on SARS-1 coming from bats. Baric developed a method to alter the RNA in a virus by cutting the strands in seperate sections then re-combining them with other viral RNA. This man is more dangerous than Daszak because he has the ability to make a virus more lethal, but his motivation is to create vaccines, not a pandemic. In summation, Daszak who was at the epi-center of all this activity, knew how risky to humanity these WIV activities were and made sure they were conducted.
REPORTER: 800,000 coronavirus deaths do you have a statement on your responsibility? And why havent you asked China to do more, to be transparent on the origins? MAY 13, 2020: Two Wuhan Institute of Virology scientists, Xing-Yi Ge and Zhengli-Li Shi, use reverse genetics to generate a chimeric virus (one that has been created by combining cells of more than one distinct genotype) closely resembling the novel coronavirus Covid-19:On the basis of these findings, we synthetically re-derived an infectious full-length SHC014 recombinant virus and demonstrate robust viral replication both in vitro and in vivo. The Nature Medicine article also mentions, Human lungs for HAE cultures were procured under University of North Carolina at Chapel Hill Institutional Review Boardapproved protocols. The Chinese governments poor record of transparency; the fact that the Wuhan Institute of Virology, a research center with facilities in the same city where the virus first appeared, was studying and creating new dangerous pathogens, including bat coronaviruses cannot be easily dismissed.
If the virus leaked out of a Chinese lab, every one through four Bio-Safety Level laboratory in the U.S. and Europe will be shut down and forced to move to more isolated locations. In 2010, by one count, there were 249 BSL-3 laboratories and seven BSL-4 laboratories in the U.S., and more than 11,000 scientists and staffers were authorized to handle the ultralethal germs on the governments select pathogen list. Jamie Metzl of the Asia Society: If zoonosis was the origin, it was a validatin of Dr. Daszak's life work. But if the pandemic started as part of a lab leak, it had the potential to do to virology what Three Mile Island and Chernobyl did to nuclear science. It could mire the field indefinitely in moratoriums and funding restrictions." So many virologists are circling the wagons around Fauci because of this and because he is the one the issues them their research grants.
In January 2021 the CCP called her "a non-party person" which is highly unlikey, considering the status of the position she reached. The CCP did this in order to distance it from her in case the truth surfaces. Chmura was educated at Kingston University Academic degree Doctor of Philosophy start time September 1, 2012 end time June 1, 2018; Columbia University academic degree Bachelor of Science Start time September 1, 2001 end time May 15, 2004. I call him Chumpura.
This is from a book that mentions Chumpura: "Quammen then narrates the race to identify the SARS agent and its (still) elusive reservoir host. Finally he joins Aleksei Chmura, an American virus hunter-cum-global gourmand. As a prelude to their fieldwork, the two men bond over clotted pigs blood, bean sprouts, red peppers, and drippy globs of durian, the worlds stinkiest fruit. Then its offminus protective respiratory gearto netting bats in the karst mountains near Guilin and later phlebotomizing them. Wrapping the chapter is an excursion to a cinderblock billet in southern China where Farmer Wei Shangzheng raises placid bamboo rats headed for human dinner plates". [Am. J. Trop. Med. Hyg., 88(4), 2013, pp. 803805 doi:10.4269/ajtmh.13-0092 Copyright © 2013 by The American Society of Tropical Medicine and Hygiene Serological Evidence of Bat SARS-Related Coronavirus Infection in Humans, China. Wang N1, Li SY2, Yang XL1, Huang HM2, Zhang YJ1, Guo H1, Luo CM1, Miller M3, Zhu G3, Chmura AA3, Hagan E3, Zhou JH4, Zhang YZ4, Wang LF5, Daszak P3, Shi ZL1] They seem to go back quite a long way Daszak helped supervise his Thesis. They wrote a quite bizarre letter back in 2007 called
"A FALL FROM GRACE TO
VIRULENCE?"
In Bruegels painting of The Fall of the Rebel Angels we are witness to a tumbling maelstrom of falling rebel angels outcast from Heaven. Within the fray stands St. Michael1 in gilded armor, and his angels-at-arms serenely in pale albs, and almost as if threshing grain, hewing and striking down this inconceivable rout. The main focus of the image and what draws the eye is the extraordinarily creative mélange of creatures; mixtures of human, animal, plant, and inanimate objects slashing and stabbing as they fall from the great battlefields in the skies. They pour down in a vast column that stretches infinitely from the luminous sun; they fall from the light to the darkness. The column of falling angels is so numerous that it widens to encompass the whole lower canvas as it approaches the viewer. With a start, then, we realize that Bruegel intends that we too are in the thick of this. Will we succumb to the multitudinous horde? Are we to be cast downward into chthonic2 chaos represented here by the heaped up gibbering phantasmagory3 against which we rail and struggle?
1.Saint Michael is an archangel, a spiritual warrior in the battle of good versus evil. He is considered a champion of justice, a healer of the sick, and the guardian of the Church. In art Saint Michael is depicted with a sword, a banner, or scales, and is often shown vanquishing Satan in the form of a dragon.
2. Concerning, belonging to, or inhabiting the underworld.
3. A fantastic sequence of haphazardly associative imagery, as seen in dreams or fever.
Clearly Bruegel intended for us to identify with St. Michael and his comrades. The classic imagery of religious battles between humanity and evil, is straightforward fodder for us to digest, but is there another metaphor here? If the fallen angels [depicted in the Bruegel painting] represent the evil mirror image of St. Michael and his cohort, so they also represent the mirror image of our own genetic kindpathogenic organisms which are otherwise just like us, but have fallen from grace through an evolutionary (not spiritual) pathway that takes them to a netherworld where they can feed only on our genes, our cells, our flesh. On closer scrutiny, we can see that Bruegel has depicted the Natural Worldspecifically chosen it as proxy for the fallen angelsfor both its fascinating wonder and horror. Thus may we surmise that nature was, in Bruegels mind, itself as strange, wondrous, and horrific as the fallen angels that he depicted in the juxtaposed forms of otherwise anatomically correct fishes, bats, and frogs? Bruegel was an urban man and precisely because of his lack of intimate knowledge of Nature, he was likely able to observe it with such precision. No doubt he was influenced during time spent in France & Italy by that unique Renaissance creation, the scientific method, or means to learn about phenomena via empirical evidence. Today, we are not unlike Bruegel as we view the wondrously diverse animals and plants that represent nature, peering beneath this palimpsest the pathogens that threaten to plague us? AIDS, SARS, Malaria, West Nile Virus, and Avian Influenza are but a few of these fallen angels that have burst from out of the Natural world to wreck havoc causing countless deaths and economic losses.
In Bruegels curious chimeras, we find yet another analogyfor the genetic recombination, mutation and evolution that negative-stranded RNA viruses in particular undergo as they shift, morph and adapt to their changing habitatanimal cells. Here St. Michael is at risk of more than just the prodding of a sword. Here he is at the mercy of the tooth and nail of glycoproteins sharpened and honed to strike with precision through the gaps in his golden armor. As Pulliam shows in this issue of EcoHealth, this battle continues in reality as human encroachment into wildlife habitat, and the increasing globalization of agriculture, trade and travel bring us into dramatic juxtaposition with a seemingly infinite number and variety of viral angels hosted by the wildlife we contact and exploit. In this prescient scene, Bruegel reminds us that our battle against novel zoonotic pathogens is far from over: They lie in wait in a multitude of surely the same overwhelming dimensions as Bruegel's column of descending angels. Perhaps he reminds us also that it is the nature of our interactions with wildlifehere represented as an evil entity for humanity to vanquishwhich cause new zoonoses to emerge in the first place. Perhaps the EcoHealth view is that if we tread carefully, we might avoid those nasty little pincers waiting to nip! Another manifesto - this one decrying the Industrial Revolution
DASZAK grew up with a hard core Nazi, his father, Bohdan Daszak, a rabid Jew hating scum and former member the OUN Youth, (Organization of Ukrainian Nazis or "Nationalists"). Peter Daszak is the son-of a Nazi death camp executioner who volunteered to murder countless Jews, and like father like son. Daszak was indoctrinated in Nazi thought by this Nazi war criminal: While protecting flora, fauna, water, and soil, the Nazis also murdered millions of human beings. The Christchurch mass murderer manifesto: "I consider myself an Eco-fascist by nature. The nation with the closest political and social values to my own is the Peoples Republic of China." As documented here, Daszack is an agent-of-influence for the Chinese Communist Party, who led the W.H.O. cover-up. Daszak never says not a bad thing about China, including commenting on their human rights abuses, such as building concentration camps for the Uyghurs. Daszak was taught to respect fascism: like that of the CCP and his father had taught him concentration camps were okay. Daddy and China are keys to the fact that he is something other than what he pretends to be. Read what Facebook members comment about him!
GIVING A SARS-LIKE VIRUS THE ABILITY TO INFECT HUMANS INVOLVED RECOMBINANT BAT CORONAVIRUSES / NOVEMBER 30, 2017:"DISCOVERY OF A RICH GENE POOL OF BAT SARS-RELATED CORONAVIRUSES PROVIDES NEW INSIGHTS INTO THE ORIGIN OF SARS CORONAVIRUS"
MANY OF THESE GoF / GoT EXPERIMENTS WERE CONDUCTED WITH VIRUSES FOUND IN HORSESHOE BATS - CLOSE RELATIVES TO THE SARS-CoV-19 VIRUS
https://onlinelibrary.wiley.com/doi/full/10.1002/ame2.12017THE BAT VIRUS STORAGE AND EXPERIMENTS WERE CONDUCTED IN BSL-2 CONSTRUCTED IN 1956 THE ELVIS ERA
BEFORE THE CORONA VIRUS OUTBREAK IN 2019 THE GAIN-OF-FUNCTION, GAIN-OF-THREAT EXPERIMENTS WERE PERFORMED IN BSL-2 LAB

CHAPTER 3
THE VIRUS IS NOT FROM MOTHER NATURE: THE FBI CORROBORATES THE LAB LEAK THEORY
FORMER FDA HEAD SCOTT GOTTLIEB ALSO MAKES THE CASE FOR THE LAB LEAK THEORY

CHAPTER 4
FAUCI, NIH, STEMMY, DASZAK, COLLINS DOCUMENTS AND INVOLVEMENT IN THE PANDEMIC
FAUCI IS A PROPONENT OF THE ANIMAL SPILL OVER THEORY BECAUSE THE LAB LEAK ONE LEADS BACK TO HIM
FRANCIS "THE TALKING MULE" COLLINS RESIGNS

INVOLVEMENT IN THE PANDEMIC
MORONICAL MD FRANCIS "THE SINGING MULE" COLLINS CHANGES SONG ABOUT SMOKING OPIUM AS IN "CHASING THE DRAGON" INTO SONG ABOUT COVID
BENEFITS AND RISKS OF INFLUENZA RESEARCH: LESSONS LEARNED ANTHONY S. FAUCI, FRANCIS S. COLLINS
THE DOCUMENTS MAKE IT CLEAR THAT ASSERTIONS BY THE NIH DIRECTOR, FRANCIS COLLINS, AND THE NIAID DIRECTOR, ANTHONY FAUCI, THAT THE NIH DID NOT SUPPORT GAIN-OF-FUNCTION RESEARCH OR POTENTIAL PANDEMIC PATHOGEN ENHANCEMENT AT WIV ARE UNTRUTHFUL.
THE US NATIONAL INSTITUTES OF HEALTH (NIH) ANNOUNCED THAT THEY WOULD RESUME FUNDING GAIN-OF-FUNCTION EXPERIMENTS INVOLVING INFLUENZA, MIDDLE EAST RESPIRATORY SYNDROME CORONAVIRUS, AND SEVERE ACUTE RESPIRATORY SYNDROME CORONAVIRUS.
FEDERAL OFFICIALS ENDED A MORATORIUM IMPOSED THREE YEARS AGO ON FUNDING RESEARCH THAT ALTERS GERMS TO MAKE THEM MORE LETHAL
"WE DEBATED UP ONE SIDE DOWN THE OTHER, AND ULTIMATELY DECIDED IF YOU WERE A HUMAN TRYING TO DESIGN A REALLY DANGEROUS CORONAVIRUS, YOU WOULD NOT DESIGN THIS ONE."
EXEMPTION 5 IS COMMONLY INTERPRETED AS SHIELDING FROM RELEASE DRAFT GOVERNMENT DOCUMENTS, RECORDS OF SENSITIVE DELIBERATIONS BEFORE DECISIONS ARE MADE, AND GOVERNMENT ATTORNEY-CLIENT DELIBERATIONS.
FRANK COLLINS CLAIMS NEITHER NIH NOR NIAID HAVE EVER APPROVED ANY GRANT THAT WOULD HAVE SUPPORTED GAIN-OF-THREAT RESEARCH ON CORONAVIRUSES THAT WOULD HAVE INCREASED THEIR TRANSMISSIBILITY OR LETHALITY FOR HUMANS

ON SANJAY GUPTA'S CNN ORIGINS OF COVID DASZAK SAID IN THE VENACULAR: "FAUCI HE BE MY HOMEBOY"
CHAPTER 5
KEY UNWITTING PERSONNEL

RALPH STEVEN BARIC



BARIC: You can engineer a virus without leaving any trace. The answers you are looking for, however, can only be found in the archives of the Wuhan laboratory.

RALPH BARIC'S ROLE IN CREATING THE PANDEMIC
 Baric, a former swim champion, described in this early paper how his lab was able to train a coronavirus, MHV, which causes hepatitis in mice, to jump species, so that it could reliably infect BHK (baby-hamster kidney) cell cultures. They did it using serial passaging: repeatedly dosing a mixed solution of mouse cells and hamster cells with mouse-hepatitis virus, while each time decreasing the number of mouse cells and upping the concentration of hamster cells. At first, predictably, the mouse-hepatitis virus couldnt do much with the hamster cells, which were left almost free of infection, floating in their world of fetal-calf serum. But by the end of the experiment, after dozens of passages through cell cultures, the virus had mutated: It had mastered the trick of parasitizing an unfamiliar rodent. A scourge of mice was transformed into a scourge of hamsters. And there was more: It is clear that MHV can rapidly alter its species specificity and infect rats and primates, Baric said. The resulting virus variants are associated with demyelinating diseases in these alternative species. (A demyelinating disease is a disease that damages nerve sheaths.) With steady prodding from laboratory science, along with some rhetorical exaggeration, a lowly mouse ailment was morphed into an emergent threat that might potentially cause nerve damage in primates. That is, nerve damage in us. A few years later, in a further round of interspecies transfer experimentation, Barics scientists introduced their mouse coronavirus into flasks that held a suspension of African-green-monkey cells, human cells, and pig-testicle cells.
Baric, a former swim champion, described in this early paper how his lab was able to train a coronavirus, MHV, which causes hepatitis in mice, to jump species, so that it could reliably infect BHK (baby-hamster kidney) cell cultures. They did it using serial passaging: repeatedly dosing a mixed solution of mouse cells and hamster cells with mouse-hepatitis virus, while each time decreasing the number of mouse cells and upping the concentration of hamster cells. At first, predictably, the mouse-hepatitis virus couldnt do much with the hamster cells, which were left almost free of infection, floating in their world of fetal-calf serum. But by the end of the experiment, after dozens of passages through cell cultures, the virus had mutated: It had mastered the trick of parasitizing an unfamiliar rodent. A scourge of mice was transformed into a scourge of hamsters. And there was more: It is clear that MHV can rapidly alter its species specificity and infect rats and primates, Baric said. The resulting virus variants are associated with demyelinating diseases in these alternative species. (A demyelinating disease is a disease that damages nerve sheaths.) With steady prodding from laboratory science, along with some rhetorical exaggeration, a lowly mouse ailment was morphed into an emergent threat that might potentially cause nerve damage in primates. That is, nerve damage in us. A few years later, in a further round of interspecies transfer experimentation, Barics scientists introduced their mouse coronavirus into flasks that held a suspension of African-green-monkey cells, human cells, and pig-testicle cells.
https://nymag.com/intelligencer/article/coronavirus-lab-escape-theory.html
BARIC WAS CONDUCTING SARS RESEARCH BEFORE A 2003 OUTBREAK IN CHAPEL HILL NORTH CAROLINA

https://media-speakerfile-pre.s3.amazonaws.com/documents/cc4e5e5d442320c20c7f76a0c3cadce51445358867.pdf
BARIC'S LAB LEAKED IN 2003 AND CAUSED A MINOR EPIDEMIC IN NORTH CAROLINA
Baric Bio Intercept
https://www.cdc.gov/about/history/sars/timeline.htm
https://epi.dph.ncdhhs.gov/cd/sars/SARSinNorthCarolina2003.pdf
HOW CAN YOU HAVE SARS-RELATED SYMPTOMS AND NOT HAVE SARS?
"THE EIGHT PREVIOUSLY REPORTED SUSPECTED SARS CASES HAVE ALL RECOVERED FROM THEIR SARS-RELATED SYMPTOMS"
https://epi.dph.ncdhhs.gov/cd/sars/SARSinNorthCarolina2003.pdf
https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5218a2.htm

GAIN OF FUNCTION / GAIN-OF-THREAT RESEARCH INVOLVES CREATION OF RECOMINANT VIRUS IN UNIVERSITY OF NORTH CAROLINA IN COLLABORATION WITH SCIENTISTS FROM WIV
UNIVERSITY OF NORTH CAROLINA PRESS RELEASE NOVEMBER 9, 2015
BARIC'S LAB RAT BITES STUDENT
REDACTIONS OF BIOSAFETY COMMITTEE DISCUSSIONS



BARIC SUPPORTS THE ZOONOTIC THEORY BECAUSE HE PLAYED A PART IN STARTING THE PANDEMIC DUE TO HIS ASSOCIATION WITH WIV SCIENTISTS
CHICOMS BLAME BARIC

THE BAT LADY

"THE BAT LADY" NEVER CONDUCTED EXPERIMENTS TO MAKE VIRUSES MORE DEADLY, JUST MORE CONTAGIOUS

TRUMP AND BIDEN BOTH SAY GET VACCINATED AGAINST THE DASZAK STRAIN
BIDEN: *smiles, walks away* pic.twitter.com/SGWNuJfdLBTWO WUHAN INSTITUTE OF VIROLOGY SCIENTISTS, XING-YI GE AND ZHENGLI-LI SHI, USE REVERSE GENETICS TO GENERATE A CHIMERICAL VIRUS CLOSELY RESEMBLING THE NOVEL CORONAVIRUS COVID-19 (WHY COULDN'T THEY HAVE DEVELOPED THE NOVEL CORONA VIRUS?)

YOUNG BAT LADY
http://www.hbkx.org.cn/ztzl/2020/zmkjgzz/info?newsid=579719f583574e0ca72e0e590366efbd
ANOTHER KEY FIGURE TO EMERGE IN THIS DEBACLE IS ALEKSEI CHMURA

https://europepmc.org/article/PMC/6178078
ALEKSEI CHMURA & PETER DASZAK'S ECO-FASCIST MANIFESTO

"A FALL FROM GRACE TO
VIRULENCE?" PETER DASZAK CORRESPONDING AUTHOR AND ALEKSEI CHMURA
CHAPTER 6
NAZI SCUM BOHDAN DASZAK MAKES THE ECO-FASCIST CHARGE CREDIBLE

DASZAK'S FATHER, BOHDAN DASZAK, WAS A NAZI DEATH CAMP EXECUTIONER

REICHSFUEHRER SS HEINRICH HIMMLER TOURING JANOWSKA DEATH CAMP LEMBERG, DISTRICT OF GALICIA, GENERAL GOVERNMENT (FORMERLY LWÓW, SECOND POLISH REPUBLIC; TODAY LVIV, UKRAINE). OPERATED BY SS OPERATIONAL SEPTEMBER 1941 NOVEMBER 1943 INMATES JEWS
In September 1941, the Germans set up a factory on Janowska Street in the northeastern suburbs of Lvov, in southeastern Poland. This factory became part of a network of factories, the German Armament Works, owned and operated by the SS. Jews were used as forced laborers, mainly in carpentry and metalwork. In October 1941, the Germans established a camp housing the forced laborers next to the factory. In addition to being a forced-labor camp for Jews, Janowska was a transit camp during the mass deportations of Polish Jews to the killing centers in 1942. Jews underwent a selection process in Janowska similar to that used at Auschwitz-Birkenau and Majdanek. Those classified as fit to work remained at Janowska for forced labor. The majority, rejected as unfit for work, were deported to Belzec and killed or were shot at the Piaski ravine, just north of the camp. In the summer and fall of 1942, thousands of Jews (mainly from the Lvov ghetto) were deported to Janowska and killed. The evacuation of the Janowska camp began in November 1943. As the Germans attempted to destroy the traces of mass murder (Aktion 1005), they forced the prisoners to open the mass graves and burn the bodies. On November 19, 1943, these prisoners staged an uprising and a mass escape attempt. A few succeeded in escaping, but most were recaptured and killed. The SS staff and their local auxiliaries murdered at least 6,000 surviving Jews from various forced-labor camps in Galicia when the Janowska camp was liquidated in November 1943. https://ukrainianjewishencounter.org/en/the-janowska-concentration-camp-what-we-know-and-dont-know/

The day began with an inspection that culminated in mass beatings and shootings of prisoners. The weak, the sick, and those who had violated discipline were either shot on the spot or brought to the area between the two barbed wire fences, where they were shot or simply expired. Death runs were also carried out. This meant that a large group of condemned prisoners had to run the gauntlet of SS men, who tripped them with their legs or carbines, beat them over the head, shot at them, etc. There were two gallows in the camp. One of them was for prisoners who wanted to commit suicide. Every day the prisoners were subjected to abuse and beatings for no reason. There is evidence indicating that every day nearly a hundred corpses were piled on the garbage dump next to the kitchen. In other words, this was a daily, systematic killing of people, but the methods used were quite different. Special methods were used to kill women, and we have testimony about sexual violence and crimes in the camp.
Peter Daszaks brother, opera singer John Daszak, revealed: "My father, Bohdan Daszak, (born March 21, 1926 died February 5, 1996 in Tameside, Greater Manchester, England) was a Ukrainian who had ended up in Britain at the end of the Second World War. Thankfully for him he was taken prisoner by the Allies and later released into the community rather than sent back to Ukraine and be dispatched with. He ended up in Manchester where I was born after he married a British woman, she was half Welsh and half English so technically I am only a quarter English [laughs]. So I was brought up here but never really felt I always fitted in. It was only when I started travelling around I realised why my father was so culturally very different to those around me."
https://seenandheard-international.com/2013/09/newjohn-daszak-genesis-of-a-new-british-siegfried-an-interview-with-jim-pritchard
John Daszak not only had a powerful mother, but his father, Bogdan Daszak, had lived a life of exceptional bravery and determination. He had managed the extraordinary feat of reaching England from the Ukraine during WWII.[As a Prisoner-of-War] When he was about 16, in March 1944, the Germans had occupied his country. They captured him by going round the villages threatening to shoot the families unless the sons joined the German army. At that time the boys hated the Russians even more, and so were willing to join the the Germans who were fighting the Russians. But since the Russians and become allies of the British, the boys became enemies of the British. Many who had been badly treated ran off and escaped. Bodgen though was an educated town boy, not a peasant like the others, so he was used for paperwork in a [Nazi death] camp.1. He was eventually taken as a POW by the British. He could not know whether his family was alive, nor could he try to contact them becase he knew that is someone found out his was alive, they would torture his family. In fact Bogdans father was tortured by the Russians. About 40 years later, the the mid-1980s, a friend spotted the name Zenon Daszak in a magazine and told Bogdan, who was thrilled to find his long lost brother alive. Zenon he said, was always practicing the violin, and though hed preferred football himself, and he used to laugh at his brothers devotion. Zenon had become head of music Conservatories in Lvov in the Ukraine, their home town, and had played in the Bolshoi orchestra. Lvov was at the time under Soviet rule and it was forbidden for a Westerner to openly contact someone there. Bogdan though managed to pass his brother a secret message, along with his childhood pet name that no one else would know. They met in 1989 in Munich where Zenon was adjudicating at an international string competition. This had to be in secret shaking off the KGB. Zenon had named one of his sons Bogdan. Another one of his sons Uri had become a well known viola player. Both brothers died in their 60s and John no longer and John no longer has contact with Zenons family. It had been particularly important to Bogdan that his family became totally British. He forbade them from visiting the Ukraine.
https://www.google.com/books/edition/Gifted_Lives/ySItCK3A4uEC
Public prosecutors in Germany have indicted a 95-year-old woman for her role supporting the Nazi killing machinery as a secretary in a concentration camp, charging her with 10,000 counts of being an accessory to murder, and complicity in attempted murders. The indictment against the woman, identified only as Irmgard Furchner under German privacy laws, followed a five-year investigation, prosecutors said. Because she was under 21 at the time of the offenses she is accused of, they said, she would be tried in a juvenile court, where she is likely to receive a milder sentence. The woman worked between June of 1943 and April of 1945 as a secretary for the camp commander at the Stutthof camp, 20 miles from the Polish city of Gdansk, which was known as Danzig under German rule at the time. Its about the concrete responsibility she had in the daily functioning of the camp, said Peter Müller-Rakow of the public prosecutors offices in Itzehoe, north of Hamburg. NYT
Those Ukrainians who did collaborate with the German occupiers did so in various ways including participating in the local administration, in German-supervised auxiliary police, Schutzmannschaft, in the German military, and serving as concentration camp guards. Nationalists in western Ukraine were among the most enthusiastic and hoped that their efforts would enable them to re-establish an independent state later on. For example, on the eve of Operation' Barbarossa, as many as 4000 Ukrainians, operating under Wehrmacht orders, sought to cause disruption behind Soviet lines. After the capture of Lviv, a highly-contentious and strategically-important city with a significant Ukrainian minority, OUN [Organization of Ukrainian Nationalists], leaders proclaimed a new Ukrainian State on June 30, 1941 and simultaneously encouraged loyalty to the new regime in the hope that they would be supported by the Germans. Already in 1939, during the German-Polish War, the OUN had been "a faithful German auxiliary." Few people noticed when they arrived in Britain in May 1947: one Labour MP, Barnett Janner, complained bitterly in the Commons that members of the Galizien division "murdered hundreds of people in cold blood", while a solitary letter in the London Evening News, signed with the correspondent's death camp number, 3399, complained that he or she had witnessed first-hand the "brutal, uncouth and bloodthirsty" behaviour of Ukrainian guards. Most newspapers devoted just one paragraph to reporting the division's arrival, however. The men were dispersed among PoW camps. Over the next three years just eight "undesirables" were deported to Germany, while some emigrated to Canada, the US or Argentina.
Daszak knew the viruses had leaking potential leading to a pandemic yet he did nothing even after the State Department deemed the WIV unsafe he continued funding the bat virus and collection work. A pandemic would delay human encroachment on nature. Daszak favors plant and animal life over human life. Daszak is not only an eco-fascist, but a eco-Fascist mole or eco-Fascist spook. What this means is that you can't tell where he is coming from, from his social media. He is not connected to the rightwing and has embedded himself in the Democratic Party.

Daszak grew up in Dukinfield, England, a coal-mining town outside of Manchester. His father was a Ukrainian conscripted to serve in the German army when the Nazis invaded who wound up a British prisoner of war in Italy, which eventually led him to Scotland and a job in a chocolate biscuit factory. He later worked as a draftsman and married a woman from Dukinfield. Daszak had 18 cages of reptiles at home. Im a lizard guy, he says. In a primary school essay, he wrote that he wanted to become a zoologist and study marmosets in the Amazon. He studied zoology at Bangor University and earned a Ph.D. in infectious diseases at the University of East London with a thesis on electron microscopy of a parasite that caused an intestinal disease in chickens A postdoctoral [Daszak worked at the School of Life Sciences, Kingston University, in Surrey, England in the 1990s. He never earned a post-graduate degree Wiki] stint at Kingston University led to a collaboration with former surgeon Andrew Wakefield, now infamous for his role in the antivaccine movement."

Wakefield asked Daszak to do electron microscopy of gut segments removed from Crohn disease patients. They found evidence of the measles virus, and Wakefielda charismatic guy, Daszak sayswent on to argue that the measles vaccine might be to blame for the disease. Subsequent studies made Daszak question the findings. No one could repeat it, he says. Wakefield was later discredited for a fraudulent study that linked vaccines to autism and has become a touchstone for vaccination skeptics. Daszak had 18 cages of reptiles at home. Im a lizard guy, he says. In a primary school essay, he wrote that he wanted to become a zoologist and study marmosets in the Amazon. He studied zoology at Bangor University and earned a Ph.D. in infectious diseases at the University of East London with a thesis on electron microscopy of a parasite that caused an intestinal disease in chickens. Science
Daszak financed DURC Dual Use Research of Concern (DURC) proceedures. The DURC experiments involved creating new forms of life, just as Mary Shelly's Doctor Frankenstein did when he created a homicidal monster. But the chance of creating a monster virus didn't bother "Dr. Daszakstein," who headed a "non-profit" called EcoHealth Alliance, because it was Doctor Daszak's intention from the start to cause a pandemic. Dr. Daszak is not a microbiologist. Daszak has no medical degree. None-the-less he plays a major part in the most dangerous experiments known to man. He's a world-class conman with a British accent and a hidden agenda, as stated, the son of a Nazi war criminal, a Ukranian who volunteered to kill Jews in a notorious death camp. Daszak never talks about the father who raised him, Bohdan. As for December 2021 Peter Daszak murdered 5 million including many Jews - and counting......Experts say that five million is an undercount. Many countries are unable to accurately record the number of people who have died from Covid-19, like India and African nations. It took nine months for the virus to kill one million people, three and a half more to reach two million, another three to claim three million and about two and a half to exceed four million.
DASZAK: "If we do the math on the previous emerging disease events the viruses tend to come from animals. Mammals cover the vast majority. You should probably include birds, because of Avian flu being such a threat. It is a stamp collecting exercise. Some of those stamps are penny blanks and you have to throw them out. Some of the viruses will be killers and some of them wont. How do we work that out from a viral sequence? It is not straight forward. First of all we are only looking at viral families that include those that have gotten to people from animals. We have narrowed it down from there. Then, when you get a sequence and it looks like a relative to their known nasty pathogen just like we did with SARS we found viruses in bats. Some of them looked very similar to SARS. We sequenced the spine protein, the protein that attaches to cells, and then you create pseudo particles, insert proteins from the viruses that combined to human cells. Each step of this you move closer and closer to could the virus becomes pathogenic in people! You narrow down the field. You end up with a small number of viruses that really do look like killers. And you look at the people in the region where the animal lives; do we see anti-bodies to that virus? In Cameroon it was a new virus coming into people. What is the activity and can we find the alternative? It is not straight forward, but there are many ways to hone it down. We had just got to do it. It makes sense for humanities sake." See video
Daszak : "We found corona virus in bats which looked exactly like SARS....My colleagues in China [at the Wuhan Institute of Virology] did this work, you create pseudoparticles1, you insert the spike proteins from those viruses, see if they bind to human cells and each step of this you move closer and closer to: a virus could really become pathogenic in people." Most of the experiments with bat viruses were performed with wild-type viruses.
1. Pseudotypes are biosafe, replication-defective retroviruses and lentiviruses that have been repurposed from gene therapy applications. They have heterologous (foreign) viral glycoproteins on their surface that originate from the viral pathogen. They thus mimic the wild- type virus with respect to cell entry, but can be handled by end users in biosafety level (BSL)-1 instead of BSL-3/4 laboratories. "Experience working with receptor mutants & pseudovirus binding assays: We have established a stable pseudovirus assay for SARS-CoV and SARS-like CoV and tested the infectivity of these spike proteins in cells expressing ACE2 from human. civet and bats (52, 68). We have demonstrated that several bat species are susceptible to the SARS-CoV and that some SARS-like CoV strains can use human ACE2 for cellular entry (Intercept 125) Minor mutations in S proteins or ACE2s greatly affected the receptor binding and finally abolish the pseudovirus entry (68, 121) (Fig. 9). Recently, we have discovered a number of alpha and beta CoVs including HKU4/5
We will use prepared pseudoviruses bearing different S proteins to infect human and bat ACE2 or DPP4 receptor expressing cells (in Hela cell model), 24 hours post infection, receptor usage by different S proteins will be determined by measuring luciferase activities. We will also induce site mutations in S proteins using site-directed mutation method, then do receptor-mutant pseudovirus binding assays. Pseudovirus infectivity on different human cell lines (A549, 293T, Caco, Huh?. and etc), primary and immortalized bat cell lines (listed below) and other mammalian cell line (mouse, pig, hamster, monkey, and ect) will be also determined by luciferase assay. The results will provide information whether bat-CoVs could use known bat and human ACE2. DPP4 or other known CoV receptors to enter cells. and allow us to determine critical receptor binding sites. viral host range, and to better predict the capacity of our Co Vs to infect people. (Intercept 118)
 Daszak continued to finance the collection of exotic bat viruses and GoF / GoT experiments that were being performed in a low level biosecurity lab at the WIV - despite the fact they were banned in the USA and despite the fact he was not a microbiologist. Daszak knew if GoF / GoT proceedures were banned in the USA, where bio-security was high, they should not be out-sourced to China, where he knew first hand bio-security was minimal. Any normal person in Daszak's position would have seen red flags. But Daszak had a hidden agenda which was to start a pandemic, so he ignored them. The U.S. House of Representatives Foreign Affairs Committee: "Was Daszak aware the funding he was providing directly supported gain-of-function research by paying for the collection of viruses the Wuhan Institute of Virology (WIV) later experimented with, even though the federal government had a moratorium on GoF research from 2014 through 2017?" Either a bat virus escaped that could directly infect humans or these procedures created a novel bat virus whose spike protein was genetically engineered to easily interface with the human respiratory system's ACE2 receptors, ostensibly to determine in which species of bat the next spillover to humans would occur. Daszak had predicted that a new virus would arise from a spillover. He called it VIRUS X. The enhanced natural bat virus that Daszak's crew created , or natural one they collected, escaped the Bio Safety Lab-2 facility, that had a much security as a dental office, and caused a pandemic setting back civilization God knows how long? Or the virus was one of the known bat-CoVs (including 630 novel CoV sequences) collected and stored at the WIV. Daszak puts the collection figures much higher. Perhaps it was the Rhinolophus Horseshoe virus or RaTG13 or RmYN02 that was altered? The virus was bio-engineered to be more contagious, not more virulent, accounting for the number of mild cases, before it began to mutate into something more deadly, such as "the Delta Mutation." The Delta Variant causes mucormycosis, Black Fungus, a rare and often deadly disease that has exploded across India on the coattails of the coronavirus pandemic. 44% of new Covid cases in NYC are attributable to this mutation. Fully immunized people with breakthrough infections of the Delta variant can spread the virus to others just as readily as unvaccinated people. The Alpha variant proved to be twice as transmissible as the original virus, and the Delta variant has mutations that turbocharged its contagiousness even more. As the virus continues to change, newer variants may turn out to be even more transmissible. In early August 2021 in Wuhan, the authorities started testing all 12 million residents after only three cases of the Delta variant were discovered. The cities of Sanmenxia and Zhuhai have also begun mass testing. In Beijing, where there are five infections, train service from 23 cities has been canceled. In the U.S. This Delta wave just rips through the unvaccinated, said Howard Markel, a medical historian at the University of Michigan. The deaths that have followed the wide availability of vaccines, he added, are absolutely needless. The variant is thought to be more contagious than previous versions of the coronavirus because the infected carry it in substantially greater amounts in their airways.
Daszak continued to finance the collection of exotic bat viruses and GoF / GoT experiments that were being performed in a low level biosecurity lab at the WIV - despite the fact they were banned in the USA and despite the fact he was not a microbiologist. Daszak knew if GoF / GoT proceedures were banned in the USA, where bio-security was high, they should not be out-sourced to China, where he knew first hand bio-security was minimal. Any normal person in Daszak's position would have seen red flags. But Daszak had a hidden agenda which was to start a pandemic, so he ignored them. The U.S. House of Representatives Foreign Affairs Committee: "Was Daszak aware the funding he was providing directly supported gain-of-function research by paying for the collection of viruses the Wuhan Institute of Virology (WIV) later experimented with, even though the federal government had a moratorium on GoF research from 2014 through 2017?" Either a bat virus escaped that could directly infect humans or these procedures created a novel bat virus whose spike protein was genetically engineered to easily interface with the human respiratory system's ACE2 receptors, ostensibly to determine in which species of bat the next spillover to humans would occur. Daszak had predicted that a new virus would arise from a spillover. He called it VIRUS X. The enhanced natural bat virus that Daszak's crew created , or natural one they collected, escaped the Bio Safety Lab-2 facility, that had a much security as a dental office, and caused a pandemic setting back civilization God knows how long? Or the virus was one of the known bat-CoVs (including 630 novel CoV sequences) collected and stored at the WIV. Daszak puts the collection figures much higher. Perhaps it was the Rhinolophus Horseshoe virus or RaTG13 or RmYN02 that was altered? The virus was bio-engineered to be more contagious, not more virulent, accounting for the number of mild cases, before it began to mutate into something more deadly, such as "the Delta Mutation." The Delta Variant causes mucormycosis, Black Fungus, a rare and often deadly disease that has exploded across India on the coattails of the coronavirus pandemic. 44% of new Covid cases in NYC are attributable to this mutation. Fully immunized people with breakthrough infections of the Delta variant can spread the virus to others just as readily as unvaccinated people. The Alpha variant proved to be twice as transmissible as the original virus, and the Delta variant has mutations that turbocharged its contagiousness even more. As the virus continues to change, newer variants may turn out to be even more transmissible. In early August 2021 in Wuhan, the authorities started testing all 12 million residents after only three cases of the Delta variant were discovered. The cities of Sanmenxia and Zhuhai have also begun mass testing. In Beijing, where there are five infections, train service from 23 cities has been canceled. In the U.S. This Delta wave just rips through the unvaccinated, said Howard Markel, a medical historian at the University of Michigan. The deaths that have followed the wide availability of vaccines, he added, are absolutely needless. The variant is thought to be more contagious than previous versions of the coronavirus because the infected carry it in substantially greater amounts in their airways.
Over all, the birthrate declined by 4 percent in 2020. Births were down most sharply in December 2000, when babies conceived at the start of the pandemic would have been born. The New York Times reported: "All over the globe, countries are confronting population stagnation and a fertility bust, a dizzying reversal unmatched in recorded history. Fewer babies cries. More abandoned homes. Toward the middle of this century, as deaths start to exceed births, changes will come that are hard to fathom." When one of SHI, BARIC, DASZAK designer viruses escaped, 3,840,528 people died and 177,559,790 were infected. In the United States, life expectancy fell by a full year in the first six months of 2020; for Black Americans, the drop was 2.7 years. [Daszak employs no blacks at his Eco-Health Alliance operation.] December 2020 had the single largest decline of any month. Over all for the year, births declined by 4 percent, the data showed. There were 3,605,201 births in the United States last year, the lowest number since 1979. The International Monetary Fund predicts that the global economy will lose over $22 trillion between 2020 and 2025. A major part of Eco-Fascism's agenda was fullfilled. To an ecofascist, in order to achieve both environmental and social purity, people of color and immigrants must sacrifice themselves for the good of the planet. From 2019 to 2020, Hispanic people experienced the greatest drop in life expectancy three years and Black Americans saw a decrease of 2.9 years. White people experienced the smallest decline, of 1.2 years. Deaths exceed births in Alabama
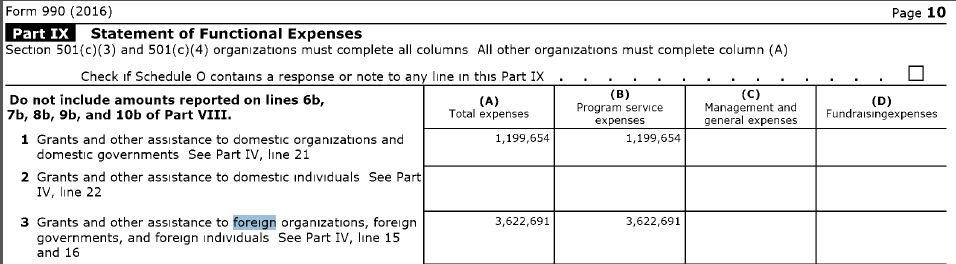
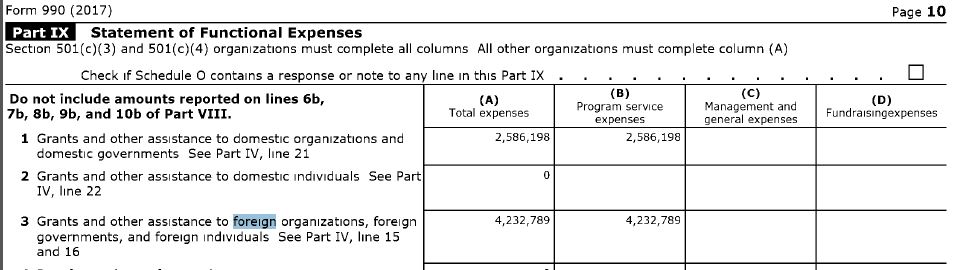
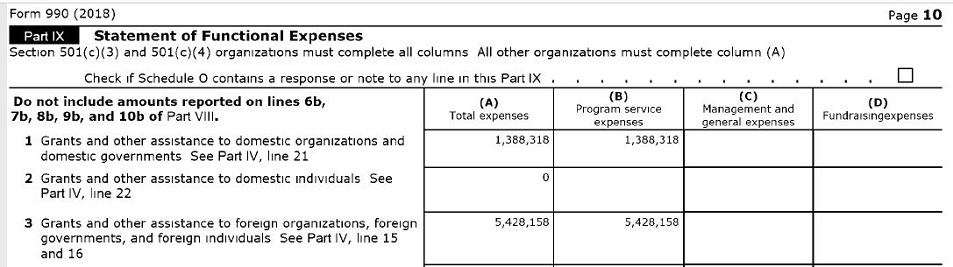
Did the organization have aggregate revenues or expenses of more than $10,000 from grantmaking, fundraising, business, investment, and program service activit1es outside the United States, or aggregate foreign investments valued at $100,000 or more? If "Yes," complete Schedule F, Parts I and IV. YES
Did the organization report on Part IX, column (A), line 3, more than $5,000 of aggregate grants or other assistance to or for foreign Individuals? If "Yes," complete Schedule F, Parts III and IV. YES
Was any supported organization not organized in the United States ("foreign supported organization")' If "Yes" and if you checked 12a or 12b to Part I, answer (b) and (c) below 4ab Did the organization have ultimate control and discretion in deciding whether to make grants to the foreign supported organization? If "Yes," describe in Part VI how the organization had such control and discretion despite being controlled or 4b supervised by or in connection with its supported organizations c. Did the organization support any foreign supported organization that does not have an IRS determination under sections 501 (c)( 3) and 509 (a) ( 1) or (2)' If "Yes," explain in Part VI what controls the organization used to ensure that all support to the foreign supported organtzation was used exclusively for section 170(c)(2)(8) purposes. LEFT BLANK

2014-2019 Daily Mail:"The US National Institutes of Health (NIH) funded bat-coronavirus research in the Wuhan Institute of Virology in China to the tune of US$3.7 million. Back in October 2014, the US government had placed a federal moratorium on gain-of-function (GoF) research altering natural pathogens to make them more deadly and infectious as a result of rising fears about a possible pandemic caused by an accidental or deliberate release of these genetically engineered monster germs.1 A portion of $3.7 million in grants awarded between 2014 and 2019 by the National Institutes of Health (NIH) funded research at the Wuhan Institute of Virology in China. However, not all of that $3.7 million went to the WIV, and not all of the funding took place under the Obama administration. Approximately $700,000 of the $3.7 million total was approved under Donald Trump."
https://asiatimes.com/2020/04/why-us-outsourced-bat-virus-research-to-wuhan
https://www.snopes.com/fact-check/obama-admin-wuhan-lab-grant
1. The funding was laundered though Daszak's ECOHEALTH ALLIANCE.
Daszak was one of the first men to find out about the virus becqause he was expecting a virus to leak and told his assets in China to be on the lookout for it. His story about how he first heard about it is full of contradictions. He told Wired: "On the night after the first Covid-19 post on December 31, 2019, which would be January 1, 2020, Dr. Peter Daszak,a disease ecologist and president of the EcoHealth Alliance, interrupted Marjorie Pollacks New Years Eve dinner [December 31, 2019] to relay details his Mandarin-speaking staff members had picked up from China. Early lab work done in the Wuhan Institute of Virology (WIV) showed the novel coronavirus behind Covid-19 had 80 percent homologysimilarity due to shared ancestrywith the 2003 coronavirus that caused SARS. Daszak wanted the ProMED volunteers to know, he says, because they are a gold standard for information. He went on: "I got hold of Marjorie Pollack on New Years Eve [December 31, 2019], and as the champagne was getting warm we realized something really serious was going on in China. We had it from a good source that this was a coronavirus and that it was 20% different to SARS. So we knew that SARS was pretty good at transmitting from person to person; we knew it had a 10% mortality rate. Thats a huge red flag. I really knew something was wrong when every single senior person that I was trying to get hold of in China was busy. I sent a really long text to George Gao, whos head of the CDC. I offered to send a team out there, to come out there and do anything to support them, and I got the shortest response ever from George, which was "Happy New Year." Wired March 5, 2020
December 31, 2019 the World Health Organization's Country Office in the Peoples Republic of China picked up a media statement by the Wuhan Municipal Health Commission from their website on cases of viral pneumonia in Wuhan, Peoples Republic of China. The night after this was January 1, 2019 First Daszak says it came from his his Mandarin-speaking staff members then he says he and Dr. Pollack had the same source in China. He was concealing the fact that he was the first to become aware of SARS-CoV-19 from his Bat Lady dupe at the WIV, Shi Zheng Li.
Dr. Shi: "I have been working on the discovery and characterization of novel viruses from bats and other wildlife since 2004. This included the discovery that Chinese horseshoe bats are the natural reservoir of SARSr-CoVs and the likely origin of SARS-CoV. My group then isolated SARSr-CoVs from bats sharing high homology with human SARS-CoV and demonstrated their interspecies transmission risk, largely confirming bats as the source of SARs" (Intercept 352)
Ira Lipkin lies and tells 9/11 revisionist Spike Lee he was the first to learn of Covid

"While much of the public debate was initially focused on the Huanan seafood market in Wuhan as the origin of the pandemic, the preponderance of evidence now suggests that the virus leaked from the Wuhan Institute of Virology. Given the WIVs demonstrated history of conducting gain-of-function experiments on coronaviruses, including genetically manipulating viruses specifically to make them infectious to humans in BSL-2 labs, as well as their possession of one of the worlds largest of coronaviruses, it is completely plausible that one or more researcher(s) was accidentally infected and carried the virus out of the lab. The evidence outlined above, combined the cover-up conducted CCP authorities, strongly suggest the Wuhan Institute of Virology as the source of the current pandemic.... After this extensive investigation, we believe it is time to call Peter Daszak to testify before Congress. There are still many outstanding questions about the type of research he funded at the WIV that only he can answer. In addition, we believe there is legislation Congress can pass that would not only hold those responsible accountable but also help to prevent a future pandemic, including but not limited to:
Institute a ban on conducting and funding any work that includes gain-of-function research until an international and legally binding standard is set, and only where that standard is verifiably being followed. Sanction the Chinese Academy of Sciences and affiliated entities. List the Wuhan Institute of Virology and its leadership on the Specially Designated Nationals and Blocked Persons List and apply additional, appropriate secondary sanctions. Authorize new sanctions for academic, governmental, and military bioresearch facilities that fail to ensure the appropriate levels of safety and information sharing."

Get to know our scientists! Andrew Huff, Ph.D. Associate Vice President Dr. Andrew Huff began his formal education after two combat tours of duty as an infantryman in Operation Enduring Freedom throughout Central America and in Iraq. After earning a bachelors degree in psychology, and masters degrees in security technologies and geographic information systems (GIS), he was appointed a research fellow at the National Center for Food Protection and Defense (NCFPD) while earning a Ph.D. in Environmental Health specializing in emerging and environmental infectious disease at the University of Minnesota. ECOHEALTH NEWSLETTER
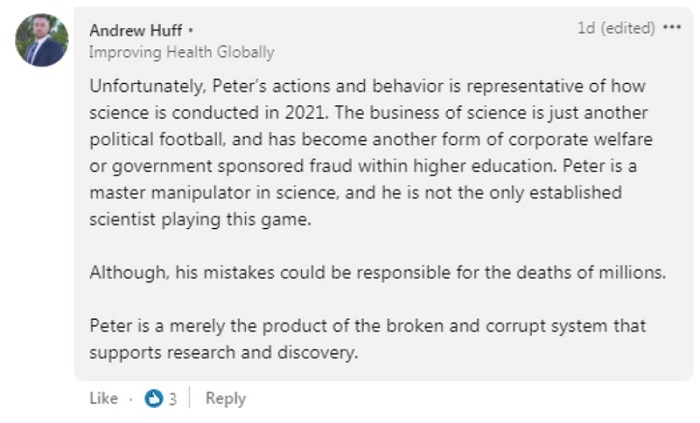

The IC [Who is the Intelligence Community?] assesses that SARS-CoV-2, the virus that causes COVID-19, probably emerged and infected humans through an initial small-scale exposure that occurred no later than November 2019 with the first known cluster of COVID-19 cases arising in Wuhan, China in December 2019. In addition, the IC was able to reach broad agreement on several other key issues. We judge the virus was not developed as a biological weapon. Most agencies also assess with low confidence that SARS-CoV-2 probably was not genetically engineered; however, two agencies believe there was not sufficient evidence to make an assessment either way. Finally, the IC assesses Chinas officials did not have foreknowledge of the virus before the initial outbreak of COVID-19 emerged. After examining all available intelligence reporting and other information, though, the IC remains divided on the most likely origin of COVID-19. All agencies assess that two hypotheses are plausible: natural exposure to an infected animal and a laboratory-associated incident. Four IC elements and the National Intelligence Council assess with low confidence that the initial SARS-CoV-2 infection was most likely caused by natural exposure to an animal infected with it or a close progenitor virusa virus that probably would be more than 99 percent similar to SARS-CoV-2. These analysts give weight to Chinas officials lack of foreknowledge, the numerous vectors for natural exposure, and other factors. One IC element assesses with moderate confidence that the first human infection with SARS-CoV-2 most likely was the result of a laboratory-associated incident, probably involving experimentation, animal handling, or sampling by the Wuhan Institute of Virology. These analysts give weight to the inherently risky nature of work on coronaviruses.
"The IC assesses that SARS-CoV-2, the virus that causes COVID-19, probably emerged and infected humans through an initial small-scale exposure" if it started at the Wuhan Market it would have been large scale "that occurred no later than November 2019 with the first known cluster of COVID-19 cases arising in Wuhan, China in December 2019. In addition, the IC was able to reach broad agreement on several other key issues. We judge the virus was not developed as a biological weapon. [If it was why are the Chinese vaccines ineffective?] Most agencies also assess with low confidence that SARS-CoV-2 probably was not genetically engineered; however, two agencies believe there was not sufficient evidence to make an assessment either way. Finally, the IC assesses Chinas officials did not have foreknowledge of the virus before the initial outbreak of COVID-19 emerged." Unless we know which Agency made each assesment the information is meaningless.
After examining all available intelligence reporting and other information, though, the IC remains divided on the most likely origin of COVID-19. All agencies assess that two hypotheses are plausible: natural exposure to an infected animal and a laboratory-associated incident. Four IC elements and the National Intelligence Council assess with low confidence that the initial SARS-CoV-2 infection was most likely caused by natural exposure to an animal infected with it or a close progenitor virusa virus that probably would be more than 99 percent similar to SARS-CoV-2. These analysts give weight to Chinas officials lack of foreknowledge, the numerous vectors for natural exposure, and other factors. One IC element assesses with moderate confidence that the first human infection with SARS-CoV-2 most likely was the result of a laboratory-associated incident, probably involving experimentation, animal handling, or sampling by the Wuhan Institute of Virology. These analysts give weight to the inherently risky nature of work on coronaviruses.Unless we know which Agency made each assesment the information is meaningless.
Analysts at three IC elements remain unable to coalesce around either explanation without additional information, with some analysts favoring natural origin, others a laboratory origin, and some seeing the hypotheses as equally likely. Variations in analytic views largely stem from differences in how agencies weigh intelligence reporting and scientific publications, and intelligence and scientific gaps. The IC judges they will be unable to provide a more definitive explanation for the origin of COVID-19 unless new information allows them to determine the specific pathway for initial natural contact with an animal or to determine that a laboratory in Wuhan was handling SARSCoV-2 or a close progenitor virus before COVID-19 emerged. The ICand the global scientific communitylacks clinical samples or a complete understanding of epidemiological data from the earliest COVID-19 cases. If we obtain information on the earliest cases that identified a location of interest or occupational exposure, it may alter our evaluation of hypotheses. Chinas cooperation most likely would be needed to reach a conclusive assessment of the origins of COVID-19. Beijing, however, continues to hinder the global investigation, resist sharing information and blame other countries, including the United States. These actions reflect, in part, Chinas governments own uncertainty about where an investigation could lead as well as its frustration the international community is using the issue to exert political pressure on China.
Three unidentified workers from the Wuhan Institute of Virology went to the hospital in November 2019 with flu-like symptoms. WAPO: "In China, people visit the hospital for routine and mild illnesses." However the intelligence came from the French who built the high containment lab at the WIV and was a result of an inquiry to the French about what to do about this accident. How to decontaminate the labs.
DASZAK: I remember specifically asking the Wuhan lab director and the staff about people who got sick, and we repeated that and pushed and asked pretty tough questions around that and they refuted it.
GUPTA: According to the Wuhan Institute of Virology on practices, they would have taken blood samples around that time.
DASZAK: Yes.
GUPTA: Did you get to see those blood samples?
DASZAK: No. No. But we did ask them. We asked them if they do that, they confirmed that they did take sample, that they tested them after the outbreak and that they were negative for COVID.
GUPTA (voice-over): The same missing pathogen database containing thousands of bat coronavirus entries that was taken offline by WIV in September of 2019.
ALINA CHAN, BROAD INSTITUTE: So this is a database that the mission was to collect all the viruses in the wild and use it to help prevent pandemics. So this is a pandemic. So why didn't they share that database with other scientists so they could figure out how dangerous this virus, where did it come from? [A Chinese news outlet accused her of filthy behavior and a lack of basic academic ethics, and readers piled on that she was a race-traitor, because of her Chinese ancestry. To scientists and editors who are directly or indirectly covering up severe research integrity issues surrounding the key SARS-2-like viruses to stop and think. If your actions obscure SARS2 origins, youre playing a hand in the death of millions of people. ]
GUPTA (on-camera): What is the status of that database now? Have you been able now then as a member of this WHO team or in any capacity to look at that data?
DASZAK: No.
GUPTA: That sounds concerning, Peter, if it is as serious and we're trying to be as thorough as possible. Maybe it mounts to nothing but I think the fact that you still haven't seen that database, it's just going to raise a lot of eyebrows as we go forward.
DASZAK: Well, rightly so. I think that, you know, China should be more open about the things that they've not released. But this is where politics comes into and when a country is under attack, and they defend and then scientists are caught in the middle of it. It's really unfortunate.
GUPTA (voice-over): Dr. Shi Zhengli and others at the Wuhan Institute of Virology did not respond to interview requests for this documentary.

"These analysts also note that Chinas investigations into the pandemics origin might not uncover evidence of a laboratory-associated incident if it involved only a small number of researchers who did not acknowledge or have knowledge of a potential infection. Updated Assessment on COVID-19 Origins"

Is the so-called respiratory disease or white lung disease of unknown cause that broke out in the United States in the second half of 2019 as new coronary pneumonia? What kind of research is being carried out in Fort Detrick and other biological laboratories in the United States? Is it related to coronavirus? How are the security measures implemented? Is it related to the origin of the global new crown epidemic? These are all questions that should be answered in the tracing of the new crown, but the US government has been covering up. In order to use the epidemic to achieve political goals, the US government suppressed scientists and suppressed professional opinions. In particular, scientists who deny the "laboratory leak theory" are subjected to cyber violence and harassment, not only affecting their normal work, but even facing personal threats.
Beijing won't accept the W.H.O's proposed second phase investigation into the origins of the Covid-19 pandemic, accusing it of arrogance and a disrespect for common sense with its return to the lab leak theory. Zeng Yixin, deputy minister of the National Health Commission, said he was very surprised when he first read about the proposed study because it places the hypothesis that Chinas breach of laboratory protocols caused the virus to leak as one of the research priorities. Zeng said none of the staff or postgraduates at the Wuhan Institute of Virology at the centre of the lab leak claims had been infected with the new coronavirus and the lab had not conducted any gain-of-function studies. Fauci approved Daszak re-purposing NIH grants to the WIV so these experiments could go foward, assuming his Deputy, Erik Stemmy, apprised him of them. South China Morning Post
GOP ORIGINS REPORT: "Therefore, it is reasonable to conclude, based on the WIVs extensive sample library and history of genetically manipulating coronaviruses, that in early September, one or more researchers became infected with SARS-CoV-2 in the lab and carried it out into the city. Based on the WIVs publications, researchers could have been exposed while experimenting with a natural virus collected from the wild or infected with a virus they genetically manipulated. Those researchers likely traveled to and from the WIV via the Wuhan metro or via the shuttle service, providing a vector for the virus to spread. This corresponds with the first signs of a growing wave of ill people in Wuhan centered around the WIVs Wuchang facility."
Doctor Anthony Fauci states we can't rely on the three scientists falling ill, intell, until China produces the scientists medical records, as if this were a real possibility. China is a "socialist" slave-state where 43% of the population, earns $150-a-month and a country that routinely launches cyber-attacks against the U.S. The Chinese insist that Covid-19 didn't orginate in Wuhan so there will never be any medical records. CCP spokeswoman Hua Chunying: "Id like to stress that if the United States truly respects facts, it should open the biological lab at Fort Detrick, Maryland, give more transparency to issues like its 200-plus overseas bio-labs, invite WHO experts to conduct origin-tracing in the United States, and respond to the concerns from the international community with real actions."
"The idea is to make prototype vaccines to protect against viruses from about 20 families that might spark a new pandemic. Using research tools that proved successful for Covid-19, researchers would uncover the molecular structure of each virus, learn where antibodies must strike it, and how to prod the body into making exactly those antibodies. It would cost a few billion dollars a year, take five years for the first crop of results and engage a huge cadre of scientists, he said." There has never been a vaccine produced by GoF / GoT experiments either before and outbreak or after one. It is junk science. No Mention of GoF experiments in new PREDICT plan
Daszak: "We have obtained a letter of support from Dr Ralph Baric, who is keen to collaborate with us initially to infect his humanized mouse model with our bat SL-CoV that uses ACE2, and subsequently to use other CoVs we identify (see Dr Ralph Baric, Letter of Support) Intercept 125. In October 2014 President Obama banned Gain-of-Threat experiments in the USA. Obama feared a global pandemic if one of the nano particle-sized viruses escaped via a leak or in an infected human or animal. Daszak was one step ahead of Obama, with help from the NIH's Fauci, Daszak had already outsourced the experiments to the WIV. Again, Gain-of-Threat experiments, called "Gain-of-Function" experiments, utilize genetic engineering to make a virus hundreds of times more transmittable than the virus nature created. GoF is also used to expand the number of hosts the virus can infect. SARS-CoV-2 was not designed to mutate rapidly making it difficult for vaccines to be developed that would work on every variant.1 But it is in so many human hosts that it does just that: e. g. "About half of adults infected in a June 2021 outbreak of the Delta variant of Covid-19 in Israel were fully inoculated with the Pfizer Inc. vaccine, prompting the government to reimpose an indoor mask requirement and other measures to contain the highly transmissible strain." However the NYT reported: "The vaccines made by Pfizer-BioNTech and Moderna set off a persistent immune reaction in the body that may protect against the coronavirus for years, scientists reported. The findings add to growing evidence that most people immunized with the mRNA vaccines may not need boosters, so long as the virus and its variants do not evolve much beyond their current forms which is not guaranteed." The paradigm for many was that Israel is the most vaccinated country in the world, that vaccinated people rarely become infected, that even more rarely do they become severely ill and that basically, with very few precautions in place, the population was very close to herd immunity. The problemwas that what was true for the original virus did not necessarily hold true for future variants coupled with waning immunity.
1. "Variant" is a mutation. That's a word no one likes to use.
Despite Obama's Executive Order, made against the wishes of NIH head Fauci, Dr. Daszak continued to "re-purpose" part of his NIH / NIAID / USAID (CIA lite) grant money to the WIV where no such ban existed, so it could continue the GoT experiments. Why was Daszak gung-ho on seeing gain-of-threat experiments continue? Daszak's cover is liberal-Democrat but he is really a Chinese agent-of-influence. Daszak's organization, Eco-Health Alliance, was first described as a foreign entity. Daszak is a Fascist, a reincarnation of his Fascist father Bohdan Daszak, but this time around, an Eco-Fascist.

Not only did Daszak orchestrate the pandemic, but he, along with Dr. Marion Koopmans, orchestrated the coverup. Daszak was the most visible presence in the bogus W.H.O. investigation that blamed the pandemic on imported frozen meat.
Daszak: "What possible infection sources in Huanan market? Could have been introduced by a person, or a product (e.g. frozen farmed wildlife INCLUDING SPECIES SUSCEPTIBLE TO COVID)..Vendors were trading wildlife products..frozen. From farms within the country. On future work amidst [Chinese] political sensitivities: I stand resolute against the political attacks that we see every day. [In Wuhan] we met scientists who are ready, willing & able to collaborate. Im looking forward to digging deeper & tracing back the origins of this virus....What we found is evidence of a way the virus could have emerged from rural China into a big city like Wuhan... at the end of the report, both the China team of experts & the WHO experts all felt this was the most likely pathway that the virus took. The WHO team examined lab work of a number of researchers in Wuhan & found "no evidence at all that any of the labs in China were working on this virus prior to the outbreak...Did they have viruses that were potential ancestor of SARS-CoV-2? Again, no evidence of that. Attempts to re-engage with China may not be the worst thing. The Trump administration taught us that the United States most effective way of destabilizing China is to destabilize itself. That this is a foolish exercise should be self-evident. We need better options."
The CGTN report quoted Daszak as saying there is a huge amount of evidence that these viruses repeatedly emerge into people from wild animals in rural areas through things like hunting and eating wildlife and that it was almost impossible for it to have come out of a lab. The Polygraph 2021 in Review: China Led in Global Disinformation On COVID-19 Pandemic
The Chinese blamed the pandemic on frozen meat imported from Barcelona, Spain. Daszak blamed it on frozen meat from China. Daszak was also behind this letter in the Lancet, a highly respected medical journal, which read in part "Conspiracy theories do nothing but create fear, rumours, and prejudice that jeopardise our global collaboration in the fight against this virus." Daszak has recused himself from Lancet Covid-19 Commission after hearing he was going to be fired!
GUPTA: The thing I think that struck me it was so early. How were you so certain at that point to go ahead and label anything that was not a natural origin a conspiracy theory?
DASZAK: Well, because the theory at the time was this is a bio- engineered virus.
GUPTA: So this was not in any way to take off the possibility that this could have still leaked from a lab even if it had not been bio- engineered?
DASZAK: That was not what was being said at the time. What was being said at the time is this is a bio-engineered virus that had been released by Chinese scientists.

In 1968, Dr. Anthony Fauci came to NIH as a clinical associate in the NIAID Laboratory of Clinical Investigation. In 1980, he was appointed chief of the Laboratory of Immunoregulation, a position he still holds. Dr. Fauci became director of NIAID in 1984 a position he still holds earning him the nickname J. Edgar Fauci for his long tenures. In when Fauci earned $417,608, making him the highest-paid federal employee at the time. NIH is the largest source of funding for medical research in the world National Institute of Health (NIH) National Institute of Allergy and Infectious Diseases (NIAID) goal is "To support a discrete, specified, circumscribed project to be performed by the named investigator(s) in an area representing his specific interest and competencies" Research Project (R01) Project 5R01AI110964-04 Application 9320765 Study Section:Clinical Research and Field Studies of Infectious Diseases Study Section (CRFS) Program Officer Erik J Stemmy approved Daszak's grants.
Dr. Erik Joseph Stemmy, PhD erik.stemmy@nih.gov BG 560 1FL RM 8E18 5601 FISHERS LN ROCKVILLE MD 20852 Mail Stop: 9825 Phone: 240-627-3380 NIAID (National Institute of Allergy and Infectious Diseases) VIRAL RESPIRATORY DISEASES SECTION (HNM563)
Eric Stemmy @NIH @NIAIDFunding at #RISymposium2016 ! HPRU RI is built upon collaboration, so we were thrilled to hear of more opportunities! pic.twitter.com/8f5VV4XlT1
— HPRU Resp Infections (@HPRURespiratory) September 26, 2016
GoF/GoT research out-sourced to China was approved by Dr. Erik Joseph Stemmy, PhD, who worked under NIH / NIAID head Anthony Fauci, and was in charge of vetting Viral Respiratory Diseases Section grants. Obama had issued an Executive Order banning these proceedures and these two highly erudite men knew farming the experiments out to China was a way of circumventing the moratorium. Violation of an Executive Order by a Federal Government employee can be a crime but in most instances, its the same penalty any employee suffers when he defies the boss: a reprimand, a demotion, firing. If you are a civil servant the head of your Department will bring administrative charges against you (as prescribed by law) based on the severity of the offense. But here's the catch "The Obama decree prevented the start of new programs, although the program started in 2014 before the Obamian moratorium, via the EcoHealth Alliance, had already secured $ 3.7 million to the Chinese researchers in Wuhan to continue deepening their studies of "epidemiological surveillance" that included the work to gain function until 2019. Part of the research was therefore carried out at the Wuhan Institute of Virology, outside the reach or control of the North American authorities, as Daszak clearly reveals in the interview we discussed. in the beginning. That is, after the deliberation process, the moratorium had been lifted in December 2017 by the Trump administration, but the previous situation of complete freedom was not returned for these investigations. What was done was to create a governmental control mechanism, a panel of experts called P3CO, to which the NIH should refer any project that includes profit of function or similar risks, to be scrutinized before the corresponding funding is granted. Quite simply, when Fauci sent the second phase of the coronavirus gain-of-function project through the EcoHealth Alliance - another $ 3.7 million for another five years, 2019-2024, for the WIV - he managed to avoid that it be raised to the consideration of the panel of experts. He managed not to notify the panel, and when he was asked to account for it much later, he argued that he did not do so because the work did not involve any biosafety danger. Extramural
When questioned by a U. S. Senator Rand Paul*, Dr. Fauci said he didn't know what types of experiments were being conducted at the WIV, although in order to get grant money the recipient has to specify what the money is for as we can see from the document reproduced below entitled "The Possibility that the COVID-19 Virus had a Wuhan Laboratory Origin by Peter Daszak." Doc Fauci's latest in the NYT: "There is this so-called intelligence that three members of the human lab were ill, requiring hospitalization. And I really wonder what the strength of that intelligence is, what the confidence in it is. It certainly has gotten bantered around. And I have said that if the intelligence is firm and real, then you should make sure we find out what the health records of those sick people are. But if its very weak, flimsy intelligence, then theres nothing there. I always keep an open mind. I feel, as do the overwhelming majority of scientists who have knowledge of virology and knowledge of evolutionary biology, that the most likely explanation for this is a natural leap from an animal reservoir to a human." Does Fauci really expect China to give us those records? Of course the Chinese will deny they exist or falsify them. They don't want to take the blame for the pandemic.
*Rand (named after the South African currency) is the son of target="_blank">Ron Paul a crypto-Nazi connected with the now defunct Liberty Lobby. It is a shame he is Fauci's #1 foe or I would work with him as I did with Congressman Henry Gonzalez and Senator Lowell Schweiker, both deceased. Ron Paul edited this statement to make it seem vacccines don't work"We know that the two doses of the vaccine offer very limited protection, if any," is where Paul's quotation ends. "The three doses, with the booster, they offer reasonable protection against hospitalization and deaths. Against deaths, I think very good, and less protection against infection."

Fauci: "We still havent found the natural source of Ebola. And thats been since 1976." The CDC: "African fruit bats are likely involved in the spread of Ebola virus and may even be the source animal (reservoir host). Scientists continue to search for conclusive evidence of the bats role in transmission of Ebola. The most recent Ebola virus to be detected (August 2018), Bombali virus, was identified in samples from bats collected in Sierra Leone." Center For Disease Control But there was no Democratic Republic of Congo Institute for the Study of Ebola viruses where it first broke out and Ebola killed 11,310 not 4 million. The search for the Ebola intermediate species revealed: "Combining results on Non Human Primates from our 4,649 samples and previous studies, it is clear that Ebola virus antibodies are not widespread among NHP which confirms that NHPs are not reservoir species and that if Ebola virus infection in NHPs occurs, few animals survive. During the W.H.O visit, the team reviewed sampling from extensive PCR tests of livestock and poultry from 31 Chinese provinces and 50,000 samples of the wild animals covering 300 different species, but not a single one was found to be infected with SARS-CoV2. https://healthpolicy-watch.news/who-experts-unable-to-find-missing-link-in-sars-cov2-transmission-in-china/
Like Daszak, Fauci often uses the term "conspiracy theory" for things he disagrees with "And any thought of that is total conspiracy theory and total flight of fantasy." There was no conspiracy between Shi, Daszak and Baric. Daszak made Shi and Baric unwitting participants.
 "No actually. I am not convinced about that [the origins of Vovid19]. I think we should continue to investigate what went on in China until we continue to find out to the best of our ability what happened. Certainly, the people who investigated it say it likely was the emergence from an animal reservoir that then infected individuals, but it could have been something else, and we need to find that out. So, you know, thats the reason why I said Im perfectly in favor of any investigation that looks into the origin of the virus." A day after Fauci made this statement The Wall Street Journal elaborated on a report included on this webpage. "Three researchers from Chinas Wuhan Institute of Virology became sick enough in November 2019 that they sought hospital care, according to a previously undisclosed U.S. intelligence report that could add weight to growing calls for a fuller probe of whether the Covid-19 virus may have escaped from the laboratory." Dr. Erik Joseph Stemmy, who also approved Daszak's grants, should also be held accountable for his actions.
"No actually. I am not convinced about that [the origins of Vovid19]. I think we should continue to investigate what went on in China until we continue to find out to the best of our ability what happened. Certainly, the people who investigated it say it likely was the emergence from an animal reservoir that then infected individuals, but it could have been something else, and we need to find that out. So, you know, thats the reason why I said Im perfectly in favor of any investigation that looks into the origin of the virus." A day after Fauci made this statement The Wall Street Journal elaborated on a report included on this webpage. "Three researchers from Chinas Wuhan Institute of Virology became sick enough in November 2019 that they sought hospital care, according to a previously undisclosed U.S. intelligence report that could add weight to growing calls for a fuller probe of whether the Covid-19 virus may have escaped from the laboratory." Dr. Erik Joseph Stemmy, who also approved Daszak's grants, should also be held accountable for his actions.
2024 Report The U.S. government has reason to believe that several researchers inside the WIV became sick in autumn 2019, before the first identified case of the outbreak, with symptoms consistent with both COVID-19 and common seasonal illness. The June 2023 ODNI Assessment entitled, Potential Links Between the Wuhan Institute of Virology and the Origin of the COVID-19 Pandemic, supported this conclusion.
Fauci: Were not talking about the Communist Chinese party. Were not talking about the Chinese military. Were talking about scientists that weve had relationships [with] for years. The Wuhan lab is a very large lab, to the tune of hundreds of millions if not billions of dollars the grant that were talking about was $600,000 over five years. I cant guarantee everything thats going on in the Wuhan lab, we cant do that, but it is our obligation as scientists and public health individuals to study the animal-human interface....A lot of what youre seeing as attacks on me, quite frankly, are attacks on science because all of the things that I have spoken about consistently from the very beginning, have been fundamentally based on science People want to fire me or put me in jail for what Ive done namely, follow the science. The most likely origin is a natural origin from an animal reservoir to human. However, we have not ruled out the possibility that there could have been a leak from the lab. I havent changed my mind.
Dr. Fauci: "Yeah, 20 years ago, people would ask me the same question.[What are you watching for next?] And if you go back over the records, which is easy to do, you would see that I consistently gave the same answer. And that is the emergence of a new virus, a respiratory borne virus that would jump species from an animal host to a human that would have two characteristics. One, it would be highly efficient in transmissibility through the respiratory route. And two, it would have a very high degree of capability of morbidity and mortality for the human population. And unfortunately, were living through my worst nightmare." This is an false equivalence: SARS surfaced 19 years ago yet it was quickly contained. SARS-CoV-19 still is deadly - June 2021 - ten thousand a week dead. NYT
REP. RAND PAUL (R), KENTUCKY: : Dr. Fauci, knowing that it is a crime to lie to Congress do you wish to retract your statement of May 11, where you claimed that the NIH never funded gain-of-function research in Wuhan?
DR. ANTHONY FAUCI, DIRECTOR OF NIH: Senator Paul I have never lied Senator Paul, I have never lied before the Congress and I do not retract that statement. This paper that you are referring to was judged by qualified staff up and down the chain as not being gain-of-function. And if anybody is lying here --Senator, it is you. Senator Paul, I have never lied before the Congress.
PAUL: But (ph) it could have been. So you're saying if you take an animal virus and you increase transmissibility to humans -- you're saying that's not gain-of-function?
FAUCI: What was -- let me finish. Yes, that is correct. And Senator Paul, you do not know what you are talking about, quite frankly. And I want to say that officially. You do not know what you are talking about.
PAUL: This is your definition that you guys wrote. It says that scientific research that increases the transmillity among -- the transmissibility among animals is gain-of-function. They took animal viruses that only occur in animals and they increased their transmissibility to humans. How you can say that is not gain-of- function --
FAUCI: It is not.
PAUL: -- it's a dance and you're dancing around this because you're trying to obscure responsibility for 4 million dying around the world from a pandemic.
FAUCI: I have to -- well, now you're getting into something. If the point that you are making is that the grant that was funded as a subaward from EcoHealth to Wuhan created SARS-CoV-2, that's where are you getting, let me finish. PAUL: We don't know if it did come from the lab, but all the evidence is pointing that it came from the lab. And there will be responsibility for those who funded the lab, including yourself.
FAUCI: -- I totally resent the lie that you are now propagating, Senator, because if you look at the viruses that were used in the experiments that were given in the annual reports that were published in the literature it is molecularly impossible.
PAUL: No one's saying those viruses caused it.
FAUCI: It -- it is -- it is molecularly --
PAUL: No one is alleging that those viruses caused the pandemic. What we're alleging is that gain-of-function research was going on in that lab and NIH funded it.
FAUCI: Can I answer the question?
PAUL: This is your definition that you guys wrote. It says that scientific research that increases the transmillity among -- the transmissibility among animals is gain-of-function. They took animal viruses that only occur in animals and they increased their transmissibility to humans. How you can say that is not gain-of- function --
FAUCI: It is not.
PAUL: -- it's a dance and you're dancing around this because you're trying to obscure responsibility for 4 million dying around the world --from a pandemic. And there will be responsibility for those who funded the lab, including yourself.
GO BACK"The question is whether benefits of such research outweigh risks. The answer is not simple. A highly pathogenic bird flu virus transmissible in humans could arise in ways not predicted by laboratory studies. And it is not clear whether this laboratory virus would behave in humans as it does in ferrets. Nonetheless, new data provide valuable insights that can inform influenza preparedness and help delineate the principles of virus transmission between species. Given these uncertainties, important information and insights can come from generating a potentially dangerous virus in the laboratory. While the World Health Organization and the Centers for Disease Control (CDC) and Prevention provide excellent public health surveillance for novel influenza strains, influenza outbreaks still occur suddenly and in unexpected places. The recent H1N1 pandemic exemplifies the problem: In 2009, a new influenza virus emerged. It was shown to have originated from an animal reservoir, and it spread so rapidly that it strained the pharmaceutical industrys capacity to prepare vaccines fast enough to blunt its spread. We do not fully understand the underlying factors that allow influenza viruses to be transmitted efficiently in humans after they emerge from different species. The ferret transmission studies were intended in part to fill these important gaps in knowledge. Understanding the biology of influenza virus transmission has implications for outbreak prediction, prevention and treatment. In defining the mutations required for mammalian transmission, public health officials are provided with genetic signatures that, like fingerprints, could help scientists more readily identify newly emergent, potentially harmful viruses, track their spread and detect threatening outbreaks. The ability to identify such viruses even a few months faster than by conventional surveillance provides critical time to slow or stop an outbreak. For example, the CDC implements public health protective measures and stockpiles antiviral drugs. Identifying threatening viruses can also facilitate the early stages of manufacturing vaccines that protect against such a virus in advance of an outbreak. In addition, determining the molecular Achilles heel of these viruses can allow scientists to identify novel antiviral drug targets that could be used to prevent infection in those at risk or to better treat those who become infected. Decades of experience tells us that disseminating information gained through biomedical research to legitimate scientists and health officials provides a critical foundation for generating appropriate countermeasures and, ultimately, protecting the public health."
https://www.washingtonpost.com/opinions/a-flu-virus-risk-worth-taking/2011/12/30/gIQAM9sNRP_story.html
"One of the goals of pandemic influenza research is to recognize and anticipate how viruses are evolving in the wild toward a phenotype that is dangerous to humans, thereby staying one step ahead of potential pandemics. In this regard, compelling research questions relevant to global health and pandemic preparedness include determining whether highly pathogenic viruses, such as H5N1, have the ability to mutate and/or reassort with another influenza virus to become readily transmissible by the airborne route among humans. If so,
"Within this context, global attention has been paid recently to two NIH-funded studies of H5N1 transmissibility and pathogenesis in ferrets. In those studies, H5N1 viruses were made transmissible via respiratory droplets among ferrets by engineering the virus; well-described and published protocols including reverse genetics, reassortment, and passaging of viruses in mammals were used. Manuscripts describing the studies have generated an unprecedented degree of discussion, concern, and disagreement among scientists, as well as the public, regarding whether the experiments should have been performed in the first place and whether they should be published in their entirety. Major sources of concern have been that the results might be used by bioterrorists to harm the public or that the virus might accidentally escape and cause a pandemic."
"However, whenever one deliberately manipulates a virus or a microbe, it is always possible, at least theoretically, that the research results could be used by bioterrorists to intentionally cause harm, or that an accidental release of a pathogen from a laboratory could inadvertently cause harm. Such research is referred to as dual-use research, as the research potentially has both positive and negative applications. A particular subset of dual-use research is referred to as dual-use research of concern or DURC. DURC is defined as life sciences research that, on the basis of current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that can be directly misapplied to pose a significant threat with broad potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, materiel, or national security. If a particular experiment is identified as DURC, that designation does not inherently mean that such research should be prohibited or not widely published. However, it does call for us to balance carefully the benefit of the research to public health, the biosafety and biosecurity conditions under which the research is conducted, and the potential risk that the knowledge gained from such research may fall into the hands of individuals with ill intent. Research that could enhance the transmissibility of H5N1viruses clearly is DURC."
"In this regard, the question of whether to publish the two H5N1 studies in ferrets has been intensively discussed by an independent federal advisory committee known as the National Science Advisory Board for Biosecurity (NSABB). On the basis of their recommendations and other evaluations, the U.S. government agreed that the research is important for the public health and should be published. However, important lessons were learned along the way and, appropriately, triggered an examination of our approach concerning the conduct, oversight, and communication of DURC. In this regard, the U.S. government announced on March 29, 2012 the U.S. Government Policy for Oversight of Life Sciences Dual Use Research of Concern. This policy document outlines, for federal departments and agencies that conduct or fund life sciences research, steps to determine whether projects fall under the definition of DURC, to assess the risks and benefits of these projects, to review them regularly, and to develop risk mitigation plans. In the process of weighing the potential risks and benefits of publishing these two manuscripts, it also became clear that, when possible, it is critical to identify research with DURC potential before the initiation of the project and, certainly, before the results are submitted for publication. Such monitoring in the case of NIH-funded research requires the concerted effort of all involved, including scientists applying for or in receipt of NIH funding and NIH program officials. Additional guidelines will be needed as well to assist biosafety committees in evaluating DURC at the institutions where the research is conducted. https://science.sciencemag.org/content/336/6088/1522.ful 22 Jun 2012
"We have not funded gain of function research on this virus in the Wuhan Institute of Virology. No matter how many times you say it, it didn't happen." -- Fauci to Rand Paul pic.twitter.com/f0kMzGeP1B
— Aaron Rupar (@atrupar) May 11, 2021
In February, 2020, 27 public health experts co-authored a Correspondence in The Lancet (Statement in support of the scientists, public health professionals, and medical professionals of China combatting COVID-19), supporting health professionals and physicians in China during the early stages of the COVID-19 pandemic. In this letter, the authors declared no competing interests. Some readers have questioned the validity of this disclosure, particularly as it relates to one of the authors, Peter Daszak. In line with guidance from the International Committee of Medical Journal Editors, medical journals ask authors to report financial and non-financial relationships that may be relevant to interpreting the content of their manuscript. There may be differences in opinion as to what constitutes a competing interest. Transparent reporting allows readers to make judgments about these interests. Readers, in turn, have their own interests that could influence their evaluation of the work in question. With these facts in mind, The Lancet invited the 27 authors of the letter to re-evaluate their competing interests. Peter Daszak has expanded on his disclosure statements for three pieces relating to COVID-19 that he co-authored or contributed to in The Lancetthe February, 2020, Correspondence, as well as a Commission Statement and a Comment for the Lancet COVID-19 Commission. The updated disclosure statement from Peter Daszak is:
Peter Daszak's remuneration is paid solely in the form of a salary from EcoHealth Alliance, a 501(c)(3) non-profit organisation. EcoHealth Alliance's mission is to develop science-based solutions to prevent pandemics and promote conservation. Funding for this work comes from a range of US Government funding agencies and non-governmental sources. All past and current funders are listed publicly, and full financial accounts are filed annually and published. EcoHealth Alliance's work in China was previously funded by the US National Institutes of Health (NIH) and the United States Agency for International Development (USAID). Neither Peter Daszak nor EcoHealth Alliance have received funding from the People's Republic of China. Peter Daszak joined the WHOChina joint global study on the animal origins of SARS-CoV-2 towards the end of 2020 and is currently a member. As per WHO rules, this work is undertaken as an independent expert in a private capacity, not as an EcoHealth Alliance staff member. The work conducted by this study was published in March, 2021. EcoHealth Alliance's work in China includes collaboration with a range of universities and governmental health and environmental science organisations, all of which are listed in prior publications, three of which received funding from US federal agencies as part of EcoHealth Alliance grants or cooperative agreements, as publicly reported by NIH. EcoHealth Alliance's work in China is currently unfunded. All federally funded subcontractees are assessed and approved by the respective US federal agencies in advance and all funding sources are acknowledged in scientific publications as appropriate. EcoHealth Alliance's work in China involves assessing the risk of viral spillover across the wildlifelivestockhuman interface, and includes behavioural and serological surveys of people, and ecological and virological analyses of animals. This work includes the identification of viral sequences in bat samples, and has resulted in the isolation of three bat SARS-related coronaviruses that are now used as reagents to test therapeutics and vaccines. It also includes the production of a small number of recombinant bat coronaviruses to analyse cell entry and other characteristics of bat coronaviruses for which only the genetic sequences are available. NIH reviewed the planned recombinant virus work and deemed it does not meet the criteria that would warrant further specific review by its Potential Pandemic Pathogen Care and Oversight (P3CO) committee. All of EcoHealth Alliance's work is reviewed and approved by appropriate research ethics committees, Institutional Animal Care and Use Committee, Institutional Review Boards for biomedical research involving human subjects, P3CO oversight administrators, and biosafety committees, as listed on all relevant publications. [Daszak is saying that Fauci approved the experiments that this researcher believes created SARS-CoV-19 and did not subject the to review by Potential Pandemic Pathogen Care and Oversight (P3CO) committee. No wonder Fauci is a proponent of Zoonotics.]
The names of the P3CO scientists are classified and their approval only applies to USG sponsored projects. "The secrecy of the screening process was the most worrisome part of government oversight of potentially dangerous research, according to Angela Rasmussen, a virologist at University of Saskatchewans Vaccine and Infectious Disease Organization, who was doing research in the United States at that time. Its not clear how they decide whats acceptable gain of function and whats not, she said. However, the star chamber nature of the process was not its biggest problem, said Richard Ebright, a molecular biologist at Rutgers who has also been one of the most vocal proponents of the lab leak theory, and a longtime advocate of stricter control of research on dangerous pathogens. An even bigger issue, he said, was that gain-of-function research was simply not being screened in accordance with the policy established by H.H.S., which includes the National Institute of Allergy and Infectious Diseases, run by Dr. Fauci. The ideal solution, he said, would be the creation of an independent body to provide the oversight of dangerous pathogen research, similar to what the Nuclear Regulatory Commission does for studies of radioactive materials.
https://www.nytimes.com/2021/06/20/science/covid-lab-leak-wuhan.html

 Why in the devil create a novel virus capable of causing a pandemic? Former New York Times journalist Nick Wade: "Ever since virologists gained the tools for manipulating a viruss genes, they have argued they could get ahead of a potential pandemic by exploring how close a given animal virus might be to making the jump to humans. And that justified lab experiments in enhancing the ability of dangerous animal viruses to infect people, virologists asserted. With this rationale, they have recreated the 1918 flu virus, shown how the almost extinct polio virus can be synthesized from its published DNA sequence, and introduced a smallpox gene into a related virus. These enhancements of viral capabilities are known as gain-of-function, gain-of-threat, experiments. With coronaviruses, there was particular interest in the Spike Proteins, which jut out all around the spherical surface of the virus and pretty much determine which species of animal it will target. In 2000 Dutch researchers, for instance, earned the gratitude of rodents everywhere by genetically engineering the Spike Protein of a mouse coronavirus so that it would attack only cats." Daszak: "Excellent thread debunking another line of argument in Wade's biased, disingenuous, & wildly erroneous piece. So, in just 1 day of Twitter responses, an @NIH statement that they did NOT fund GoF studies at WIV & the Furin cleavage site is NOT a 'smoking gun'. I guess Wade got paid & the conspiracists got their chance to point fingers. Meanwhile, somewhere in a wildlife farm in SE Asia..."
Why in the devil create a novel virus capable of causing a pandemic? Former New York Times journalist Nick Wade: "Ever since virologists gained the tools for manipulating a viruss genes, they have argued they could get ahead of a potential pandemic by exploring how close a given animal virus might be to making the jump to humans. And that justified lab experiments in enhancing the ability of dangerous animal viruses to infect people, virologists asserted. With this rationale, they have recreated the 1918 flu virus, shown how the almost extinct polio virus can be synthesized from its published DNA sequence, and introduced a smallpox gene into a related virus. These enhancements of viral capabilities are known as gain-of-function, gain-of-threat, experiments. With coronaviruses, there was particular interest in the Spike Proteins, which jut out all around the spherical surface of the virus and pretty much determine which species of animal it will target. In 2000 Dutch researchers, for instance, earned the gratitude of rodents everywhere by genetically engineering the Spike Protein of a mouse coronavirus so that it would attack only cats." Daszak: "Excellent thread debunking another line of argument in Wade's biased, disingenuous, & wildly erroneous piece. So, in just 1 day of Twitter responses, an @NIH statement that they did NOT fund GoF studies at WIV & the Furin cleavage site is NOT a 'smoking gun'. I guess Wade got paid & the conspiracists got their chance to point fingers. Meanwhile, somewhere in a wildlife farm in SE Asia..."
https://nicholaswade.medium.com/origin-of-covid-following-the-clues
Using NIH grants and later Daszak's re-purposed grant money, Shi Zheng Li conducted experiments based on a 2005 report by herself and Daszak that proved bat SARS-related CoV (SARSr-CoV) was closely related to human SARS-CoV, implying a bat origin of human SARS. Since then, more bat SARSr-CoV isolates were identified in China. Genome identities of these other bat SARSr-CoVs are as high as 92% to human SARS-CoV, but their major receptor binding spike proteins cannot use the human virus entry receptor ACE2 cells that line the lungs. Shi Zheng Li's motivation was to discover which of these viruses would mutate allowing it to enter ACE-2 receptors. Using Gain-of-Threat bio-engineering the scientists at the WIV altered the receptor binding spike so it do just that making it a threat to humans. Shi Zheng Li's rational behind this was an effort to develop a vaccine for this new virus before there was a spillover from bats to an intermediate species and then to human beings or directly from bats to human beings but the road to Hell is paved with good intentions. Nick Wade contacted the National Institute of Allergy and Infectious Diseases NIAID about this funding and it responded, At no time did NIAID fund gain-of-function research to be conducted at WIV. As to the question of whether the Chinese lab did perform such research, NIAID says: It is impossible for us to be aware of nor can we account for all of their activities. The money was laundered through Daszak but originally came from NIAID. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6466186/
Microbiologists were divided into two camps ProGoFer's and Anti-GoFers. Fauci was a big proGoFer. In fact, when a controversy arose over making Avian Flu transmittable to humans Fauci wrote an editorial in WAPO defending it. If Fauci hadn't been a supporter of GoF / GoT and cracked down on Daszak, reported him to the FBI, instead of looking the other way, none of this would have happened. Fauci could have cut off the subaward grant money going to the WIV for years INTERCEPT DOC page 91 et.al. in a heartbeat. Not doing so, as a USG employees, Fauci, and his Deputy, Erik Stemmy, who signed off on the grants, were liable for violating the Executive Order banning GoF. Not only will either of these men face any criminal charges for their actions, but will remain in their NIH positions.
"If one weighs the benefits verses risk of creating highly transmissible and highly fatal flu strains it should be clear that this research is better not done. If it is, then, at the very least, only labs with the highest level of safety protocols (P4) should be allowed to work with such strains, lesser safeguards are a breach of common sense. It is not an over reaction to think that a release of the most virulent strains might result in a pandemic which would be much worse than the pandemic of 1917, which killed more than 20 million people. Many strains being worked with today have a much higher fatality rate and could result in hundreds of millions, or billions of deaths." NYT comments: David Green Brooklyn NY June 30, 2014
 SEPTEMBER 2014 David Relman: "My greatest fear is that someone will create a highly contagious and highly pathogenic infectious agent that does not currently exist in nature, publish its genetic blueprint, allow it to escape the laboratory by accident, or else enable a malevolent person or persons to synthesize the agent with the intention of releasing it in a deliberate manner. Although these may be unlikely scenarios, they could have catastrophic consequences, which is why I and others feel that we need to sensitize everyone to these possibilities and decide how to manage these risks ahead of time. I want to be clear: I am not opposed to laboratory work on dangerous pathogens, especially if they are known to exist in nature. Rather, I am opposed to high-risk experiments and, in particular, those that seek to create novel, dangerous pathogens that cannot be justified by well-founded expectations of near-term, critical benefits for public health benefits that clearly outweigh the risks, and benefits that cannot be achieved through other means."
SEPTEMBER 2014 David Relman: "My greatest fear is that someone will create a highly contagious and highly pathogenic infectious agent that does not currently exist in nature, publish its genetic blueprint, allow it to escape the laboratory by accident, or else enable a malevolent person or persons to synthesize the agent with the intention of releasing it in a deliberate manner. Although these may be unlikely scenarios, they could have catastrophic consequences, which is why I and others feel that we need to sensitize everyone to these possibilities and decide how to manage these risks ahead of time. I want to be clear: I am not opposed to laboratory work on dangerous pathogens, especially if they are known to exist in nature. Rather, I am opposed to high-risk experiments and, in particular, those that seek to create novel, dangerous pathogens that cannot be justified by well-founded expectations of near-term, critical benefits for public health benefits that clearly outweigh the risks, and benefits that cannot be achieved through other means."
https://med.stanford.edu/news/all-news/2014/09/5-questions--david-relman-on-risks-of-creating-new-pathogens.html
 Dr. Latham: "Not only are there clear reasons to doubt that the wet market was the source, but it also requires you to accept the most staggering coincidence. After all, given that wet markets are widespread in China indeed across much of Asia, how remarkable is it that the viruss zoonotic leap from animal to human just happened to take place in one that has a lab under 300 meters away, and another within 10 kilometres, that is researching not just bat coronaviruses, but the most closely related ones to the outbreak?" If you believe this is co-incidence you just fell off a turnip truck. Latham believes that SARS-CoV-19 close relative RaTG13 mutated in the miner's lungs in which it was discovered, was brought to the WIV and escaped.
Dr. Latham: "Not only are there clear reasons to doubt that the wet market was the source, but it also requires you to accept the most staggering coincidence. After all, given that wet markets are widespread in China indeed across much of Asia, how remarkable is it that the viruss zoonotic leap from animal to human just happened to take place in one that has a lab under 300 meters away, and another within 10 kilometres, that is researching not just bat coronaviruses, but the most closely related ones to the outbreak?" If you believe this is co-incidence you just fell off a turnip truck. Latham believes that SARS-CoV-19 close relative RaTG13 mutated in the miner's lungs in which it was discovered, was brought to the WIV and escaped.
https://www.independentsciencenews.org/commentaries/a-proposed-origin-for-sars-cov-2-and-the-covid-19-pandemic/
GO BACK
The documents make it clear that assertions by the NIH Director, Francis Collins, and the NIAID Director, Anthony Fauci, that the NIH did not support gain-of-function research or potential pandemic pathogen enhancement at WIV are untruthful.
— Richard H. Ebright (@R_H_Ebright) September 7, 2021
The materials further reveal that the the grants also supported the construction--in Wuhan--of novel chimeric MERS-related coronaviruses that combined spike genes from one MERS-related coronavirus with genetic information from another MERS-related coronavirus.
— Richard H. Ebright (@R_H_Ebright) September 7, 2021
 Richard Ebright, a molecular biologist and biodefence expert at Rutgers University in Piscataway, New Jersey, told the Daily Mail that the odds of this new virus containing such unusual features and occurring naturally were possible but improbable. Ebright said scientists at the Wuhan Institute of Virology were creating chimeric coronaviruses (new hybrid micro-organisms) and seeking funding to test their ability to infect human cells while using procedures that leave no sign of human manipulation. Asked about the chance of a leak, he replied: "There definitely is a possibility. But there is no basis to say a high probability. It is clear that the Wuhan Institute of Virology was systematically constructing novel chimeric coronaviruses and was assessing their ability to infect human cells and human-ACE2-expressing mice.
Richard Ebright, a molecular biologist and biodefence expert at Rutgers University in Piscataway, New Jersey, told the Daily Mail that the odds of this new virus containing such unusual features and occurring naturally were possible but improbable. Ebright said scientists at the Wuhan Institute of Virology were creating chimeric coronaviruses (new hybrid micro-organisms) and seeking funding to test their ability to infect human cells while using procedures that leave no sign of human manipulation. Asked about the chance of a leak, he replied: "There definitely is a possibility. But there is no basis to say a high probability. It is clear that the Wuhan Institute of Virology was systematically constructing novel chimeric coronaviruses and was assessing their ability to infect human cells and human-ACE2-expressing mice.
"Other experiments in the University of North Carolina study show that the virus in wild bats would need to evolve to pose any threat to humans a change that may never happen, although it cannot be ruled out. Baric and his team reconstructed the wild virus from its genome sequence and found that it grew poorly in human cell cultures and caused no significant disease in mice. The only impact of this work is the creation, in a lab, of a new, non-natural risk, agrees Richard Ebright. Both Ebright and Wain-Hobson are long-standing critics of gain-of-function research. In their paper, the study authors also concede that funders may think twice about allowing such experiments in the future. "Scientific review panels may deem similar studies building chimeric viruses based on circulating strains too risky to pursue," they write, adding that discussion is needed as to "whether these types of chimeric virus studies warrant further investigation versus the inherent risks involved.
 Simon Wain-Hobson, a virologist at the Pasteur Institute in Paris, points out that the researchers have created a novel virus that grows remarkably well in human cells. If the virus escaped, nobody could predict the trajectory, he says. Wain-Hobson disapproves of the study because, he says, it provides little benefit, and reveals little about the risk that the wild SHC014 virus in bats poses to humans. Other scientists at the Pasteur Institute are part of the cover-up because they built the WIV. "Coronavirus outbreak in France not comes directly from China or Italy, say French scientists A genetic study conducted by scientists from France's Pasteur Institute suggests that the spread of the coronavirus epidemic in France is very little linked to cases imported from China." http://www.xinhuanet.com/english/2020-04/30/c_139018917.htm
Simon Wain-Hobson, a virologist at the Pasteur Institute in Paris, points out that the researchers have created a novel virus that grows remarkably well in human cells. If the virus escaped, nobody could predict the trajectory, he says. Wain-Hobson disapproves of the study because, he says, it provides little benefit, and reveals little about the risk that the wild SHC014 virus in bats poses to humans. Other scientists at the Pasteur Institute are part of the cover-up because they built the WIV. "Coronavirus outbreak in France not comes directly from China or Italy, say French scientists A genetic study conducted by scientists from France's Pasteur Institute suggests that the spread of the coronavirus epidemic in France is very little linked to cases imported from China." http://www.xinhuanet.com/english/2020-04/30/c_139018917.htm
ALSO SEE: https://www.nature.com/news/engineered-bat-virus-stirs-debate-over-risky-research
https://peterdaszak.com/wade.htm
https://www.nature.com/articles/nm.3985

England & Wales, Civil Registration Marriage Index, 1916-2005 Peter Daszak Spouse JANET D COTTINGHAM https://twitter.com/janetcottingha1 (Married Feb 1997) Is this a co-incidence or is Daszak hiding his Ukrainian background? Daszak's daughter interned at Eco-Health Alliance.


Kaspar Sebastian Bohdan Daszak is 20 years old and lives in an apartment in Stockholm. He lives with his mother Jacqueline Miura Daszak and John Ramstedt whose grandfather Gustaf John Ramstedt was a Finnish Charge D'Affairs to Japan.. BORN 8/27/2000. John's son. His home is valued at $1,627,859.80 United States Dollars
https://www.hitta.se/kaspar+daszak/stockholm/person/VgPwmnoooK
CV: Peter Daszak holds a B.Sc. in Zoology in 1987 at University College of North Wales (UCNW), AKA Bangor University. Bangor is ranked #601-650 in QS Global World Rankings 2021 and 61st in the United Kingdom. Bio sketch Intercept docs "Bangor University (UK) BSc. (hons) 07/86 Zoology"
CV: Daszak earned a Ph.D. in parasitic infectious diseases in 1994 at University of East London. One would think he would return to graduate school to attain a degree in microbiology or infectious diseases but he lacks the mental capacity. Bio sketch Intercept docs: University of East London (UK) Ph.D 03/93 Infectious Diseases

Q. How did you become involved in zoology?
A. I was always interested in nature as a kid so I wanted to do zoology ever since I was eight years old I wanted to be a zoologist and I was on that truck. I did a zoology degree. I was going to work. I'm a reptile fan. I wanted to work on reptiles and then in England you have in your third year, you have to pick a research project, and I was late to the table and the only one left on reptiles was no good behavior stuff. It was ultra structure of a parasite in a lizard's gallbladder. I'll do it anyway but it was it was so fascinating I just got hooked on research. I got hooked on seeing something for the first time and getting a result that tells you something that no one else has seen it. I found that fascinating. I got a PhD in a similar field. I got a PhD in coccidiosis now that was the parasite. I did it in England, in London, near University of East London.
Q. Okay and did you do a postdoc after that?
A. Yeah I did a couple of postdocs. It was in a pretty bad time in England. There were one year postdocs and kept getting refunded but it's not great support.
Q. What areas were they in?
A. Similar stuff parasitology. I was a sort of general because I could do what from across country. I volunteered at CDC and it was during the Nipah virus outbreak and I was in the lab doing electronic respond the first samples.

Subject: Confidential -A new bat-origin coronavirus emerging in pigs in China discovered under our NIAID ROl Importance: High
Dear Dr Fauci and NIAID colleagues,
It was a pleasure to meet you again today. I've attached an unpublished paper, currently in the second round of review with Nature that describes a novel bat-origin Coronavirus (SADS-CoV: Swine Acute Diarrheal Syndrome coronavirus) that recently spilled over into pig farms in Southern China, leading to the death of over 25,000 piglets in 5+ farms in Guandong Province. The virus originates in the same group of bats as SARS-CoV, and emerged in the same place. It's not known to be zoonotic (we've tested 35+ pig farm workers with an antibody assay and none are positive. The pig farm owners tell us the virus is now under control, thanks to culling and separation of infected herds. It's not yet known if this virus has appeared elsewhere, but we are looking. We're also doing assays to find out if it can infect human cells in the lab- so far no evidence of this. I hope this paper is of interest. You should know that this work was supported by a NIAID ROl that Erik Stemmy is the Program Officer for, and that I'm PI on, with Zhengli Shi as co-Pl. If you want any other information at all, please don't hesitate to email or call and I'd be happy to come over to NIAID to brief you further. I'll also let you know if/when it will be published so that we can try to foster some publicity as appropriate. Cheers, Peter Judicial Watch
Laura A. Pone Grants Management Officer NATIONAL INSTITUTE OF ALLERGY AND INFECTIOUS DISEASES
Grants Management Specialist: Jenny L. Greer
 Grants Management Specialist: Adam Graham Phone: Fax: 301-493-0597 Assistant Grants Policy Officer at National Institutes of Health, Office of Extramural Research Dates Employed Mar 2016 Jan 2019 Employment Duration 2 yrs 11 mos
Grants Management Specialist: Adam Graham Phone: Fax: 301-493-0597 Assistant Grants Policy Officer at National Institutes of Health, Office of Extramural Research Dates Employed Mar 2016 Jan 2019 Employment Duration 2 yrs 11 mos
Evaluate management aspects of grant applications, including development of criteria and standards for planning, implementation, and evaluation of grant proposals.
Monitor and execute all phases of awards of numerous mechanisms, including multi-project Cooperative Agreements, Small Business Innovative Research, Small Business Technology Transfer, Career Development Awards, Fellowships, Conference, and Training Program Grants.
Provide guidance on statutes, regulations, policies, and procedures relevant to the award program in general and individual applications and proposals.
Communications liaison for scientific program officers, institutional business officials, and principal investigators.
Grants Management Coordinator for:
o Global Infectious Disease Research Administrations Development Awards
o Bi-Lateral Research Programs - China, India, Brazil, Indonesia
o Vaccine Action Program (VAP) Small Research Grant Program (R03)
o Systems Biology: The Next Generation for Infectious Diseases (Cooperative Agreements)
American University Degree Master of Arts (M.A.) Field Of Study International Economic Relations Dates attended or expected graduation 2007 2010 Specialization in the East Asia Region Significant Research Paper - Systemic Risk and Financial Regulation Recipient of the School of International Service Study Abroad Scholarship for study abroad in MalaysiaUniversity of Florida Degree Bachelor of Arts (B.A.) Field Of Study Political Science & Philosophy Dates attended or expected graduation 2001 2005
Grants Management Specialist: Shaun W Gratton Mount Airy, MD Age: 29 years old (301) 831-0856 kgratton@aol.com
Although Baric's "no-see-um" method was used to create SARS-CoV-19 making it almost impossible to determine the virus was unnatural Shi left some fingerprints:
"Notably, this approach surprisingly revealed that the binding energy between SARS-CoV-2 Spike Protein and ACE2 was highest for humans out of all species tested, suggesting that SARS-CoV-2 Spike Protein is uniquely evolved to bind and infect cells expressing human ACE2. This finding is particularly surprising as, typically, a virus would be expected to have highest affinity for the receptor in its original host species, e.g. bat, with a lower initial binding affinity for the receptor of any new host, e.g. humans. However, in this case, the affinity of SARS-CoV-2 is higher for humans than for the putative original host species, bats, or for any potential intermediary host species. Although bats carry many coronaviruses including SARS-CoV, a relative of SARS-CoV-2, direct evidence for existence of SARS-CoV-2 in bats has not been found. As highlighted by our data, the binding strength of SARS-CoV-2 for bat ACE2 is considerably lower than for human ACE2,suggesting that even if SARS-CoV-2 did originally arise from a bat precursor it must later have adapted its Spike Protein to optimise its binding to human ACE2. There is no current explanation for how, when or where this might have happened. Instances of direct human infection by coronaviruses or other bat viruses is rare with transmission typically involving an intermediate host. For example, lyssaviruses such as Hendra are periodically transmitted from bats to horses and then to humans who contact the infected horse. Similarly, SARS-CoV was shown to be transmitted from bats to civet cats and from them to humans. To date, a virus identical to SARS-CoV-2 has not been identified in bats or any other non-human species, making its origins unclear. To date, the most closely related coronavirus to SARS-CoV-2, is the bat coronavirus, BatCoV RaTG1, which has 96% whole-genome identity to SARS-CoV-2. 50. The fact that SARS-CoV-2 has also not been found in any likely intermediate host raises questions of the origins of the original SARS-CoV-2."
https://arxiv.org/ftp/arxiv/papers/2005/2005.06199.pdf
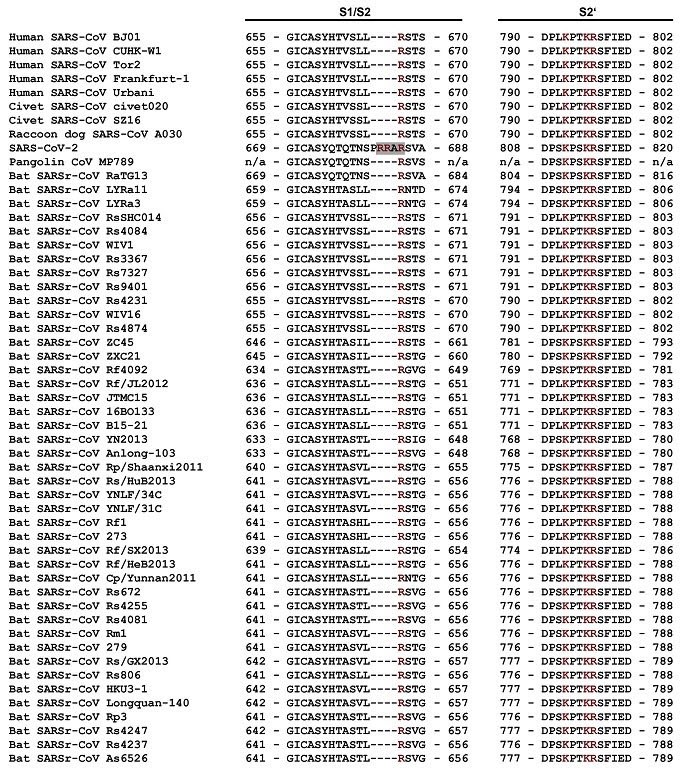
 No clear evolutionary pathway has been identified that would explain the presence of SARS-CoV-2s furin polybasic cleavage site, especially given its enhanced pathogenic significance. It is, therefore, not an unreasonable alternative to assume that the unique furin polybasic cleavage site found in SARS-CoV-2 and in no other close relatives may be the result of genetic manipulation. SARS-CoV-2 has a Furin Polybasic Cleavage Site PRRA that is not found in either bat, and pangolin viruses that were genetically similar to SARS-CoV-2. This insertion interface makes it easy for the virus to infect humans because the spike protein on the virus immediately interacts with furin at the polybasic cleavage site. Furin is an enzyme found abundantly in the body. Furin assists the virus envelope in merging with the cell membrane to infect it. Similar furin-like sites were also found in other viruses such as HIV and Ebola causing scientists to explore their similarities to SARS-CoV-2. This insertion allows the virus to skip animal to human transmission infect humans. SARS-CoV-2 has not been found in nature as SARS CoV-1 was. One can say it hasn't yet been found in nature but since the Corona Plague scientists have been looking desperately for this missing link. The coronaviruses causing SARS CoV-1 were descended from coronaviruses affecting masked civets and camels. Their genetic similarity was found to be 99 percent. This level of similarity was not found between bat and pangolin viruses or any other living creature, and SARS-CoV-2, because this is a creation of SHI ZHENG LI. Some scientists claim that inserting a new gene in a virus is like substituting a red brick for a black brick in a structure, but new insertion techniques, like passaging, leave no trace of human intervention.
No clear evolutionary pathway has been identified that would explain the presence of SARS-CoV-2s furin polybasic cleavage site, especially given its enhanced pathogenic significance. It is, therefore, not an unreasonable alternative to assume that the unique furin polybasic cleavage site found in SARS-CoV-2 and in no other close relatives may be the result of genetic manipulation. SARS-CoV-2 has a Furin Polybasic Cleavage Site PRRA that is not found in either bat, and pangolin viruses that were genetically similar to SARS-CoV-2. This insertion interface makes it easy for the virus to infect humans because the spike protein on the virus immediately interacts with furin at the polybasic cleavage site. Furin is an enzyme found abundantly in the body. Furin assists the virus envelope in merging with the cell membrane to infect it. Similar furin-like sites were also found in other viruses such as HIV and Ebola causing scientists to explore their similarities to SARS-CoV-2. This insertion allows the virus to skip animal to human transmission infect humans. SARS-CoV-2 has not been found in nature as SARS CoV-1 was. One can say it hasn't yet been found in nature but since the Corona Plague scientists have been looking desperately for this missing link. The coronaviruses causing SARS CoV-1 were descended from coronaviruses affecting masked civets and camels. Their genetic similarity was found to be 99 percent. This level of similarity was not found between bat and pangolin viruses or any other living creature, and SARS-CoV-2, because this is a creation of SHI ZHENG LI. Some scientists claim that inserting a new gene in a virus is like substituting a red brick for a black brick in a structure, but new insertion techniques, like passaging, leave no trace of human intervention.
The IC assesses that public claims that some distinguishing features in SARS-CoV-2 are the result of genetic engineering are not diagnostic of genetic engineering. The IC has been evaluating how SARS-CoV-2 could have developed these features and notes that the furin cleavage site (FCS)a region in the spike protein that enables infection and has been the topic of open-source debatecan also be consistent with a natural origin of the virus. We do not fully understand the diversity of natural coronaviruses or how often they recombine, suggesting that there are plausible natural means by which these features in SARS-CoV-2 could have emerged beyond what we currently understand.
For example, the author of an article in April notes the SARS-CoV-2s FCS is unique among known betacoronaviruses. The author argues that such features are rare and so well-adapted for human infection that they are more likely emerged from laboratory work than from natural selection.
Although an IC review of scientific literature has indicated that no known betacoronaviruses in the same subgenus have this FCS in the same region of the spike protein as SARS-CoV-2, similar FCSs are present in the same region of the spike protein as other naturally occurring coronaviruses, according to scientific articles.
Coronaviruses discovered in Laotian bats are surprisingly adept at infecting human cells, showing that such deadly features can indeed evolve outside of a lab. Three of the Laos coronaviruses were unusual: They carried a molecular hook on their surface that was very similar to the hook on the virus that causes Covid-19, called SARS-CoV-2. Like SARS-CoV-2, their hook allowed them to latch onto human cells. That really puts to bed any notion that this virus had to have been concocted, or somehow manipulated in a lab, to be so good at infecting humans, said Michael Worobey, a University of Arizona virologist who was not involved in the work.
With the novel viruses here described, the pool of sequences found in Rhinolopus spp. allows the reconstitution of a genome sufficiently close to that of SARS-CoV-2 without the need to hypothesize recombination or natural selection for increased RBD affinity for hACE2 in an intermediate host before spillover38, nor natural selection in humans following spillover39. However, we found no furin site in any of these viruses on sequences determined from original fecal swab samples, devoid of any bias associated with counterselection of the furin site by amplification in Vero cells. Lack of furin cleavage may be explained by insufficient sampling in bats, or by acquisition of the furin cleavage site through passages of the virus in an alternate host or during an early poorly symptomatic unreported circulation in humans. Finally, where these intergenomic recombinations arose and the epidemiological link with the first human cases remains to be established...Despite the absence of the furin site in these novel bat sarbecoviruses, phylogenetic reconstruction of this fragment, key for the virus tropism and host spectrum, revealed that Laotian R. malayanus BANAL-52, R. pusillus BANAL-103, and R. marshalli BANAL-236 coronaviruses are the closest ancestors of SARS-CoV-2 known to date. Identification of strains of animal origin with a furin cleavage site may require additional sampling...To conclude, our results pinpoint the presence of new bat sarbecoviruses that seem to have the same potential for infecting humans as early strains of SARS-CoV-2. People working in caves, such as guano collectors, or certain ascetic religious communities who spend time in or very close to caves, as well as tourists who visit the caves, are particularly at risk of being exposed. Further investigations are needed to assess if such exposed populations have been infected by one of these viruses, if these infections are associated with symptoms, and whether they could confer protection against subsequent SARS-CoV-2 infections. Zimmer NYT
Zimmer took over the science beat after Donald MacNeil was fired for using the word nigger in a lecture on racist language. While he was still at the Times Donald wrote "I was offended by Nicholas Wade's attacks on Dr. Anthony S. Fauci of the National Institutes of Health and Peter Daszak of the EcoHealth Alliance, both of whom I have known for years; I know both are dedicated to saving lives, and they have always told me the truth or what they honestly believed to be the truth at the time, because evidence sometimes changes. They are now both getting death threats, and that is repulsive."
Zimmer earned his BA from Yale University. He wrote for Discover Magazine and later gained attention in 2013 when he started writing his column Matter in The New York Times. Also see NYT
Authors: Xin Li, Liang Wang, Peipei Liu, Hongying Li, Shuting Huo, Kexin Zong, Shiyan Zhu, , Yuanyuan Guo, Libiao Zhang, Ben Hu, Yu Lan, ALEKSEI CHMURA https://orcid.org/0000-0001-5544-0431, Guizhen Wu, Peter Daszak, William J. Liu and target="_blank">George F. GaoALEKSEI CHMURA: SHADY ECO-CON MAN ECOHEALTH ALLIANCE VICE PRESIDENT WHO WORKS WITH DASZAK ORCHESTRATES ARTICLE TO SHOW FURIN
A NOVEL POTENTIALLY RECOMBINANT RODENT CORONAVIRUS WITH A POLYBASIC CLEAVAGE SITE IN THE SPIKE PROTEIN August 25, 2021
ABSTRACT
"The emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has reignited global interest in animal coronaviruses and their potential for human transmission. While bats are thought to be the wildlife reservoir of SARS-CoV and SARS-CoV-2, the widespread human coronavirus OC43 is thought to have originated in rodents. We sampled 297 rodents and shrews representing eight species in three municipalities of southern China. We report coronavirus prevalence of 23.3% and 0.7% in Guangzhou and Guilin, respectively, with samples from urban areas having significantly higher coronavirus prevalence than those from rural areas. We obtained three coronavirus genome sequences from Rattus norvegicus, including a Betacoronavirus RCoV-GCCDC3, an Alphacoronavirus RCoV-GCCDC5 and a novel Betacoronavirus RCoV-GCCDC4. Recombination analysis suggests that there was a potential recombinant event involving RCoV-GCCDC4, Murine hepatitis virus and Longquan Rl rat coronavirus. Furthermore, we uncovered a polybasic cleavage site RARR in the spike (S) protein of RCoV-GCCDC4, which is dominant in RCoV. These findings provide further information on the potential for inter-species transmission of coronaviruses and demonstrate the value of a One Health approach to virus discovery. Importance: surveillance of viruses among rodents within rural and urban areas of South China identified three rodent coronaviruses RCoV-GCCDC3, 4 and 5, one of which was identified as a novel potentially recombinant coronavirus with a polybasic cleavage site in the spike (S) protein. Through reverse transcription PCR screening of coronaviruses, we found that coronavirus prevalence in urban areas is much higher than that in rural areas. Subsequently, we obtained three coronavirus genome sequences by deep sequencing. After different method-based analyses, we found that RCoV-GCCDC4 was a novel potentially recombinant coronavirus with a polybasic cleavage site in S protein, dominant in RCoV. This newly identified coronavirus RCoV-GCCDC4 with its potentially recombinant genome and polybasic cleavage site provides a new insight into the evolution of coronaviruses. Furthermore, our results provide further information on the potential for inter-species transmission of coronaviruses and demonstrate the necessity of a One Health approach for zoonotic disease surveillance.
 1. Originally identified in 1990, furin is a cellular endoprotease that proteolytically activates many proprotein substrates ranging from pathogenic agents to growth factors, receptors, and extracellular matrix proteins. The furin cleavage site is a small region of SARS-CoV-2 Spike Protein just 12 units of its 30,000-unit genome. A virus usually acquires inserts like this by accidentally exchanging genomic units with another virus when both invade the same cell. But no other known virus in SARS2s group has this 12-unit insert. No one has yet found the human population in which the virus might have evolved this way. The insert also contains entities known as arginine codons, which are common in humans but not in coronaviruses like SARS2. Since 1992 the virology community has known that the one sure way to make a virus deadlier is to give it a furin cleavage site, writes Dr. Steven Quay, a biotech entrepreneur interested in the origins of SARS2. "At least 11 such experiments have been published, including one by Dr. Shi." David Baltimore, an eminent virologist and former president of the California Institute of Technology stated: When I first saw the furin cleavage site in the viral sequence, with its arginine codons, I said to my wife it was the smoking gun for the origin of the virus. These features make a powerful challenge to the idea of a natural origin for SARS2, he said. Baltimore is currently President Emeritus and distinguished Professor of Biology at Caltech. Awarded the Nobel Prize at the age of 37 for research in virology, Baltimore has profoundly influenced national science policy on such issues as recombinant DNA research and the AIDS epidemic. After being presssured by the Fauci Mafia, Baltimore walked some of this back.
1. Originally identified in 1990, furin is a cellular endoprotease that proteolytically activates many proprotein substrates ranging from pathogenic agents to growth factors, receptors, and extracellular matrix proteins. The furin cleavage site is a small region of SARS-CoV-2 Spike Protein just 12 units of its 30,000-unit genome. A virus usually acquires inserts like this by accidentally exchanging genomic units with another virus when both invade the same cell. But no other known virus in SARS2s group has this 12-unit insert. No one has yet found the human population in which the virus might have evolved this way. The insert also contains entities known as arginine codons, which are common in humans but not in coronaviruses like SARS2. Since 1992 the virology community has known that the one sure way to make a virus deadlier is to give it a furin cleavage site, writes Dr. Steven Quay, a biotech entrepreneur interested in the origins of SARS2. "At least 11 such experiments have been published, including one by Dr. Shi." David Baltimore, an eminent virologist and former president of the California Institute of Technology stated: When I first saw the furin cleavage site in the viral sequence, with its arginine codons, I said to my wife it was the smoking gun for the origin of the virus. These features make a powerful challenge to the idea of a natural origin for SARS2, he said. Baltimore is currently President Emeritus and distinguished Professor of Biology at Caltech. Awarded the Nobel Prize at the age of 37 for research in virology, Baltimore has profoundly influenced national science policy on such issues as recombinant DNA research and the AIDS epidemic. After being presssured by the Fauci Mafia, Baltimore walked some of this back.
One indication that this virus was made more deadly and communicable by the Chinese scientists encouraged by Daszak and Fauci is that is doesn't act like any other virus did. It is far more deadly and causes many other symptons other than respiratory disease. It attacks children with a Kawasaki disease - an illness that causes inflammation (swelling and redness) in blood vessels throughout the body. It happens in three phases, and a lasting fever usually is the first sign. The condition most often affects kids younger than 5 years old. National Geographic reports on these weird symptoms but is afraid to even suggest they may be a result of Gain-of-Purpose experiments. Lynne Turner-Stokes, professor of rehabilitation medicine at Kings College, says Covid is a multi-system disease which can potentially affect any organ. It causes microvascular problems and clots. Lungs, brain, skin, kidneys and the nervous system may be affected. Neurological symptoms can be mild (headache) or severe (confusion, delirium, coma). Turner-Stokes says its uncertain why the illness is sometimes so protracted. One explanation is that the bodys immune system goes into overdrive, with an ongoing reaction. Another is that the symptoms are virus-driven. Either way, she says there can be a recrudescence of symptomatology. Or, as she also puts it using more colloquial language, the whole caboodle comes back. The most common post-Covid symptom is parosmia, loss of smell. which scientists attribute to the ACE2 invasion. Paediatric inflammatory multisystem syndrome temporally associated with SARS-CoV-2 (PIMS-TS) is a new, rare, post-infectious complication of SARS-CoV-2 infection in children. In a study of veterans, Covid survivors were 35 percent more likely than other patients to have long-term kidney damage or declines in kidney function.
https://www.nationalgeographic.com/science/2020/05/kawasaki-stroke-why-coronavirus-weirdest-symptoms-are-only-emerging-now-cvd
https://www.theguardian.com/world/2020/may/15/weird-hell-professor-advent-calendar-covid-19-symptoms-paul-garner
Detailed Study of Post Covid Symptoms
"There is a bidirectional relationship between Covid-19 and diabetes. On the one hand, diabetes is associated with an increased risk of severe Covid-19. On the other hand, new-onset diabetes and severe metabolic complications of preexisting diabetes, including diabetic ketoacidosis and hyperosmolarity for which exceptionally high doses of insulin are warranted, have been observed in patients with Covid-19.1-3 These manifestations of diabetes pose challenges in clinical management and suggest a complex pathophysiology of Covid-19related diabetes." https://www.nejm.org/doi/full/10.1056/NEJMc2018688
In general, SARS begins with a high fever (temperature greater than 100.4°F [>38.0°C]). Other symptoms may include headache, an overall feeling of discomfort, and body aches. Some people also have mild respiratory symptoms at the outset. About 10 percent to 20 percent of patients have diarrhea. After 2 to 7 days, SARS patients may develop a dry cough. Most patients develop pneumonia. https://www.cdc.gov/sars/about/fs-sars.html A study published in the Journal of International Audiology that looked at nearly 60 case reports and studies found that 15 percent of adults with Covid-19 reported symptoms of tinnitus. The study of 100 patients from 21 states, published in The Annals of Clinical and Translational Neurology, found that 85 percent of them experienced four or more neurological issues like brain fog, headaches, tingling, muscle pain and dizziness. A small number of Covid patients who had never experienced mental health problems are developing severe psychotic symptoms weeks after contracting the coronavirus. A condition, which the Centers for Disease Control and Prevention are calling Multisystem Inflammatory Syndrome in Children, has shaken widespread confidence that children were largely spared from the pandemic. Instead of targeting lungs as the primary coronavirus infection does, it causes inflammation throughout the body and can cripple the heart.

1970s, Researchers were learning for the first time to move genes from one organism to another to make bacteria produce human insulin. From the start, critics worried that such experiments could accidentally create deadly pathogens if they escaped from labs.
1995 Abstract: Coronaviruses are common respiratory and gastrointestinal pathogens of mammals and birds. Not only do they cause about 1520% of the common colds in humans, they are also occasionally associated with infections of the lower respiratory tract and central nervous system. The prototype, mouse hepatitis virus (MHV), contains a 32 kb genomic RNA which encodes two large orfs at the 5' end, designated orf la and orf lb. Orf lb contains highly conserved polymerase, helicase and metal binding motifs typical of viral RNA polymerases while orf 1 a contains membrane and cysteine rich domains, and serine-and poliovirus 3c-like protease motifs1. The large size of the genome coupled with its unique replication strategy and high recombination frequencies during mixed infection predict a considerable capacity to evolve. Ralph S. Baric Kaisong FuWan Chen / Boyd Yount University of North Carolina
https://link.springer.com/chapter/10.1007/978-1-4615-1899-0_91
OCTOBER 2003 Baric published the results of his research: "A previously undescribed coronavirus (CoV) is the etiologic agent responsible for severe acute respiratory syndrome (SARS). Using a panel of contiguous cDNAs that span the entire genome, we have assembled a full-length cDNA of the SARS-CoV Urbani strain1 and have rescued molecularly cloned SARS viruses (infectious clone SARS-CoV) that contained the expected marker mutations inserted into the component clones. Recombinant viruses2 replicated as efficiently as WT virus and both were inhibited by treatment with the cysteine proteinase inhibitor (2S,3S)-transepoxysuccinyl-L-leucylamido-3-methylbutane ethyl ester. In addition, subgenomic transcripts were initiated from the consensus sequence ACGAAC in both the WT and infectious clone SARS-CoV. Availability of a SARS-CoV full-length cDNA provides a template for manipulation of the viral genome, allowing for the rapid and rational development and testing of candidate vaccines and therapeutics against this important human pathogen. 1. The Urbani strain was sequenced by the US Centers for Communicable Diseases in Atlanta, Georgia. It is a common strain in Asia, named after Carlo Urbani, who was an infectious disease specialist at the World Health Organization's office in the Vietnamese capital, Hanoi. He helped to identify the disease as a coronavirus but later died from it. 2. Viral recombination occurs when viruses of two different parent strains coinfect the same host cell and interact during replication to generate virus progeny that have some genes from both parents.
2004 The Chinese and French governments signed a cooperation agreement on fighting and preventing new diseases, stressing the active cooperation between China and France in the construction of high-level biosafety laboratories and the system construction of biosafety laws and regulations etc. In order to implement the spirit of Sino-French agreement, in 2005, Wuhan Institute of Virology undertook the task of building a national biosafety laboratory of Wuhan, Chinese Academy of Sciences. With nearly 10 years of unremitting efforts, the laboratory completed the physical facilities in January 2015. In August 2016, it obtained the recognition and authentication certificate for the critical protection equipment installation and commissioning.
2004 according to Scientific American, a team from the WIV led by virologist Shi Zhengli began visiting caves in southern China, hoping to find the cause of SARS. They captured bats and took blood, saliva, and fecal samples, and tested the samples for viruses back in Wuhan.
FEBRUARY 2005 ABSTRACT: The genomes of transmissible gastroenteritis virus (TGEV) and Mouse Hepatitis Virus (MHV) have been generated with a novel construction strategy that allows for the assembly of very large RNA and DNA genomes from a panel of contiguous cDNA subclones. Recombinant viruses generated from these methods contained the appropriate marker mutations and replicated as efficiently as wild-type virus. The MHV cloning strategy can also be used to generate recombinant viruses that contain foreign genes or mutations at virtually any given nucleotide. MHV molecular viruses were engineered to express green fluorescent protein (GFP), demonstrating the feasibility of the systematic assembly approach to create recombinant viruses expressing foreign genes. The systematic assembly approach was used to develop an infectious clone of the newly identified human coronavirus, the serve acute respiratory syndrome virus (SARS-CoV). Our cloning and assembly strategy generated an infectious clone within 2 months of identification of the causative agent of SARS, providing a critical tool to study coronavirus pathogenesis and replication. The availability of coronavirus infectious cDNAs heralds a new era in coronavirus genetics and genomic applications, especially within the replicase proteins whose functions in replication and pathogenesis are virtually unknown.
JANUARY 19, 2006 "In further embodiments, the present invention provides a helper cell for producing an infectious, multiplication-defective, coronavirus particle, comprising: (a) a SARS coronavirus replicon RNA comprising a coronavirus packaging signal and a heterologous RNA sequence, wherein said replicon RNA lacks a sequence encoding at least one coronavirus structural protein; and/or (b) at least one separate helper RNA encoding the at least one coronavirus structural protein absent from the replicon RNA, said helper RNA lacking a coronavirus packaging signal; wherein the combined expression of the replicon RNA and the helper RNA produces an infectious, multiplication-defective coronavirus particle. Thus, the present invention includes the embodiment of a helper cell comprising a helper RNA encoding at least one coronavirus structural protein and the embodiment of a helper cell comprising a SARS coronavirus replicon RNA comprising a coronavirus packaging signal and a heterologous RNA sequence, wherein said replicon RNA lacks a sequence encoding at least one coronavirus structural protein.
The present invention additionally provides a method of making infectious, multiplication-defective, coronavirus particles, comprising: a) providing the helper cell of this invention; and b) producing coronavirus particles in the helper cell....Multiplication-defective or replication-defective as used herein means that the replicon RNA contained within viral particles produced according to the present invention does not itself contain sufficient genetic information to allow for the production of new infectious viral particles. 2006 Baric was granted a patent for his invisible method of fabricating a full-length infectious clone using the seamless, no-seem method. But this time, it wasnt a clone of the mouse-hepatitis virus it was a clone of the entire deadly human SARS virus, the one that had emerged from Chinese bats, via civets, in 2002. The Baric Lab came to be known by some scientists as the Wild Wild West. Also in 2006 Baric hoping to come up with a vaccine strategy for SARS, produced noninfectious virus replicon particles (or VRPs) using the Venezuelan-equine-encephalitis virus1 which they fitted with various SARS Spike Proteins. Then, wearing Tyvek suits and two pairs of gloves each, and working in a biological safety cabinet in a BSL-3-certified laboratory, they cloned and grew recombinant versions of the original SARS virus in an incubator in a medium that held African-green-monkey cells. When they had grown enough virus, the scientists swapped out one kind of Spike Protein for a carefully chosen mutant, and they challenged their prototype vaccine with it in mice. https://nymag.com/intelligencer/article/coronavirus-lab-escape-theory.html
1. VEEV was developed as a biological weapon by the United States and Soviet Union during the Cold War. It was manufactured as an incapacitating agent, designed to cause severe symptoms that would not only weaken the opposing military, but also require valuable resources to contain and treat.
2006 Shi and Daszak collaborated with a researcher in Australia to publish Review of bats and SARS in Emerging Infectious Diseases, a peer-reviewed journal published monthly by the U.S. Centers for Disease Control and Prevention. Shi was again listed as the second author, and the work was funded by the same PRC and NIH/NSF grants referenced above.
DECEMBER 2006 Shi Zheng Li a researcher at Wuhan Institute of Virology co-authors an article with Peter Daszak describing whole-genome sequences of two novel bat coronaviruses from Chinese horseshoe bats that target the ACE2 - ANGIOTENSIN-CONVERTING ENZYME 2 receptor, just like SARS-CoV-2 does1 Abstract: Bats have been identified as a natural reservoir for an increasing number of emerging zoonotic viruses, including henipaviruses and variants of rabies viruses. Recently, we and another group independently identified several horseshoe bat species (genus Rhinolophus) as the reservoir host for a large number of viruses that have a close genetic relationship with the coronavirus associated with severe acute respiratory syndrome (SARS). Our current research focused on the identification of the reservoir species for the progenitor virus of the SARS coronaviruses responsible for outbreaks during 20022003 and 20032004. In addition to SARS-like coronaviruses, many other novel bat coronaviruses, which belong to groups 1 and 2 of the 3 existing coronavirus groups, have been detected by PCR.2 The discovery of bat SARS-like coronaviruses and the great genetic diversity of coronaviruses in bats have shed new light on the origin and transmission of SARS coronaviruses. 1. In 2000, when a different coronavirus struck, Michael Farzan and his team figured out how it was getting into human cells: targeting a specific receptor called ACE2 found on certain cells. A chimeric virus is a genetic chimerism or chimera is a single organism composed of cells with more than one distinct genotype. It is a virus that has not been found in nature.
2. Polymerase Chain Reaction (PCR) is a method to rapidly make millions to billions of copies (complete copies or partial copies) of a specific DNA sample, allowing scientists to take a very small sample of DNA and amplify it (or a part of it) to a large enough amount to study in detail.
A similar Shi Zheng Li and Daszak paper.
2007 Baric said that we had entered the Golden Age of Coronavirus Genetics. Baric is basically the inventor of GoT.
2007 "A group of SARS-like CoVs (SL-CoVs) has been identified in horseshoe bats. SL-CoVs and SARS-CoVs share identical genome organizations and high sequence identities, with the main exception of the N terminus of the Spike Protein (S), known to be responsible for receptor binding in CoVs. In this study, we investigated the receptor usage of the SL-CoV S by combining a HIV human immunodeficiency virus-based pseudovirus system with cell lines expressing the ACE2 molecules of human, civet, or horseshoe bat. In addition to full-length S of SL-CoV and SARS-CoV, a series of S chimeras was constructed by inserting different sequences of the SARS-CoV S into the SL-CoV S backbone. Several important observations were made from this study. First, the SL-CoV S was unable to use any of the three ACE2 molecules as its receptor. Second, the SARS-CoV S failed to enter cells expressing the bat ACE2. Third, the chimeric S covering the previously defined receptor-binding domain gained its ability to enter cells via human ACE2, albeit with different efficiencies for different constructs. Fourth, a minimal insert region (amino acids 310 to 518) was found to be sufficient to convert the SL-CoV S from non-ACE2 binding to human ACE2 binding, indicating that the SL-CoV S is largely compatible with SARS-CoV S PROTEIN both in structure and in function. The significance of these findings in relation to virus origin, virus recombination, and host switching is discussed." https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2258702
ANALYSIS: The three most important phrases in this abstract as they relate to SARS-CoV-2 are: "First, the SL-CoV S was unable to use any of the three ACE2 molecules as its receptor." Second "We investigated the receptor usage of the SL-CoV S by combining a human immunodeficiency virus-based pseudovirus system with cell lines expressing the ACE2 molecules of human, civet, or horseshoe bat" "Third, the chimeric S covering the previously defined receptor-binding domain gained its ability to enter cells via human ACE2."
In laymans terms: We found this SARS-like virus ("S") that couldnt infect human cells. So, we combined S with parts of HIV, which does infect human cells, to see if this new S could infect human cells. The new S ("chimeric S") can infect human cells. We find that WIV was involved in this study in the Materials and Methods section: "Materials and Methods A MAb against p24 of HIV was generated by the HIV group of the Wuhan Institute of Virology (unpublished results). Rabbit polyclonal antibodies against ACE2 of the bat R. pearsonii (RpACE2) was generated using a recombinant RpACE2 protein expressed in Escherichia coli at our laboratory at the Wuhan Institute of Virology, following standard procedures. All 10 scientists who are associated with this paper are also associated with the Wuhan Institute of Virology.
2024 Report: The June 2023 ODNI Assessment supported this conclusion and went further, stating, [s]cientists at the WIV have created chimeras, or combinations of SARS-like coronaviruses through genetic engineering, attempted to clone other unrelated viruses, and used reverse genetic cloning techniques on SARS-like coronaviruses. The June 2023 ODNI Assessment continued, [s]ome of the WIVs genetic engineering projects on coronaviruses involved techniques that could make it difficult to detect intentional changes. In 2018, a year before the outbreak, EcoHealth, in partnership with the WIV, in a
grant application to DARPA proposed to create a virus with SARS-CoV-2s defining features. In their application to DARPA, EcoHealth and its WIV partners stated their intent to create a SARS-like virus with a furin cleavage site, which is the exact same feature that made humans susceptible to COVID-19 infection.21
FEBRUARY 2008 In this study, a human immunodeficiency virus (HIV)-based pseudovirus system was employed to address these issues.1 Our results indicated that the SL-CoV S protein is unable to use ACE2 proteins of different species for cell entry and that SARS-CoV S protein also failed to bind the ACE2 molecule of the horseshoe bat, Rhinolophus pearsonii. However, when the RBD of SL-CoV S was replaced with that from the SARS-CoV S, the hybrid S PROTEIN was able to use the huACE2 [HumanACE2] for cell entry, implying that the SL-CoV S proteins are structurally and functionally very similar to the SARS-CoV S. These results suggest that although the SL-CoVs discovered in bats so far are unlikely to infect humans using ACE2 as a receptor, it remains to be seen whether they are able to use other surface molecules of certain human cell types to gain entry. It is also conceivable that these viruses may become infectious to humans if they undergo N-terminal sequence variation, for example, through recombination with other CoVs, which in turn might lead to a productive interaction with ACE2 or other surface proteins on human cells.
1. Whether it is possible to construct an ACE2-binding SL-CoV S protein by replacing the RBD with that from SARS-CoV S proteins is also unknown.
The significance of these findings is as follows. First, the failure of SARS-CoV S protein to use bat RpACE2 as a receptor suggests that despite the presence of a diverse group of SL-CoVs in horseshoe bats, they are unlikely to be the natural reservoir of the immediate progenitor virus for SARS-CoV. It is therefore important to continue the search for the reservoir of SARS-CoV in other bat and wildlife species. It is also important to conduct such searches in different geographical locations because live-animal markets in southern China source their animals from all over China and from foreign countries and it is possible that the natural host of SARS-CoV was not indigenous to the site of the first SARS outbreak. Second, as predicted, sequence variation in the N-terminal region of the SL-CoV S protein rendered it incapable of using ACE2 as a receptor for cell entry. However, the ACE2-binding activity of SARS-LIKE-CoVs was easily acquired by the replacement of a relatively small sequence segment of the S protein from the SARS-CoV S sequence, highlighting the potential dangers posed by this diverse group of viruses in bats. It is now well documented that bat species, including horseshoe bats, can be infected by different CoVs. Coinfection by different CoVs in an individual bat has also been observed. Knowing the capability of different CoVs to recombine both in the laboratory and in nature, the possibility that SL-CoVs may gain the ability to infect human cells by acquiring S sequences competent for binding to ACE2 or other surface proteins of human cells can be readily envisaged. This could occur if the same bat cells carry receptors for both types of viruses. In this context, it could be concluded that the particular horseshoe bat species investigated in the current study is unlikely to be the putative mixing host. However, one cannot rule out the possibility that a different horseshoe bat or a different bat species might have a functional ACE2, thus giving it the ability to act as a mixing host. This work was jointly funded by a State Key Program for Basic Research Grant (2005CB523004) from the Chinese Ministry of Science and Technology, a special fund from the president of the Chinese Academy of Sciences (no. 1009), the Knowledge Innovation Program Key Project of the Chinese Academy of Sciences. SEPTEMBER 2008 Using a range of in vitro techniques (including infection in bat cell culture), examine the pathogenesis of these new viruses, and a pool of available bat viruses which have not yet emerged in humans. This multidisciplinary approach represents the first, concerted effort to understand the depth and breadth of the process of emergence within a key group of wildlife hosts associated with the recent emergence of SARS, Nipah, Hendra, Ebola and Marburg viruses. https://grantome.com/grant/NIH/R01-AI079231-02
2009 The WIV began working with PREDICT, a new program established at USAID to train and fund scientists to test high-risk areas for new viruses. By identifying unknown viruses before they spilled over into humansto find them before they find us, as Shi Zhengli put itresearchers hoped to establish an early-warning system. PREDICT worked in dozens of countries, but the WIV was one of its linchpins, and Shi Zhengli became famous as Chinas Bat Woman.
2011 Ron Fouchier created an airborne Avian Flu virus that could infect humans. Baric "They created a virus like H5N1 which has a 50 percent mortality rate and they trained it how to be efficiently transmitted in mammals. And so that's scary." The virus, H5N1, causes bird flu, which rarely infects people but has an extraordinarily high death rate when it does. Since the virus was first detected in 1997, about 600 people have contracted it, and more than half have died. Nearly all have caught it from birds, and most cases have been in Asia. Scientists have watched the virus, worrying that if it developed the ability to spread easily from person to person, it could create one of the deadliest pandemics ever. A government advisory panel, the National Science Advisory Board for Biosecurity, overseen by the National Institutes of Health, has asked two journals, Science and Nature, to keep certain details out of reports that they intend to publish on the research. The panel said conclusions should be published, but not experimental details and mutation data that would enable replication of the experiments. Dr. Anthony Fauci, head of the National Institute of Allergy and Infectious Diseases, said the research addressed important public health questions, but added, Im sure there will be some people who say these experiments never should have been done. Dr. Fauci said staff members at the institutes followed the results of the research and flagged it as something that the biosecurity panel should evaluate.
The potential for the modified H5N1 strain to cause a human pandemic if it ever slipped out of containment sparked sharp criticism. Rutgers molecular biologist Richard Ebright told Science Magazine that the research "should never have been done." "When I first heard about the experiments that make highly pathogenic avian influenza transmissible," says Philip Dormitzer, vice president and chief scientific officer of viral vaccines at Pfizer, "I was interested in the science but concerned about the risks of both the viruses themselves and of the consequences of the reaction to the experiments." 2011 or 2012 M.I.T. REPORTED: By taking thousands of samples from guano, fecal swabs, and bat tissue, and searching those samples for genetic sequences similar to SARS, Shis team began to discover many closely related viruses. In a cave in Yunnan Province in 2011 or 2012, they discovered the two closest, which they named WIV1 and SHC014. Shi managed to culture WIV1 in her lab from a fecal sample and show that it could directly infect human cells, proving that SARS-like viruses ready to leap straight from bats to humans already lurked in the natural world. This showed, Daszak and Shi argued, that bat coronaviruses were a substantial global threat. Scientists, they said, needed to find them, and study them, before they found us. Many of the other viruses couldnt be grown, but Barics system provided a way to rapidly test their spikes by engineering them into similar viruses. When the chimera he made using SHC014 proved able to infect human cells in a dish, Daszak told the press that these revelations should move this virus from a candidate emerging pathogen to a clear and present danger. To others, it was the perfect example of the unnecessary dangers of gain-of-function science. The only impact of this work is the creation, in a lab, of a new, non-natural risk, the Rutgers microbiologist Richard Ebright, a longtime critic of such research, told Nature. To Baric, the situation was more nuanced. Although his creation might be more dangerous than the original mouse-adapted virus hed used as a backbone, it was still wimpy compared with SARScertainly not the supervirus Senator Rand Paul would later suggest. In the end, the NIH clampdown never had teeth. It included a clause granting exceptions if head of funding agency determines research is urgently necessary to protect public health or national security. Not only were Barics studies allowed to move forward, but so were all studies that applied for exemptions. The funding restrictions were lifted in 2017 and replaced with a more lenient system.
2011 "The World Health Organization has expressed concern that US attempts to block publication of aspects of recent research on the H5N1 avian influenza virus may undermine an international agreement on sharing information on the virus. The research relates to genetic mutations that might increase the viruss risk of transmission to humans. W.H.O. raised its concerns in a statement released on December 30, 2011 and urged that information gathered through such research be shared in the manner delineated in its pandemic influenza preparedness (PIP) framework which came into effect in May 2011." December 30, 2011 "The World Health Organization (WHO) takes note that studies undertaken by several institutions on whether changes in the H5N1 influenza virus can make it more transmissible between humans have raised concern about the possible risks and misuses associated with this research. W.H.O. is also deeply concerned about the potential negative consequences. However, W.H.O. also notes that studies conducted under appropriate conditions must continue to take place so that critical scientific knowledge needed to reduce the risks posed by the H5N1 virus continues to increase." JUNE 21, 2012 The paper, by scientists at Erasmus Medical Center in the Netherlands, identified five mutations apparently necessary to make the bird flu virus spread easily among ferrets, which catch the same flus that humans do. There is always a risk, Dr. Anthony S. Fauci, the director of the National Institute for Allergy and Infectious Diseases, said in a telephone news conference held by OCTOBER 2012 This award supports a workshop that will explore the potential for in the area of ecology and evolution of infectious diseases. The workshop be held in Kunming China in October 2012. Investigators from the United States and China will discuss research priorities and ways to collaborate and explore the potential for joint US and China research in the area of ecology and evolution of infectious disease. https://grantome.com/grant/NSF/DEB-1257513
2013 The WIV discovered SARS-CoV, the cause of SARS, in a cave in Yunnan Province. SHI ZHENGLI team found that the bats of southern China were full of viruses, especially coronaviruses. Over 10 years, her team collected more than 10,000 samples from bats in the region and discovered hundreds of new coronaviruses, including some with the ability to infect humans. Many bats harbored multiple viruses, and there were alarming signs that the viruses were recombining with each otherswapping chunks of genetic code as they replicated, producing novel viruses with new abilities.
OCTOBER 30, 2013 Recently, the team led by Dr. Zhengli Shi from Wuhan Institute of Virology, Chinese Academy of Sciences, and Dr. Peter Daszak from Ecohealth Alliance identified SL-CoVs in Chinese horseshoe bats that were 95% identical to human SARS-CoV and were able to use human Angiotensin-Converting Enzyme 2 (ACE2) receptor for docking and entry. Remarkably, they isolated the first known live bat SL-CoV that replicates in human and related cells. Their findings provide clear evidence that some SL-CoVs circulating in bats are capable of infecting and replicating in human cells 2014 EcoHealth Alliance received a $3.4 million grant from the National Institute of Allergy and Infectious Diseases in 2014, which the nonprofit used, working with its Wuhan institute collaborators, to study coronaviruses in bats roosting in caves in China and how the viruses infect people. https://www.wsj.com/articles/nih-presses-u-s-nonprofit-for-information-on-wuhan-virology-lab-11597829400
Brand-new pathogens. In 2014, the NIH awarded a five-year, $3.75 million grant to EcoHealth Alliance to study the risk that more bat-borne coronaviruses would emerge in China, using the same kind of Gain-of-Threat techniques Baric had pioneered. Some of that work was to be subcontracted to the Wuhan Institute of Virology. (MIT Tech Review)
JULY 11, 2014 "After potentially serious back-to-back laboratory accidents, federal health officials announced that they had temporarily closed the flu and anthrax laboratories at the Centers for Disease Control and Prevention in Atlanta and halted shipments of all infectious agents from the agencys highest-security labs. In one episode, at least 62 C.D.C. employees may have been exposed to live anthrax bacteria after potentially infectious samples were sent to laboratories unequipped to handle them. Employees not wearing protective gear worked with bacteria that were supposed to have been killed but may not have been. All were offered a vaccine and antibiotics, and the agency said it believed no one was in danger. In a second accident, a C.D.C. lab accidentally contaminated a relatively benign flu sample with a dangerous H5N1 bird flu strain that has killed 386 people since 2003. Fortunately, a United States Agriculture Department laboratory realized that the strain was more dangerous than expected and alerted the C.D.C. Two of six vials of smallpox recently found stored in a National Institutes of Health laboratory since 1954 contained live virus capable of infecting people.
"As this report was being finalized, CDC leadership was made aware that earlier this year a culture of lowpathogenic avian influenza was unintentionally cross-contaminated at a CDC influenza laboratory with a highly pathogenic H5N1 strain of influenza and shipped to a BSL-3, select-agent laboratory operated by the United States Department of Agriculture (USDA). The CDC influenza laboratory where this incident occurred is now closed and will not reopen until adequate improvements are put in place. Although CDC is continuing to investigate and review this matter, Attachment A provides current information on the incident and the agency's response."1 1. COMMENT: Most of these accidents involved the shipment of live anthrax bacteria initially believed to be inactivated exposed lab technicians to contamination.
In OCTOBER 2014 in response to researchers' fears and some lab incidents, the federal government imposed a moratorium on all Gain-of-Function / Gain-of-Threat research, freezing the work. OCTOBER 17, 2014 "Summary: The White House Office of Science and Technology Policy and Department of Health and Human Services today announced that the U.S. Government is launching a deliberative process to assess the potential risks and benefits associated with a subset of life sciences research known as gain-of-function studies." OCTOBER 2014 Gain-of-Function / Gain-of-Threat studies, or research that improves the ability of a pathogen to cause disease, help define the fundamental nature of human-pathogen interactions, thereby enabling assessment of the pandemic potential of emerging infectious agents, informing public health and preparedness efforts, and furthering medical countermeasure development. Gain-of-Function / Gain-of-Threat studies may entail biosafety and biosecurity risks; therefore, the risks and benefits of Gain-of-Function / Gain-of-Threat research must be evaluated, both in the context of recent U.S. biosafety incidents and to keep pace with new technological developments, in order to determine which types of studies should go forward and under what conditions. In light of recent concerns regarding biosafety and biosecurity, effective immediately, the U.S.Government (USG) will pause new USG funding for gain-of-function / gain-of-threat research on influenza, MERS or SARS viruses, as defined below. This research funding pause will be effective until a robust and broad deliberative process is completed that results in the adoption of a new USG Gain-of-Function research policy. Restrictions on new funding will apply as follows: New USG funding will not be released for gain-of-function research projects that may be reasonably anticipated to confer attributes to influenza, MERS, or SARS viruses such that the virus would have enhanced pathogenicity and/or transmissibility in mammals via the respiratory route. The research funding pause would not apply to characterization or testing of naturally occurring influenza, MERS, and SARS viruses, unless the tests are reasonably anticipated to increase transmissibility and/or pathogenicity." http://www.phe.gov/s3/dualuse/Documents/gain-of-function.pdf 2014-2019 Daily Mail:"The US National Institutes of Health (NIH) funded bat-coronavirus research in the Wuhan Institute of Virology in China to the tune of US$3.7 million. Back in October 2014, the US government had placed a federal moratorium on gain-of-function (GoF) research altering natural pathogens to make them more deadly and infectious as a result of rising fears about a possible pandemic caused by an accidental or deliberate release of these genetically engineered monster germs.1 A portion of $3.7 million in grants awarded between 2014 and 2019 by the National Institutes of Health (NIH) funded research at the Wuhan Institute of Virology in China. However, not all of that $3.7 million went to the WIV, and not all of the funding took place under the Obama administration. Approximately $700,000 of the $3.7 million total was approved under Donald Trump." 1. The funding was laundered though Daszak's ECOHEALTH ALLIANCE.
JUNE 1, 2014 "Develop predictive models of bat CoV emergence risk and host range. A combined modeling approach will include phylogenetic analyses of host receptors and novel CoV genes (including functional receptor binding domains); a fused ecological and evolutionary model to predict host-range and viral sharing; and mathematical matrix models to examine evolutionary and transmission dynamics. Test predictions of CoV inter-species transmission. Predictive models of host range (i.e. emergence potential) will be tested experimentally using reverse genetics, pseudovirus and receptor binding assays, and virus infection experiments across a range of cell cultures from different species and humanized mice." https://grantome.com/grant/NIH/R01-AI110964-04 1. This is the money sent to the WIV
JUNE 6, 2014 "We will use Spike Protein sequence data,1 infectious clone technology,2 in vitro and in vivo infection experiments and analysis of receptor binding to test the hypothesis that % divergence thresholds in Spike Protein sequences predict spillover potential. We will combine these data with bat host distribution, viral diversity and phylogeny, human survey of risk behaviors and illness, and serology to identify SARSr-CoV spillover risk hotspots across southern China. Together these data and analyses will be critical for the future development of public health interventions and enhanced surveillance to prevent the re-emergence of SARS or the emergence of a novel SARSr-CoV. Project Start 2014-06-01 / Project End 2020-04-24 / Budget Start 2019-07-24 / Budget End 2020-04-24 / Support Year 6 / Fiscal Year 2019 / Total Cost / Indirect Cost" https://grantome.com/grant/NIH/R01-AI110964-06
1. The features that make SARS-CoV-2 more deadly include insertions of specific stretches of amino acids into two virus proteins, the nucleocapsid and the spike...In particular, the insertions in the spike protein are predicted, from protein structure analysis, to facilitate the recognition of the coronavirus receptors on human cells and the subsequent penetration of the virus into those cells. Finding these features in animal coronavirus isolates could predict the jump to humans and the severity of disease caused by such isolates. JUNE 6, 2014 1R01 A1110964 Daszak(PI) 06/01/14-05/31/19 Understanding the Risk of Bat Coronavirus Emergence The goal of this work is to conduct ecological and virological studies on bats in China that harbor SARS-like coronaviruses, and conduct behavioral risk surveys and testing in people, with a goal of identifying risk factors
for further spillover of SARS-like Co Vs, and help identify the likely drivers of the SARS-CoV outbreak in 2003. INTERCEPT Page 26
MARCH 20, 2015 "In one aspect, the present invention provides a chimeric coronavirus spike protein comprising, in orientation from amino to carboxy terminus: a) a first region comprising a portion of a coronavirus Spike Protein ectodomain that precedes the receptor binding domain (RBD) as located in a nonchimeric coronavirus spike protein, of a first coronavirus; b) a second region comprising a coronavirus Spike Protein receptor binding domain (RBD) of a second coronavirus that is different from said first coronavirus; c) a third region comprising a portion of a coronavirus Spike Protein S1 domain as located in a nonchimeric coronavirus Spike Protein immediately downstream of the RBD, contiguous with a portion comprising a coronavirus spike protein S2 domain as located immediately upstream of a fusion protein domain in a nonchimeric coronavirus Spike Protein, wherein said third region is of said first coronavirus; and d) a fourth region comprising a portion of a coronavirus Spike Protein from the start of the fusion protein domain through the carboxy terminal end as located in a nonchimeric coronavirus spike protein of a third coronavirus that is different from said first coronavirus and said second coronavirus.https://patents.justia.com/patent/9884895
The Spike Protein (S) of SARS-CoV-2, which plays a key role in the receptor recognition and cell membrane fusion process, is composed of two subunits, S1 and S2. The S1 subunit contains a receptor-binding domain that recognizes and binds to the host receptor Angiotensin-Converting Enzyme 2, ACE2 while the S2 subunit mediates viral cell membrane fusion by forming a six-helical bundle via the two-heptad repeat domain. In this review, we highlight recent research advance in the structure, function and development of antivirus drugs targeting the (S).
A large number of glycosylated Spike Proteins cover the surface of SARS-CoV-2 and bind to the host cell receptor Angiotensin-Converting Enzyme 2 (ACE2), mediating viral cell entry. When the Spike Protein binds to the receptor, TM protease serine 2 (TMPRSS2), a type 2 TM serine protease located on the host cell membrane, promotes virus entry into the cell by activating the Spike Protein. Once the virus enters the cell, the viral RNA is released, polyproteins are translated from the RNA genome, and replication and transcription of the viral RNA genome occur via protein cleavage and assembly of the replicasetranscriptase complex. Viral RNA is replicated, and structural proteins are synthesized, assembled, and packaged in the host cell, after which viral particles are released."
It is obvious that Daszak and his associates were experimenting with S1 and S2 SARS proteins and its coupling with the ACE2 receptors making them fit like a plug in an AC outlet and increasing their infectivity.
OCTOBER 2015 Zoonotic virus spillover from wildlife was most frequent in and around human dwellings and in agricultural fields, as well as at interfaces with occupational exposure to animals (hunters, laboratory workers, veterinarians, researchers, wildlife management, zoo and sanctuary staff). Primate hosts were most frequently cited as the source of viruses transmitted by direct contact during hunting and in laboratories, while rodent hosts were more likely to be implicated in transmission by indirect contact in and around human dwellings and in agricultural fields. https://www.nature.com/articles/srep14830
NOVEMBER 9, 2015 Two Wuhan Institute of Virology scientists, Xing-Yi Ge and Shi Zhengli, use reverse genetics to generate a chimeric virus (one that has been created by combining cells of more than one distinct genotype) closely resembling the novel coronavirus Covid-19: On the basis of these findings, we synthetically re-derived an infectious full-length SHC014 recombinant virus and demonstrate robust viral replication both in vitro and in vivo. The Nature Medicine article also mentions, Human lungs for HAE cultures were procured under University of North Carolina at Chapel Hill Institutional Review Boardapproved protocols. Shi joined with an American researcher, Ralph Baric of University of North Carolina, to generate an artificial coronavirus (a chimera), SHC014-MA15, by recombining a SARS coronavirus extracted from an infected mouse as backbone and a section of genome representing the Spike Protein of the SARS-like coronavirus, SHC014, which Shi's colleague found in 2012. This chimera were tested in vitro (test tube) and in vivo (live animals) and showed that it was capable of viral replication and could cause "robust refection to human airway culture and mice, particularly aggressive to aged mice."Hong Kong Chinese News JUNE 2021: "Those 50 and older continue to make up the bulk of Covid-19 deaths. Among that cohort, white Americans are driving the shifts in death patterns. At the height of the pandemic, those who were white and aged 75 and older accounted for more than half of all Covid-19 deaths. Now, they make up less than a third." To examine the emergence potential (that is, the potential to infect humans) of circulating bat CoVs, we built a chimeric virus encoding a novel, zoonotic CoV spike proteinfrom the RsSHC014-CoV sequence that was isolated from Chinese horseshoe bats1in the context of the SARS-CoV mouse-adapted backbone. The hybrid virus allowed us to evaluate the ability of the novel spike protein to cause disease independently of other necessary adaptive mutations in its natural backbone. Using this approach, we characterized CoV infection mediated by the SHC014 spike protein in primary human airway cells and in vivo, and tested the efficacy of available immune therapeutics against SHC014-CoV. Together, the strategy translates metagenomics data to help predict and prepare for future emergent viruses.https://www.nature.com/articles/nm.3985 November 12, 2015 In October 2014, the US government imposed a moratorium on federal funding of such research on the viruses that cause SARS, influenza and MERS (Middle East respiratory syndrome, a deadly disease caused by a virus that sporadically jumps from camels to people). The latest study was already under way before the US moratorium began, and the US National Institutes of Health (NIH) allowed it to proceed while it was under review by the agency, says Ralph Baric, an infectious-disease researcher at the University of North Carolina at Chapel Hill, a co-author of the study. The NIH eventually concluded that the work was not so risky as to fall under the moratorium, he says. But Wain-Hobson disapproves of the study because, he says, it provides little benefit, and reveals little about the risk that the wild SHC014 virus in bats poses to humans." https://www.nature.com/news/engineered-bat-virus-stirs-debate-over-risky-research-1.18787
DECEMBER 2015 The emergence of Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) and Middle East Respiratory Syndrome (MERS)-CoV underscores the threat of cross-species transmission events leading to outbreaks in humans. In this study, we examine the disease potential for SARS like CoVs currently circulating in Chinese horseshoe bat populations. Utilizing the SARS-CoV infectious clone, we generated and characterized a chimeric virus expressing the spike of bat coronavirus SHC014 in a mouse adapted SARS-CoV backbone. The results indicate that group 2b viruses encoding the SHC014 spike in a wild type backbone can efficiently utilize multiple ACE2 receptor orthologs, replicate efficiently in primary human airway cells, and achieve in vitro titers equivalent to epidemic strains of SARS-CoV. Additionally, in vivo experiments demonstrate replication of the chimeric virus in mouse lung with notable pathogenesis. Evaluation of available SARS-based immune-therapeutic and prophylactic modalities revealed poor efficacy; both monoclonal antibody and vaccine approaches failed to neutralize and protect from CoVs utilizing the novel spike protein. Importantly, based on these findings, we synthetically rederived an infectious full length SHC014 recombinant virus and demonstrate robust viral replication both in vitro and in vivo. Together, the work highlights a continued risk of SARS-CoV reemergence from viruses currently circulating in bat populations SARS-LIKE-CLUSTER-OF-CIRCULATING-BAT-CORONAVIRUSES-SHOWS-POTENTIAL-FOR-HUMAN-EMERGENCE
DECEMBER 29, 2015 Yang XL1, Hu B1, Wang B1, Wang MN1, Zhang Q1, Zhang W1, Wu LJ1, Ge XY1, Zhang YZ2, PETER DASZAK P3, Wang LF4, SHI ZHENGLI ZL5 Author information [All worked at Wuhan Institute of Virology] Journal of Virology, Dec 29, 2015, 90(6):3253-3256 DOI: 10.1128/JVI.02582-15 PMID: 26719272 PMCID: PMC4810638 NIAID NIH HHS (2)? Grant ID: R01 / AI110964 19 publications Grant ID: R01AI110964 3 publications Google search of Grant ID: R01AI110964 takes us to NIH: National Institute of Allergy and Infectious Diseases (Dr. FAUCI) "We report the isolation and characterization of a novel bat coronavirus which is much closer to the severe acute respiratory syndrome coronavirus (SARS-CoV) in genomic sequence than others previously reported, particularly in its S gene. Cell entry and susceptibility studies indicated that this virus can use ACE2 as a receptor and infect animal and human cell lines. Our results provide further evidence of the bat origin of the SARS-CoV and highlight the likelihood of future bat coronavirus emergence in humans." We recently isolated a bat SL-CoV-1 strain (WIV1) and constructed an infectious clone of another strain (SHC014); significantly, these strains are closely related to SARS-CoV and capable of using the same cellular receptor (angiotensin-converting enzyme 2 [ACE2] as SARS-CoV-1. Despite the high similarity in genomic sequences and receptor usage of these two strains, there is still some difference between the N-terminal domains of the S proteins of SARS-CoV-1 and other SL-CoVs, indicating that other unknown SL-CoV-1s are circulating in bats." https://europepmc.org/article/pmc/pmc4810638
"In 2016 MIT: Daszak and Shi published a paper reporting how the Chinese lab had engineered different versions of WIV1 and tested their infectiousness in human cells. The paper announced that the WIV had developed its own reverse-genetics system, following the Americans lead. It also included a troubling detail: the work, which was funded in part by the NIH grant, had been done in a BSL-2 lab. That meant the same viruses that Daszak was holding up as a clear and present danger to the world were being studied under conditions that, according to Richard Ebright, matched the biosafety level of a US dentists office. Ebright believes one factor at play was the cost and inconvenience of working in high-containment conditions. The Chinese labs decision to work at BSL-2, he says, would have effectively increas[ed] rates of progress, all else being equal, by a factor of 10 to 20a huge edge. Work at the WIV was indeed progressing quickly. In 2017, Daszak and Shi followed with another study, also at BSL-2, that one-upped Barics work in North Carolina. The WIV had continued to unearth dozens of new SARS-like coronaviruses in bat caves, and it reported making chimeras with eight of them by fusing the spikes of the new viruses to the chassis of WIV1. Two of them replicated well in human cells. They were, for all intents and purposes, brand-new pathogens. The revelation that the WIV was working with SARS-like viruses in subpar safety conditions has led some people to reassess the chance that SARS-CoV-2 could have emerged from some type of laboratory incident. Thats screwed up, the Columbia University virologist Ian Lipkin, who coauthored the seminal paper arguing that covid must have had a natural origin, told the journalist Donald McNeil Jr. It shouldnt have happened. People should not be looking at bat viruses in BSL-2 labs. My view has changed.
But the WIV was not breaking any rules by working at BSL-2, says Filippa Lentzos, a biosecurity expert at Kings College London There are no enforceable standards of what you should and shouldnt do. Its up to the individual countries, institutions, and scientists. And in China, she says, the vertiginous rise of high-tech biological research has not been accompanied by an equivalent increase in oversight. In an email, Zhengli Shi said she followed Chinese rules that are similar to those in the US. Safety requirements are based on what virus you are studying. Since bat viruses like WIV1 havent been confirmed to cause disease in human beings, her biosafety committee recommended BSL-2 for engineering them and testing them and BSL-3 for any animal experiments. In response to questions about the decision to do the research in BSL-2 conditions, Peter Daszak forwarded a statement from EcoHealth Alliance stating that the organization must follow the local laws of the countries in which we work and that the NIH had determined the research was not gain-of-function. EARLY 2016 Shi sent Baric a fresh bat virus Spike Protein, and Baric inserted it into the backbone of a human SARS virus and then used that infectious clone to attack human airway cells.
MARCH 14, 2016 The emergence of Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) and Middle East respiratory syndrome (MERS)-CoV highlights the continued risk of cross-species transmission leading to epidemic disease. This manuscript describes efforts to extend surveillance beyond sequence analysis, constructing chimeric and full-length zoonotic coronaviruses to evaluate emergence potential. Focusing on SARS-like virus sequences isolated from Chinese horseshoe bats, the results indicate a significant threat posed by WIV1-CoV. Both full-length and chimeric WIV1-CoV readily replicated efficiently in human airway cultures and in vivo, suggesting capability of direct transmission to humans. In addition, while monoclonal antibody treatments prove effective, the SARS-based vaccine approach failed to confer protection. Together, the study indicates an ongoing threat posed by WIV1-related viruses and the need for continued study and surveillance. "The virus readily and efficiently replicated in cultured human airway tissues, suggesting an ability to potentially jump directly to humans, reported the University of North Carolina [Baric] website. This time, they also used the bat-human hybrid virus to infect transgenic humanized mice that grew human ACE2 protein. The mice, young and old, lost weight and died, proving, again, that this particular bat virus was potentially poised to emerge in human populations. It was an ongoing threat, Baric wrote. https://www.pnas.org/content/113/11/3048
MAY 2016 This report further defines gain-of function research of concern to describe the subset of studies that have been the subject of recent debate and have raised potential biosafety and biosecurity implications. These are gain-of function studies with the potential to generate pathogens with pandemic potential in humans by exhibiting high transmissibility and high virulence. The RBA (RISK AND BENEFIT ASSESSMENTS) contains a number of other useful analyses as well, including background and contextual information on the biology of influenza and coronavirus, historical analysis of naturally-occurring seasonal and pandemic influenza and coronavirus outbreaks, an examination of the potential proliferation of GOF research, and analysis of the potential loss of public trust in science that could result if a laboratory incident involving GOF research were to occur. The biosecurity risk assessment shows that the most probable threats involve insiders who have direct access to dangerous pathogens or outsiders [Daszak] who collaborate with or subvert insiders.
In addition to the agents and toxins on the list, the select agent regulations apply to some genetic elements, including nucleic acids that are immediate precursors to infectious forms of any select agent viruses (i.e., complete positive strand RNA viral genomes), as well as some nucleic acids that encode select toxins. [Baric's experiments bringing extinct viruses back to life.] Select agent regulations also apply to genetically-modified select agents and toxins. Restricted experiments are described in the regulations and involve the deliberate transfer of or selection for a drug resistance trait to select agents that are not known to acquire the trait naturally.
As with all life sciences research involving pathogens, GOF studies entail inherent biosafety and biosecurity risks. GOF research involving the generation of pathogens with pandemic potential involves the greatest risks. A laboratory accident involving such a pathogen could potentially release a pathogen that could spread rapidly and efficiently through the human population.
Manipulations that increase transmissibility, increase pathogenicity, and enable a pathogen to more readily spread through the population have the greatest potential to increase risk; in some strains even a moderate increase might be a concern...GOFROC entails the generation of pathogensperhaps novel pathogenswith anticipated pandemic potential. The risks associated with such studies are uncertain but potentially significant. It is possible that generating a laboratory pathogen with pandemic potential introduces a risk of a pandemic, albeit a low probability risk, that did not exist before that pathogen was generated...The pathogen generated is likely highly transmissible and likely capable of wide and uncontrollable spread in human populations. To be considered highly transmissible the pathogen must be judged to have the capacity for sustained secondary transmission among humans, particularly, but not exclusively, by the respiratory route.
Importantly, a proposed experiment need not involve the simultaneous enhancement of both phenotypes. Thus, research involving a naturally-occurring pathogen that exhibits one of the above attributes would be considered GOFROC if a study were anticipated to confer the second attribute to the agent (while retaining the first attribute). Other studies may generate a pathogen with the above attributes after a series of manipulations that enhance the phenotypes separately but ultimately result in a pathogen with both attributes. Any route of experimentation that is anticipated to ultimately generate a pathogen that exhibits both of the characteristics above would be considered GOFROC [Gain-of-Function Research of Concern] and should be reviewed carefully before it can be funded. Pathogen characteristics recommended for consideration in the RA (RISK ASSESSMENT) and BA (BENEFIT ASSESSMENT) The RA and BA should include analysis of the risks and benefits associated with GoF experiments that
are anticipated to increase the pandemic potential of pathogens. Toward this end, the following characteristics, which may be conferred to pathogens during the conduct of GoF studies, should be
considered:
Scientists who were far more educated and experienced than the mere Zoologist Daszak knew the potential was there to cause a pandemic. But Daszak had an agenda so he made sure they continued.
NOVEMBER 2016 ABSTRACT: Bats harbor severe acute respiratory syndrome (SARS)-like coronaviruses (SL-CoVs) from which the causative agent of the 2002-2003 SARS pandemic is thought to have originated. However, despite the fact that a large number of genetically diverse SL-CoV sequences have been detected in bats, only two strains (named WIV1 and WIV16) have been successfully cultured in vitro. These two strains differ from SARS-CoV only in containing an extra Open Reading Frame (ORF) (named ORFX), between ORF6 and ORF7, which has no homology to any known protein sequences. In this study, we constructed a full-length cDNA clone of SL-CoV WIV1 (rWIV1), an ORFX deletion mutant (rWIV1-X), and a Green Fluorescent Protein (GFP)1-expressing mutant (rWIV1-GFP-X). Northern blotting and fluorescence microscopy indicate that ORFX was expressed during WIV1 infection. A virus infection assay showed that rWIV1-X replicated as efficiently as rWIV1 in Vero E6, Calu-3, and HeLa-hACE2 cells. Further study showed that ORFX could inhibit interferon production and activate NF-B. Our results demonstrate for the first time that the unique ORFX in the WIV1 strain is a functional gene involving modulation of the host immune response but is not essential for in vitro viral replication. Lei-Ping Zeng,a Yu-Tao Gao, Xing-Yi Ge, Qian Zhang, a Cheng Peng, Xing-Lou Yang, a Bing Tan, Jing Chen, Aleksei A. Chmura, Peter Daszak, Zheng-Li Shia Key Laboratory of Special Pathogens, Wuhan Institute of Virology, Chinese Academy of Sciences, Wuhan, China; EcoHealth Alliance, New York, New York, USA https://www.ecohealthalliance.org/wp-content/uploads/2016/11/Zeng-etal_Bat-SARSlike_2016.pdf
1. Green fluorescent protein (GFP) is a protein in the jellyfish Aequorea Victoria that exhibits green fluorescence when exposed to light. The protein has 238 amino acids, three of them (Numbers 65 to 67) form a structure that emits visible green fluorescent light. ... Biologists use GFP as a marker protein.
2017 Abstract "Emergent and preemergent coronaviruses (CoVs) pose a global threat that requires immediate intervention. Rapid intervention necessitates the capacity to generate, grow, and genetically manipulate infectious CoVs in order to rapidly evaluate pathogenic mechanisms, host and tissue permissibility, and candidate antiviral therapeutic efficacy. CoVs encode the largest viral RNA genomes at about 28-32,000 nucleotides in length, and thereby complicate efficient engineering of the genome. Deconstructing the genome into manageable fragments affords the plasticity necessary to rapidly introduce targeted genetic changes in parallel and assort mutated fragments while maximizing genome stability over time. In this protocol we describe a well-developed reverse genetic platform strategy for CoVs that is comprised of partitioning the viral genome into 5-7 independent DNA fragments (depending on the CoV genome), each subcloned into a plasmid for increased stability and ease of genetic manipulation and amplification. Coronavirus genomes are conveniently partitioned by introducing type IIS or IIG restriction enzyme recognition sites that confer directional cloning. Since each restriction site leaves a unique overhang between adjoining fragments, reconstruction of the full-length genome can be achieved through a standard DNA ligation comprised of equal molar ratios of each fragment. Using this method, recombinant CoVs can be rapidly generated and used to investigate host range, gene function, pathogenesis, and candidate therapeutics for emerging and preemergent CoVs both in vitro and in vivo" Work at the WIV was indeed progressing quickly. In 2017, Daszak and Shi followed with another study, also at BSL-2, that one-upped Barics work in North Carolina. The WIV had continued to unearth dozens of new SARS-like coronaviruses in bat caves, and it reported making chimeras with eight of them by fusing the spikes of the new viruses to the chassis of WIV1. Two of them replicated well in human cells. They were, for all intents and purposes, brand-new pathogens. The revelation that the WIV was working with SARS-like viruses in subpar safety conditions has led some people to reassess the chance that SARS-CoV-2 could have emerged from some type of laboratory incident. Thats screwed up, the Columbia University virologist Ian Lipkin, who coauthored the seminal paper arguing that covid must have had a natural origin, told the journalist Donald McNeil Jr. It shouldnt have happened. People should not be looking at bat viruses in BSL-2 labs. My view has changed. But the WIV was not breaking any rules by working at BSL-2, says Filippa Lentzos, a biosecurity expert at Kings College London There are no enforceable standards of what you should and shouldnt do. Its up to the individual countries, institutions, and scientists. And in China, she says, the vertiginous rise of high-tech biological research has not been accompanied by an equivalent increase in oversight. In an email, Zhengli Shi said she followed Chinese rules that are similar to those in the US. Safety requirements are based on what virus you are studying. Since bat viruses like WIV1 havent been confirmed to cause disease in human beings, her biosafety committee recommended BSL-2 for engineering them and testing them and BSL-3 for any animal experiments. In response to questions about the decision to do the research in BSL-2 conditions, Peter Daszak forwarded a statement from EcoHealth Alliance stating that the organization must follow the local laws of the countries in which we work and that the NIH had determined the research was not gain-of-function. "Since bat viruses like WIV1 havent been confirmed to cause disease in human beings, her biosafety committee recommended BSL-2 for engineering them and testing them and BSL-3 for any animal experiments." Shi authored a paper with Baric about bat viruses spilling over into animal than human populations that crowned her The Bat Lady.
JANUARY 2017 FEBRUARY 22, 2017 The Wuhan Institute of Virology, Chinas first BIOSAFETY LEVEL-4 LAB (BSL-4), is certified to work on the most dangerous pathogens. Observes Nature, But worries surround the Chinese lab, too. The SARS virus has escaped from high-level containment facilities in Beijing multiple times. The Wuhan Institute of Virology is the only BSL-4 virology lab in China staffed with two Chinese virologists, SHI ZHENGLI and Xing-Yi Ge, who both previously worked at a University of North Carolina at Chapel Hill lab, which had already bio-engineered an incredibly virulent strain of bat coronavirus1.
1. The GoF/GoT experiments were carried out in a BSL-3 and BSL-2 Lab. Other reports say the BSL-4 was postponed until 2018.
In 2017, she and her colleagues at the Wuhan lab published a paper about an experiment in which they created new hybrid bat coronaviruses by mixing and matching parts of several existing ones including at least one that was nearly transmissible to humans in order to study their ability to infect and replicate in human cells.
APRIL 2017
Peter Daszak admitting his EcoHealth Alliance groups research WAS in fact funded through Dr Anthony Faucis NIH....... pic.twitter.com/y9UBQso1CG NOVEMBER 30, 2017 Nature Medicine: After a detective hunt across China, researchers chasing the origin of the deadly SARS virus have finally found their smoking gun. In a remote cave in Yunnan province, virologists have identified a single population of horseshoe bats that harbours virus strains with all the genetic building blocks of the one that jumped to humans in 2002, killing almost 800 people around the world. And wielding the smoking gun, besides a cast of usual suspects (Xing-Yi Ge and PETER DASZAK ) was none other than SHI ZHENGLI (Zhengli Shi in Chinese), a virologist who is often called Chinas bat woman by her colleagues because of her virus-hunting expeditions in bat caves over the past 16 years. That Nature Medicine: report was based on an article published in San Francisco-based PLOS Pathogens and jointly funded by National Natural Science Foundation of China. https://www.nature.com/articles/d41586-017-07766-9
"In the current study, we successfully cultured an additional novel SARSr-CoV Rs4874 from a single fecal sample using an optimized protocol and Vero E6 cells. Its S protein shared 99.9% aa sequence identity with that of previously isolated WIV16 and it was identical to WIV16 in RBD. Using the reverse genetics technique we previously developed for WIV1, we constructed a group of infectious bacterial artificial chromosome (BAC) clones with the backbone of WIV1 and variants of S genes from 8 different bat SARSr-CoVs. Only the infectious clones for Rs4231 and Rs7327 led to cytopathic effects in Vero E6 cells after transfection. The other six strains with deletions in the RBD region, Rf4075, Rs4081, Rs4085, Rs4235, As6526 and Rp3 failed to be rescued, as no cytopathic effects was observed and viral replication cannot be detected by immunofluorescence assay in Vero E6 cells. In contrast, when Vero E6 cells were respectively infected with the two successfully rescued chimeric SARSr-CoVs, WIV1-Rs4231S and WIV1-Rs7327S, and the newly isolated Rs4874, efficient virus replication was detected in all infections. To assess whether the three novel SARSr-CoVs can use human ACE2 as a cellular entry receptor, we conducted virus infectivity studies using HeLa cells with or without the expression of human ACE2. All viruses replicated efficiently in the human ACE2-expressing cells. The results were further confirmed by quantification of viral RNA using real-time RT-PCR." NOVEMBER 30, 2017 "To assess whether the three novel SARSr-CoVs can use human ACE2 as a cellular entry receptor, we conducted virus infectivity studies using HeLa cells with or without the expression of human ACE2. All viruses replicated efficiently in the human ACE2-expressing cells." Isn't that great. They aimed the virus at human lung tissue that contains ACE2 receptors. This is Gain-of-Function. MIT biologist Kevin Esvelt reviewed a paper that appears to have been published with financial assistance from the Daszak sub-contract grant. According to Esvelt, certain techniques that the researchers used met the definition of Gain-of-Function research. DECEMBER 19, 2017 The US National Institutes of Health (NIH) announced that they would resume funding Gain-of-Function experiments involving influenza, Middle East respiratory syndrome coronavirus, and severe acute respiratory syndrome coronavirus. A moratorium had been in place since October, 2014. At the time, the NIH had stated that the moratorium will be effective until a robust and broad deliberative process is completed that results in the adoption of a new US Government Gain-of-Function research policy. This process has now concluded. It was spearheaded by the National Science Advisory Board for Biosecurity (NSABB) and led to the development of a new framework for assessing funding decisions for research involving pathogens with enhanced pandemic potential. The release of the framework by the Department of Health and Human Services (HHS), of which NIH is part, signalled the end of the funding pause." Francis S. Collins, M.D., Ph.D. Director, National Institutes of Health." It should be noted that the names of the scientists who make these determinations will be TOP SECRET MAY 17, 2017 At a hotel in central Wuhan in May 2017, a former commander at USAMRIID participated in a workshop with the WIV's top scientists. The workshop was called the Second China-US Workshop on the Challenges of Emerging Infections, Laboratory Safety and Global Health Security. Gain-of-function research, laboratory risks and gene editing were key topics during the conference held from May 17-19. Shi and George F. Gao, the director at the Chinese Centre for Disease Control and Prevention, gave presentations at the conference hosted by Chinas Academy of Sciences and attended by the director of Wuhan Institute of Virology BSL-4 laboratory, Yuan Zhiming. (What Really Happened In Wuhan by Sharri Markson 25/5/21)
JANUARY 19, 2018 U.S. Embassy in Beijing takes unusual step of repeatedly sending U.S. science diplomats to the Wuhan Institute of Virology. One visit raises so many red flags, that a cable is sent noting, a serious shortage of appropriately trained technicians and investigators needed to safely operate this high-containment laboratory. During interactions with scientists at the WIV laboratory, they noted the new lab has a serious shortage of appropriately trained technicians and investigators needed to safely operate this high-containment laboratory, the researchers also showed that various SARS-like coronaviruses can interact with ACE2, the human receptor identified for SARS-coronavirus. This finding strongly suggests that SARS-like coronaviruses from bats can be transmitted to humans to cause SARS-like diseases. From a public health perspective, this makes the continued surveillance of SARS-like coronaviruses in bats and study of the animal-human interface critical to future emerging coronavirus outbreak prediction and prevention. (JANUARY 19, 2018 , DOS cable, drafted by two officials from the embassys environment, science and health sections who met with the WIV scientists.)
JANUARY 2018 Shi was appointed Principal Investigator for a new Strategic Priority Research Program of the Chinese Academy of Sciences (grant no. XBD29010101, $1.35 million USD), investigating genetic evolution and transmission mechanism of important bat-borne viruses. This project, especially with its focus on transmission mechanisms, aligns with the first focus area
mentioned above. That same month, Shi began work on a project titled Study on the evolutionary mechanism of bat SARS-like coronavirus adapted to host receptor molecules and the risk of crossspecies infection. The project was funded at a value of roughly $850,000 USD (grant no. 31770175) and is slated to run until December 2021. This grant aligns with the second focus area, the
description of which specifically mentions replicating and modifying coronaviruses. [GOP ORIGINS]
MARCH 2, 2019 It is highly likely that future SARS- or MERS-like coronavirus outbreaks will originate from bats, and there is an increased probability that this will occur in China Shi Zhengli and her colleagues wrote in a 2019 paper that now seems chillingly prescient. Therefore, the investigation of bat coronaviruses becomes an urgent issue for the detection of early warning signs. "Why China? Two bat CoVs caused outbreaks in China; it is thus urgent to study the reasons to avoid future outbreaks. China is the third largest territory and is also the most populous nation in the world. A vast homeland plus diverse climates bring about great biodiversity including that of bats and bat-borne virusesmost of the ICTV coronavirus species were named by Chinese scientists studying local bats or other mammals. The majority of the CoVs can be found in China. Moreover, most of the bat hosts of these CoVs live near humans, potentially transmitting viruses to humans and livestock. Chinese food culture maintains that live slaughtered animals are more nutritious, and this belief may enhance viral transmission. In November 2012, the first case of SARS was recorded in Foshan city, Guangdong Province, China. In 2005, two independent Chinese groups reported the first bat SARS-related CoV (SARSr-CoV) that was closely related to human SARS-CoV, implying a bat origin of the latter. Since then, more bat SARSr-CoV isolates were identified in China. Genome identities of these bat SARSr-CoVs are as high as 92% to human SARS-CoV, but their major receptor binding spike proteins cannot use the human virus entry receptor ACE2. Whether they are the progenitor viruses of SARS-CoV is debatable. In 2013, the isolation of a bat SARSr-CoV that uses the ACE2 receptor provided the strongest evidence of the bat origin of SARS-CoV. Furthermore, the building blocks for SARS-CoV were identified from eleven different SARSr-CoV viral strains in a five-year surveillance program in a cave inhabited by multiple species of horseshoe bats in Yunnan Province, China It is generally believed that bat-borne CoVs will re-emerge to cause the next disease outbreak. In this regard, China is a likely hotspot. The challenge is to predict when and where, so that we can try our best to prevent such outbreaks. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6466186/ March 27, 2018 The Wuhan Institute of Virology issues a news release in English after the last of the U.S. Embassy visits. The news release was removed the week of April 6, 2020, although it remains archived on the internet.
APRIL 2018 Have you seen this report? What is the connection between Piglets virus and NIAID? The scientists that helped the farmers identified the virus are from Wuhan Institute of Virology, Dr. Zhengli Shi's team. Dr. Shi is the Chinese collaborator on a ROB grant award to EcoHealth in NYC. Her lab has published the connection of SARS-Iike coronavirus found in cave bats in
Southwestern China to the SARS that caused human infection in 2003. NIAID funding has supported her research on SARS-Iike coronaviruses in China. Her team has developed tests to identify SARS-Iike coronaviruses. The test was used to identify the virus that caused piglets death at the farm. I learned this story last year when she visited embassy with EcoHealth to USAID on the Global Vi rome Project.
Dr. Shi was the researcher who made the arrangement for me to visit WIV's P41ab. Attached is the research paper and NIAID grant was acknowledged. JUDICIAL WATCH DOCS
APRIL 2018 Wang Yanyi: "Some of the speculation [of a lab leak] is based on an article the WIV published in Nature in April 2018, saying it discovered a novel coronavirus originating from bats. In February 2020, WIV published another article in Nature saying another novel coronavirus from bats was discovered, and the similarity between this virus and the Sars-CoV-2 is up to 96.2 per cent. Actually, the virus mentioned in the 2018 article wasnt SARS-CoV-2. The virus in the article mainly causes diarrhoea and death among piglets. It was later named SADS. The genome sequence of SADS is only 50 per cent similar to that of SARS-CoV-2. Its a rather big difference. The bat coronavirus that had a 96.2 per cent genomic similarity to Sars-CoV-2 was RaTG-13. From the perspective of many non-professionals, the similarity rate of 96.2 per cent is a very high number. But coronavirus is one of the RNA viruses that have the largest genomes. Take the SARS-CoV-2 for example. Its entire genome contains about 30,000 bases. The difference of a percentage of 3.8 means the difference of over 1,100 nucleotide positions. In the natural world, it takes a long period of time for a virus to naturally evolve and mutate to become Sars-CoV-2. Although the researchers identified the genome sequence of RaTG-13 they did not isolate nor obtain the live virus of RaTG-13. Thus, there is no possibility of us leaking RaTG-13." Wang Yanyi said the institute now had three strains of live viruses, including one that was 96 per cent genomically similar to the SARS virus. But their highest similarity to SARS-CoV-2 was only 79.8 per cent." This was a startling admission. if they had viruses 80% similar why not SARS-COV-2?" DECEMBER 2019 I had never expected this kind of thing to happen in Wuhan, in central China,Shi Zhengli told Scientific American. "Wuhan is a skyscraper-filled metropolis of 11 million people hundreds of miles from the bat-friendly caves of southern China. Could they have come from our lab? Shi Zhengli described the next few weeks as the most stressful of her life. She claimed she frantically searched her labs records, looking for signs of an accident or inappropriate disposal, only relaxing once the genetic code of the new virus was sequenced and didnt match the coronaviruses in her lab. That really took a load off my mind. I had not slept a wink for days. The Chinese government shows no signs of releasing Shis labs records so we have to take her self-serving lies for fact. In any case, while Shis comments were meant to be reassuring, they actually implied something unsettling. Most of us mistakenly believe that the risk of a biolab-based pandemic is infinitesimal. But clearly Shi Zhengli didnt rule out an accidental escape from her lab. Although we will never get to see her records we do know that Shi Zhengli work at the time involved altering the Spike Protein of Corona viruses found in bats:
Shi Zhengli: "During the past two decades, three zoonotic coronaviruses have been identified as the cause of large-scale disease outbreaksSevere Acute Respiratory Syndrome (SARS), Middle East Respiratory Syndrome (MERS), and Swine Acute Diarrhea Syndrome (SADS). SARS and MERS emerged in 2003 and 2012, respectively, and caused a worldwide pandemic that claimed thousands of human lives, while SADS struck the swine industry in 2017. They have common characteristics, such as they are all highly pathogenic to humans or livestock, their agents originated from bats, and two of them originated in China. Thus, it is highly likely that future SARS- or MERS-like coronavirus outbreaks will originate from bats, and there is an increased probability that this will occur in China. Therefore, the investigation of bat coronaviruses becomes an urgent issue for the detection of early warning signs, which in turn minimizes the impact of such future outbreaks in China. The purpose of the review is to summarize the current knowledge on viral diversity, reservoir hosts, and the geographical distributions of bat coronaviruses in China, and eventually we aim to predict virus hotspots and their cross-species transmission potential." SEPTEMBER 12, 2019 It has not been published since and has not been made available to World Health Organisation investigators, who did not ask for it during their Wuhan visit earlier this year. Shi has claimed the virus database was taken offline after 3000 hacking attempts. Vanity Fair reported "They paid one visit to the Wuhan Institute of Virology, where they met with Shi Zhengli, as recounted in an annex to the mission report. One obvious demand would have been access to the WIVs database of some 22,000 virus samples and sequences, which had been taken offline. At an event convened by a London organization on March 10, 2021 Daszak was asked whether the group had made such a request. He said there was no need: Shi Zhengli had stated that the WIV took down the database due to hacking attempts during the pandemic. Absolutely reasonable, Daszak said. And we did not ask to see the data
. As you know, a lot of this work has been conducted with EcoHealth Alliance
. We do basically know whats in those databases. There is no evidence of viruses closer to SARS-CoV-2 than RaTG13 in those databases, simple as that. The GOP ORIGINS REPORT: KATIE BO WILLIAMS, CNN INTELLIGENCE AND NATIONAL SECURITY REPORTER: Just a few months before the world becomes aware that there is this dangerous virus circulating in Wuhan, the WIV removes from the internet this catalog of 22,000 viral samples that it had been working on, that it had been studying.
GUPTA (on-camera): What happened to the bat coronavirus database in September of 2019?
DASZAK: They told us they were revising it and making it more searchable and that they would then get hacked over and over again so they didn't put it back up. That's what they told us.
GUPTA: Do you believe that?
DASZAK: Well, it's not my role to believe or not believe. I'm a scientist. I look at data and information and we look at it objectively.
GUPTA: You're a scientist but I'm saying, you know, you do have to look at history as well and there's been concerns that there's been a lack of transparency.
DASZAK: Right. When I look in that database, it was simply a list of samples with sequences attached. Most of which had been published, probably the vast majority by now, so we knew what was in those data bases.
GUPTA: If it was mostly out there.
DASZAK: Yes.
GUPTA: Much of it published, why were they so concerned about it being hacked?
DASZAK: Well, I don't know. I mean, more importantly, people who think that that database has the answers to SARS-COVI-2, I really doubt that.
One of the more informative suppressions of data was the closure of China’s main database on bat and other viruses. Zhengli Shi, China’s leading expert on bat coronaviruses, told the BBC that it was taken off line because of numerous hacking attempts. It’s conceivable that in January 2020, after the pandemic broke out, people without authorized access might have tried to hack into the database. But in fact the database went off line on September 12, 2019. Who would have wanted to hack into a bat virus database back then? More likely, that is the date at which Chinese authorities realized they had a virus escape problem. Bulletin of Atomic Scientists
OCTOBER 2019 A report obtained by the London-based NBC News Verification Unit says there was no cellphone activity in a high-security portion of the Wuhan Institute of Virology from October 7, 2019 through October 24, 2019, and that there may have been a "hazardous event" sometime between Oct. 6 2019 and Oct. 11 2019. "Would be interesting if someone analyzed commercial telemetry data at & near Wuhan lab from Oct-Dec 2019," Marco Rubio tweeted. "If it shows dramatic drop off in activity compared to previous 18 months it would be a strong indication of an incident at lab & of when it happened." OCTOBER 14-19, 2019 CCP orders roadblocks around WIV.
OCTOBER 25, 2019 In a move that worries many public health experts, the federal government is quietly shutting down a surveillance program for dangerous animal viruses that someday may infect humans. The United Nations Environment Program estimates that a new animal disease that can also infect humans is discovered every four months. Ending the program, experts fear, will leave the world more vulnerable to lethal pathogens like Ebola and MERS that emerge from unexpected places, such as bat-filled trees, gorilla carcasses and camel barns. The end of the program is definitely a loss, said Peter Daszak, president of the EcoHealth Alliance, a nonprofit global health organization that received funding from the program. PREDICT was an approach to heading off pandemics, instead of sitting there waiting for them to emerge and then mobilizing. Thats expensive." NOVEMBER 2019 a genetically modified Coronavirus escaped from the Bio Safety Level, BSL-2 lab in Wuhan. These experiments, assuming they should be conducted at all, should have been performed in a BSL-4 lab, but the WIV didn't have one until 2018 and many of these experiments were performed much earlier and their results were stored at the WIV. BSL-4 is for viruses to which there is no known vaccine or therapeutic. Daszak should stand trial in USDC/SDNY. If found guilty he should face the death penalty for mass-murder and treason. Dust off old sparky and execute him. This is personal. I came minutes away from dying of SARS-CoV-19 but came back to life after doctors beat on my chest, then I spent a month in Lenox Hill Hospital in Manhattan with a lung tube in my chest as I had a collapsed lung. The disease left one of my fingers paralyzed. I witnessed the agony and suffering the other patients went through first hand.
NOVEMBER 2019 At least four countries who sent delegations to the MILITARY WORLD GAMES have now confirmed the presence of SARS-CoV-2 or COVID-19 cases within their borders in November and December 2019, before the news of an outbreak first became public. NOVEMBER 2019 The WSJ reported: "Three researchers from Chinas Wuhan Institute of Virology became sick enough in November 2019 that they sought hospital care, according to a previously undisclosed U.S. intelligence report." While it is not clear what had sickened them they had symptoms similar to SARS-CoV-19, These were not the janitors, said the former State Department official. They were active researchers. The dates were among the absolute most arresting part of the picture, because they are smack where they would be if this was the origin. An intelligence analyst working with David Asher sifted through classified channels and turned up a report that outlined why the lab-leak hypothesis was plausible. It had been written in May by researchers at the Lawrence Livermore National Laboratory, which performs national security research for the Department of Energy. But it appeared to have been buried within the classified collections system. The Wuhan Institute of Virology has not come across such cases, the Bat Lady wrote to the NYT, If possible, can you provide the names of the three to help us check?
NOVEMBER 18, 2019 The Wuhan Institute of Virology posts a job opening that seeks to fill 1-2 post-doctoral fellows who will take bats as the research object and provide answers regarding the molecular mechanism that can co-exist with Ebola and SARS-associated coronaviruses for a long time without causing disease, and its relationship with flight (aerosols) and longevity. Virology, immunology cell biology and multiple comics (refers to biological science study fields that end with -omics) are used to compare the differences between humans and other mammals. Candidates are to send their resumes to Peng Zhou (??) Ph.D., the Leader of the Bat Virus Infection and Immunization Group at the Wuhan Institute of Virology.
NOVEMBER 21, 2019 Daszak tweeted: "Weve made great progress with bat SARS-related CoVs, IDing 50 novel strains, sequencing Spike Protein genes, IDing ones that bind to human cells, using recombinant viruses, humanized mice to see SARS-like signs, and showing some dont respond to MAbs, vaccines."
DECEMBER 9, 2019 In a video interview Daszak mentioned the testing of modified coronaviruses on human cells and humanized mice in the WIV, just weeks before the first cases of COVID-19 were announced in the city of Wuhan, China. Daszak has dismissed the possibility of a lab leak, insisting since the start of the pandemic it's a "conspiracy theory" without providing evidence for his claim. Daszak: "Real conspiracy behind well-funded, politically motivated social media campaigns to create false narrative on COVID origins. Add Falun Gong/Epoch Times, 1000s of 'independent researchers', pseudo-virologists from Fintech1 /bioinformatics2 & right-wing media."
1. Fintech refers to the integration of technology into offerings by financial services companies in order to improve their use and delivery to consumers.
It primarily works by unbundling offerings by such firms and creating new markets for them. Startups disrupt incumbents in the finance industry by expanding financial inclusion and using technology to cut down on operational costs.
2. The leading journal in its field, Bioinformatics publishes the highest quality scientific papers and review articles of interest to academic and industrial researchers. Its main focus is on new developments in genome bioinformatics and computational biology
TRANSCRIPTION: "There are dozens and dozens of small spill overs going on on the planet at any one time which we just never see. Sure they never amplify. There are a few cases in there just absorbed in the illness the overall illness of the population right they could say so "who cares?" you know and that's one argument. But our strategy is any one of those could become pandemic there's a lot of stochasticity1 and what happens then? So if we look at all of them understand patterns try and reduce the number of spillover events we've got you know but if you say these are diverse coronaviruses and you can't vaccinate against them they're no antivirals what do we do? You're a virologist you know all the stuff; you can manipulate them in the lab pretty easily. It is just Spike Protein that drives a lot of what happens with the anonymous zoonotic risk. So you can get the sequence. You can build the protein and we work with Ralph Baric at University of North Carolina who does this insert into backbone of another virus and do some work in the lab so you can get more predictive. When you find a sequence you got this diversity. Now the logical progression for vaccines is, if you're going to develop a vaccine for SARS people are going to use. Let's try and insert some of these others and get a better vaccine. If you see something emerging it gives a head start on making a vaccine or a therapeutic. You know better knowledge of where they are as well so you can put your money into this clinics that matter and that's one of the big things that we've been trying to push. There's a lot of the word PREDICT or the anticipating, forecasting pandemics. IT DOESN'T MEAN YOU CAN START THEM. That's the problem there's so... What we're trying to say in a global scale if we can show where the most likely come from the species in most likely's origin and the people most like to get affected a global actor like W.H.O. our national government can better allocate resources to the highest risk. It's very simple so you're doing a lot of activities those are just a few of all the ones you can find." 1. The quality of lacking any predictable order or plan
DECEMBER 2019 - JANUARY 2020 Daszak deleted the e-mails containing his first reaction to the outbreak since he knew very well what had transpired. https://usrtk.org/biohazards-blog/scientists-masked-involvement-in-lancet-letter-on-covid-origin
GO BACK
DECEMBER 19, 2019 NYT - "Federal officials ended a moratorium imposed three years ago on funding research that alters germs to make them more lethal. Such work can now proceed, said Dr. Francis S. Collins, the head of the National Institutes of Health, but only if a scientific panel decides that the benefits justify the risks." JANUARY 5, 2020 "Pneumonia of unknown cause China Disease outbreak news January 5, 2020. On December 31, 2019, the W.H.O. China Country Office was informed of cases of pneumonia of unknown etiology (unknown cause) detected in Wuhan City, Hubei Province of China. According to the authorities, some patients were operating dealers or vendors in the Huanan Seafood market. Based on the preliminary information from the Chinese investigation team, no evidence of significant human-to-human transmission and no health care worker infections have been reported. Based on information provided by national authorities, W.H.O.s recommendations on public health measures and surveillance of influenza and severe acute respiratory infections still apply.
W.H.O. does not recommend any specific measures for travellers. In case of symptoms suggestive of respiratory illness either during or after travel, travellers are encouraged to seek medical attention and share travel history with their healthcare provider. W.H.O. advises against the application of any travel or trade restrictions on China based on the current information available on this event." JANUARY 2020 "Zheng-Li Shi, of the Wuhan Institute of Virology, is a senior author of the draft paper (not yet peer reviewed and so far available only in preprint) that gave nCoV-2019 its identity and name. It was Ms. Shi and her collaborators who, back in 2005, showed that the SARS pathogen was a bat virus that had spilled over into people. Ms. Shi and colleagues have been tracing coronaviruses in bats since then, warning that some of them are uniquely suited to cause human pandemics. In a 2017 paper, they set out how, after nearly five years of collecting fecal samples from bats in the Yunnan cave, they had found coronaviruses in multiple individuals of four different species of bats, including one called the intermediate horseshoe bat, because of the half-oval flap of skin protruding like a saucer around its nostrils. The genome of that virus, Ms. Shi and her colleagues have now announced, is 96 percent identical to the Wuhan virus that has recently been found in humans. And those two constitute a pair distinct from all other known coronaviruses, including the one that causes SARS. In this sense, nCoV-2019 is novel and possibly even more dangerous to humans than the other coronaviruses. I say possibly because so far, not only do we not know how dangerous it is, we cant know. Outbreaks of new viral diseases are like the steel balls in a pinball machine: You can slap your flippers at them, rock the machine on its legs and bonk the balls to the jittery rings, but where they end up dropping depends on 11 levels of chance as well as on anything you do. This is true with coronaviruses in particular: They mutate often while they replicate, and can evolve as quickly as a nightmare ghoul. Peter Daszak, the president of EcoHealth Alliance, a private research organization based in New York that focuses on the connections between human and wildlife health, is one of Ms. Shis longtime partners. Weve been raising the flag on these viruses for 15 years ever since SARS. He was a co-author of the 2005 bats-and-SARS study, and again of the 2017 paper about the multiple SARS-like coronaviruses in the Yunnan cave. Mr. Daszak told me that, during that second study, the field team took blood samples from a couple of thousand Yunnanese people, about 400 of whom lived near the cave. Roughly 3 percent of them carried antibodies against SARS-related coronaviruses. We dont know if they got sick. We dont know if they were exposed as children or adults, Mr. Daszak said. But what it tells you is that these viruses are making the jump, repeatedly, from bats to humans. In other words, this Wuhan emergency is no novel event. Its part of a sequence of related contingencies that stretches back into the past and will stretch forward into the future, as long as current circumstances persist. So when youre done worrying about this outbreak, worry about the next one. Or do something about the current circumstances. "Wuhan Institute of Virology senior scientist Zhengli Shi, who led the re-naming effort, described in an email to University of North Carolina virologist Ralph Baric, a fierce discussion among Chinese virologists over the name SARS-CoV-2. Deyin Guo, former dean of Wuhan Universitys School of Biomedical Sciences and co-author of the name-change proposal, wrote to CSG members that they had failed to consult their naming decision with virologists including the first discovers [sic] of the virus and the first describers of the disease from mainland China. It is not appropriate to use one disease-based virus name (like SARS-CoV) to name all other natural viruses that belong to the same species but have very different properties, he wrote in the correspondence sent on behalf of himself and five other Chinese scientists. The group proposed an alternative name Transmissible acute respiratory coronavirus (TARS-CoV). Another option, they said, could be Human acute respiratory coronavirus (HARS-CoV). JANUARY 24, 2020 Analysis published in the Lancet: "From JANUARY 10, 2020, we enrolled a family of six patients who travelled to Wuhan from Shenzhen between Dec 29, 2019 and Jan 4, 2020. Of six family members who travelled to Wuhan, five were identified as infected with the novel coronavirus. Additionally, one family member, who did not travel to Wuhan, became infected with the virus after several days of contact with four of the family members. None of the family members had contacts with Wuhan markets or animals, although two had visited a Wuhan hospital. Five family members (aged 3666 years) presented with fever, upper or lower respiratory tract symptoms, or diarrhoea, or a combination of these 36 days after exposure. They presented to our hospital (The University of Hong Kong-Shenzhen Hospital, Shenzhen) 610 days after symptom onset. They and one asymptomatic child (aged 10 years) had radiological ground-glass lung opacities." JANUARY 2020 The Shanghai laboratory where researchers published the worlds first genome sequence of the deadly coronavirus that causes Covid-19 has been shut down.
The laboratory at the Shanghai Public Health Clinical Centre was ordered to close for rectification on January 12, 2020 a day after Professor Zhang Yongzhens team published the genome sequence on open platforms. It closed temporarily the following day.
JANUARY 2020 Relying on the major science and technology infrastructure, this project aims to cultivate national high-level biosafety talents, to output significant scientific and technological breakthroughs and achievements, and to promote the scientific and technological support capabilities for biosafety and public health. According to the scientific and technological development programs of China, Chinese Academy of Sciences (CAS) and Wuhan Institute of Virology (WIV), CAS, the Call Announcement 2020 of Advanced Customer Cultivation Project of Wuhan National Biosafety Laboratory, CAS is released. Please apply for the project accordingly. The specific contents are as below...The research achievements attained during the project implementation, including theses, monographs, patents, software and database etc. shall be marked with Funded by Advanced Customer Cultivation Project of Wuhan National Biosafety Laboratory, Chinese Academy of Sciences. Any achievements unmarked will not be counted in the assessment. Wuhan Institute of Virology Chinese Academy of Sciences Jan 15th, 2020 JANUARY 3, 2020 Wuhan Institute of Virologys director general, Dr. WANG YANYI, messaged her colleagues, saying the National Health Commission told her the labs SARS-CoV-2 data shall not be published on social media and shall not be disclosed to the media. And on JANUARY 3, 2020 the commission sent this document, never posted online, but saved by researchers, telling labs to destroy SARS-CoV-2 samples or send them to the depository institutions designated by the state. Late Friday [May 16, 2020] the Chinese government admitted to the destruction
but said it was for public safety. The Chinese government explanation for the destruction of SARS-CoV-2 samples has no scientific credibility. For purposes of public safety any samples would surely be stored and studied, exactly as with the ones that were isolated from patients, and their RNA genomes decoded and published. (PBS News Hour May 22, 2020)
The National Health Commission is dictated to by the Leading Party Members' Groups. "Article 46 A leading Party members' group may be formed in the leading body of a central or local state organ, people's organization, economic or cultural institution or other non-Party unit. The group
plays the role of the core of leadership. Its main tasks are: to see to it that the Party's line, principles and policies are implemented, to discuss and decide on matters of major importance in its unit, to do well in managing affairs concerning cadres, to unite with the non-Party cadres and the masses in fulfilling the tasks assigned by the Party and the state and to guide the work of the Party organization of the unit and those directly under it."
FEBRUARY 2020 When the labs Shi Zhengli and colleagues published a paper with the viruss partial genome, they omitted any mention of the special sequence that supercharges the virus or the rare double CGG section. Yet the fingerprint is easily identified in the data that accompanied the paper. Was it omitted in the hope that nobody would notice this evidence of the gain-of-function origin? But in a matter of weeks virologists Bruno Coutard and colleagues published their discovery of the sequence in CoV-2 and its novel supercharged site. Double CGG is there; you only have to look. They comment in their paper that the protein that held it may provide a gain-of-function capability to the virus, for efficient spreading to humans. Wall Street Journal Opinion
FEBRUARY 27, 2020 In early 2018, during a meeting at the World Health Organization in Geneva, a group of experts I belong to (R&D Blueprint) coined the term Disease X We were referring to the next pandemic, which would be caused by an unknown, novel pathogen that hadnt yet entered the human population. As the world stands today on the edge of the pandemic precipice, its worth taking a moment to consider whether Covid-19 is the disease our group was warning about. Disease X, we said back then, would likely result from a virus originating in animals and would emerge somewhere on the planet where economic development drives people and wildlife together. Disease X would probably be confused with other diseases early in the outbreak and would spread quickly and silently; exploiting networks of human travel and trade, it would reach multiple countries and thwart containment. Disease X would have a mortality rate higher than a seasonal flu but would spread as easily as the flu. It would shake financial markets even before it achieved pandemic status. In a nutshell, Covid-19 is Disease X. Even as there are signs that the epidemics spread might be slowing in China, multiple communities and countries have now reported sustained transmission in their midst. The number of confirmed cases has exploded in South Korea in recent days. In Italy, villages and towns are on lockdown, Fashion Week in Milan has been disrupted and festivals are being canceled while public health authorities search for patient zero to identify who else is likely infected and may spread the disease in Europe. Iran appears to have become a new hub of transmission. The looming pandemic will challenge us in new ways, as people try to evade quarantines, and misinformation campaigns and conspiracy theorists ply their trade in open democracies. But as the world struggles to respond to Covid-19, we risk missing the really big picture: Pandemics are on the rise, and we need to contain the process that drives them, not just the individual diseases. Plagues are not only part of our culture; they are caused by it. The Black Death spread into Europe in the mid-14th century with the growth of trade along the Silk Road. New strains of influenza have emerged from livestock farming. Ebola, SARS, MERS and now Covid-19 have been linked to wildlife. Pandemics usually begin as viruses in animals that jump to people when we make contact with them. These spillovers are increasing exponentially as our ecological footprint brings us closer to wildlife in remote areas and the wildlife trade brings these animals into urban centers. Unprecedented road-building, deforestation, land clearing and agricultural development, as well as globalized travel and trade, make us supremely susceptible to pathogens like coronaviruses. Yet the worlds strategy for dealing with pandemics is woefully inadequate. Across the board, from politicians to the public, we treat pandemics as a disaster-response issue: We wait for them to happen and hope a vaccine or drug can be developed quickly in their aftermath. But even as Covid-19 rages, there still is no vaccine available for the SARS virus of 2002-3, nor for HIV/AIDS or Zika or a host of emerging pathogens. The problem is that between outbreaks, the will to spend money on prevention wanes, and the market for vaccines and drugs against sporadic viral diseases isnt enough to drive research and development. During its World Health Assembly in 2016, the W.H.O. set up the R&D Blueprint to bridge this gap and announced a priority list of pathogens that most threaten global health and for which no vaccines or drugs were in the pipeline. SARS made the list, as did MERS, Nipah, Ebola and other rare but serious diseases caused by epidemic viruses. The Coalition for Epidemic Preparedness Innovations a global partnership between public, private, philanthropic and civil society organizations launched at Davos in 2017 stepped up to the plate and sourced funding to develop vaccines and therapeutics against some of these. To escape from the Age of Pandemics, well need to treat them as a public health issue and start working on prevention in addition to responses. Our first goal should be to broaden our armory against potential mass epidemics. When some of us added Disease X to the W.H.O.s priority list two years ago, we wanted to make the point that its not sufficient to develop vaccines and drugs for known agents when the next big one is likely to be a different pathogen a virus close to SARS, say, but not close enough that the same vaccine can work against both. As Covid-19 strikes today and a spate of other pathogens are ready to emerge in the future, we continue to butt up against nature. Scientists estimate that there are 1.67 million unknown viruses of the type that have previously emerged in people. Discovering and sequencing them should be a priority a simple case of know your enemy. In the aftermath of SARS, research on coronaviruses originating in bats has discovered more than 50 related viruses, some of which have the potential to infect people; this information can now be used to test for broad-action vaccines and drugs. Scaling up this effort to cover all viral families, as the Global Virome Project proposes to do, is a logical first step toward prevention. A radical shift is also needed in the way that tests, vaccines and drugs are designed so that entire groups of pathogens are targeted instead of individual pathogens that are already known. The National Institute of Allergy and Infectious Diseases in the United States is working on a universal flu vaccine that would cover all known strains of influenza; a universal coronavirus vaccine, an Ebola-virus vaccine and others will also be needed. With a smaller investment, we can also try to get ahead of pandemics by working with communities in hot spots of emerging diseases. Disease surveillance should be focused on farmers, rural communities and anyone who has extensive contact with wildlife, to look for unusual illnesses, test for novel pathogens and work with people to develop alternatives to high-risk activities such as the wildlife trade. Pandemics are like terrorist attacks: We know roughly where they originate and whats responsible for them, but we dont know exactly when the next one will happen. They need to be handled the same way by identifying all possible sources and dismantling those before the next pandemic strikes. Peter Daszak is a disease ecologist and the president of EcoHealth Alliance, in New York. FEBRUARY 3, 2020 Thanks for sharing. Yes, I saw this earlier today and both Eddie and myself are actually quoted in it . It's a great article, but the problem is that our phylogenetic analyses aren't able to answer whether the sequences are unusual at individual residues, except if they are completely off. On a phylogenetic tree the virus looks totally normal and the close clustering with bats suggest that bats serve as the reservoir. The unusual features of the virus make up a really small part of the genome (<0.1%) so one has to look really closely at all the sequences to see that some of the features (potentially) look engineered.. We have a good team lined up to look very critically at this, so we should know much more at the end of the weekend. I should mention that after discussions earlier today, Eddie, Bob, Mike, and myself all find the genome inconsistent with expectations from evolutionary theory. But we have to look at this much more closely and there are still further analyses to be done, so those opinions could still change.
When Anderson realized that the NIH and Fauci were involved he said that his was preliminary research because his employer, The Scripts Institute, received most of it funding from the NIH and you don't bite the hand that feeds you. On FEBRUARY 19, 2020, The Lancet, among the most respected and influential medical journals in the world, published a statement that roundly rejected the lab-leak hypothesis, effectively casting it as a xenophobic cousin to climate change denialism and anti-vaxxism. Signed by 27 scientists, the statement expressed solidarity with all scientists and health professionals in China and asserted: We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin.
"Yet there have been no preprints or official scientific reports on the sampling, so it's not clear which, if any, animals tested positive. "Until you consistently isolate the virus out of a single species, it's really, really difficult to try and determine what the natural host is," says Kristian Andersen, an evolutionary biologist at Scripps Research. While the analyses above suggest that SARS-COV-2 may bind
human ACE2 with high affinity, computational analyses predict that the interaction is not ideal so it could not be man-made otherwise the binding would be ideal? Doesn't that depend on skill of the scientist or scientists altering the virus?
GO BACK
The study carefully examined whether key elements of the virus, particularly the spike protein on its surface, appeared engineered. They did not. The spike didnt optimally bind to its receptor, ACE-2, and the interaction between the two proteins was unpredictable even using the most advanced computer algorithms. Another key feature often cited as evidence of laboratory origin is the furin cleavage site, where the spike protein is cut in half to activate viral material for entry into cells. The viruses most closely related to SARS-CoV-2 dont have this site, but many others do, including other human coronaviruses. The furin site of SARS-CoV-2 has odd features that no human would design. Its sequence is suboptimal, meaning its cleavage by the enzyme furin is relatively inefficient. Any skilled virologist hoping to give a virus new properties this way would insert a furin site known to be more efficient. The SARS-CoV-2 site has more of the hallmarks of sloppy natural evolution than a human hand. Indeed, a timely analysis last year showed convincingly that it is a product of genetic recombination, a natural feature of coronavirus replication and evolution. WAPO
The virus was not man made because it is likely that SARS-COV-2-LIKE VIRUSES with partial or full POLYBASIC CLEAVAGE SITE PRRA will be discovered in other species It has yet to be discovered.
SARS-CoV-2 could have been derived from a new virus backbone
Anderson's bias against trump affects his research. Tweet deleted
FEBRUARY 3, 2020 Since the outbreak of severe acute respiratory syndrome (SARS) 18 years ago, a large number of SARS-related coronaviruses (SARSr-CoVs) have been discovered in their natural reservoir host, bats. Previous studies have shown that some bat SARSr-CoVs have the potential to infect humans. Here we report the identification and characterization of a new coronavirus (2019-nCoV), which caused an epidemic of acute respiratory syndrome in humans in Wuhan, China. The epidemic, which started on December 12, 2019, had caused 2,794 laboratory-confirmed infections including 80 deaths by January 26, 2020. Full-length genome sequences were obtained from five patients at an early stage of the outbreak. The sequences are almost identical and share 79.6% sequence identity to SARS-CoV. Furthermore, we show that 2019-nCoV is 96% identical at the whole-genome level to a bat coronavirus. Pairwise protein sequence analysis of seven conserved non-structural proteins domains show that this virus belongs to the species of SARSr-CoV. In addition, 2019-nCoV virus isolated from the bronchoalveolar lavage fluid of a critically ill patient could be neutralized by sera from several patients. Notably, we confirmed that 2019-nCoV uses the same cell entry receptor—Angiotensin Converting Enzyme II (ACE2)—as SARS-CoV. Characterization of full-length genome sequences from patients infected with a new coronavirus (2019-nCoV) shows that the sequences are nearly identical and indicates that the virus is related to a bat coronavirus." This is true. 2019-nCoV is related as a Coronavirus found in Horseshoe Bats because a natural SARS-CoV virus was modified by inducing mutations and a monster virus was created.
FEBRUARY 2020 The rapid, open, and transparent sharing of data on this outbreak is now being threatened by rumours and misinformation around its origins. We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin. Scientists from multiple countries have published and analysed genomes of the causative agent, severe acute respiratory syndrome coronavirus (SARS-CoV-2), and they overwhelmingly conclude that this coronavirus originated in wildlife, as have so many other emerging pathogens. This is further supported by a letter from the presidents of the US National Academies of Science, Engineering, and Medicine and by the scientific communities they represent. Conspiracy theories do nothing but create fear, rumours, and prejudice that jeopardise our global collaboration in the fight against this virus. We support the call from the Director-General of WHO to promote scientific evidence and unity over misinformation and conjecture. We want you, the science and health professionals of China, to know that we stand with you in your fight against this virus. MARCH 17, 2020 Nick Wade: "A statement which had enormous influence in shaping public attitudes was a letter (in other words an opinion piece, not a scientific article) published on March 17, 2020 in the journal Nature Medicine. Its authors were a group of virologists led by Kristian G. Andersen of the Scripps Research Institute. Our analyses clearly show that SARS-CoV-2 is not a laboratory construct or a purposefully manipulated virus, the five virologists declared in the second paragraph of their letter. True, some older methods of cutting and pasting viral genomes retain tell-tale signs of manipulation. But newer methods, called no-see-um or seamless approaches, leave no defining marks. Nor do other methods for manipulating viruses such as serial passage, the repeated transfer of viruses from one culture of cells to another. If a virus has been manipulated, whether with a seamless method or by serial passage, there is no way of knowing that this is the case. Dr. Andersen and his colleagues were assuring their readers of something they could not know."
MARCH 2020 "Despite extensive contact tracing of early cases during the COVID-19 pandemic, there have been no reported cases related to any laboratory staff at the WIV and all staff in the laboratory of Dr. Shi Zhengli were reported to be seronegative for SARS-CoV-2 when tested in March 2020 10.
MARCH 2020 "ABSTRACT: The SARS-CoV-2 epidemic started in late December 2019 in Wuhan, China, and has since impacted a large portion of China and raised major global concern. Herein, we investigated the extent of molecular divergence between SARS-CoV-2 and other related coronaviruses. Although we found only 4% variability in genomic nucleotides between SARS-CoV-2 and a bat SARS-related coronavirus (SARSr-CoV; RaTG13), the difference at neutral sites was 17%, suggesting the divergence between the two viruses is much larger than previously estimated. Our results suggest that the development of new variations in functional sites in the receptor-binding domain (RBD) of the spike seen in SARS-CoV-2 and viruses from pangolin SARSr-CoVs are likely caused by natural selection besides recombination. Population genetic analyses of 103 SARS-CoV-2 genomes indicated that these viruses had two major lineages (designated L and S), that are well defined by two different SNPs1 that show nearly complete linkage across the viral strains sequenced to date. We found that L lineage was more prevalent than the S lineage within the limited patient samples we examined. The implication of these evolutionary changes on disease etiology remains unclear. These findings strongly underscores the urgent need for further comprehensive studies that combine viral genomic data, with epidemiological studies of coronavirus disease 2019 (COVID-19)." It is an a priori asssumption that these anomalies were natural.
1. To make new cells, an existing cell divides in two. But first it copies its DNA so the new cells will each have a complete set of genetic instructions. Cells sometimes make mistakes during the copying process - kind of like typos. These typos lead to variations in the DNA sequence at particular locations, called single nucleotide polymorphisms, or SNPs (pronounced "snips").
MARCH 2020 Retraction of earlier article: "Oxford University publication Nature "Engineered bat virus stirs debate over risky research: lab-made corona virus related to SARS can infect human cells" "Editors note, We are aware that this story is being used as the basis for unverified theories that the novel coronavirus causing COVID-19 was engineered. There is no evidence that this is true; scientists believe that an animal is the most likely source of the coronavirus. APRIL 14, 2020 "The cables argued that the United States should give the Wuhan lab further support, mainly because its research on bat coronaviruses was important but also dangerous." washingtonpost.com/opinions/2020/04/14/state-department-cables-warned-safety-issues-wuhan-lab-studying-bat-coronaviruses/ In 2020 WIV tried to recruit a new staff http://english.whiov.cas.cn
GO BACK
APRIL 17, 2020 Exemption 5 is commonly interpreted as shielding from release draft government documents, records of sensitive deliberations before decisions are made, and government attorney-client deliberations. These types of documents deserve some protection. But agencies stretch the exemption to inappropriately cover other information, such as records that may paint the agency in a bad light, records that reveal problems, and records that contain embarrassing information.
APRIL 28, 2020 "A grant to a New York nonprofit ECOHEALTH ALLIANCE aimed at detecting and preventing future outbreaks of coronaviruses from bats has been canceled by the National Institutes of Health, Politico reports, apparently at the direction of President Donald Trump because the research involved the Wuhan Institute of Virology in China. On April 14, 2020 The Washington Post published a column highlighting State Department cables about concerns regarding safety at the institute." Dr. Shi Zhengli: "I have conducted collaborative research with Dr. Peter Daszak, President and PI of EcoHealth Alliance. We have established a good relationship in the fields of virus surveillance and pathogen discovery. Our research team has found a variety of coronaviruses with different sequences in bat populations. [Her researchers also created new viruses"]. Some of them have the potential to spread to humans and animals, such as SARS-related coronavirus, MERS-related coronavirus, SADS-related coronavirus, etc. The findings provide important clues for the prevention and control of infectious diseases. We dont understand the NIH termination of funding support for our collaborative project and feel it is absolutely absurd."
APRIL 2020 Daszak: What worries me the most is that we are going to miss the next emerging disease, that moves from one part of the planet to another, wiping out people as it moves along. Daszak said the purpose of his non-profit was to develop tools that could help researchers create diagnostics, treatments and vaccines for human outbreaks. MAY 1, 2020 "U.S. officials are investigating the Wuhan Institute of Virology as a potential source of the coronavirus pandemic. The request for records of gifts or contracts from the Wuhan Institute of Virology and its researcher Dr. Shi Zhengli known for her work on bats, is of a broader department investigation into possible faulty financial disclosures of foreign money by the Texas group of universities. As one of the nation's 14 Bio Safety Level 4 laboratories, the Galveston National Laboratory at UTMB belongs to the National Institute of Allergy and Infectious Diseases (NIAID) Biodefense Laboratory Network. It has collaborated with more than 70 countries and with scientists from the U.S. and abroad on biosafety and biosecurity, as part of its broad mission to advance global scientific collaboration. While it receives no financial support or gifts from global scientific laboratories, UTMB complies with obligations to report fiduciary relationships as required by statute." MAY 10, 2020 In the journal Current Biology, researchers describe a recently identified bat coronavirus that is SARS-CoV-2's closest relative in some regions of the genome and which contains insertions of amino acids at the junction of the S1 and S2 subunits of the virus's Spike Protein in a manner similar to SAR-CoV-2. While it's not a direct evolutionary precursor of SARS-CoV-2, this new virus, RmYN02, suggests that these types of seemingly unusual insertion events can occur naturally in coronavirus evolution, the researchers say. "Since the discovery of SARS-CoV-2 there have been a number of unfounded suggestions that the virus has a laboratory origin," says senior author Weifeng Shi, director and professor at the Institute of Pathogen Biology at Shandong First Medical University in China. "In particular, it has been proposed the S1/S2 insertion is highly unusual and perhaps indicative of laboratory manipulation. Our paper shows very clearly that these events occur naturally in wildlife. This provides strong evidence against SARS-CoV-2 being a laboratory escape." MAY 13, 2020: Two Wuhan Institute of Virology scientists, Xing-Yi Ge and Zhengli-Li Shi, use reverse genetics to generate a chimeric virus (one that has been created by combining cells of more than one distinct genotype) closely resembling the novel coronavirus Covid-19:On the basis of these findings, we synthetically re-derived an infectious full-length SHC014 recombinant virus and demonstrate robust viral replication both in vitro and in vivo. The Nature Medicine article also mentions, Human lungs for HAE cultures were procured under University of North Carolina at Chapel Hill Institutional Review Boardapproved protocols. The Chinese governments poor record of transparency; the fact that the Wuhan Institute of Virology, a research center with facilities in the same city where the virus first appeared, was studying and creating new dangerous pathogens, including bat coronaviruses cannot be easily dismissed.
MAY 2020 Alice Latinne, Ben Hu, Kevin J. Olival, Guangjian Zhu, Libiao Zhang, Hongying Li, Aleksei A. Chmura, Hume E. Field, Carlos Zambrana-Torrelio, Jonathan H. Epstein, Bei Li, Wei Zhang, Lin-Fa Wang, Zheng-Li Shi, Peter Daszak This article is a preprint and has not been certified by peer review. Importantly, the closest known relative of SARS-CoV-2, a SARS-related virus, was found in a Rhinolophus sp. bat in this regionf, although it is important to note that our survey was limited to China, and that the bat hosts of this virus also occur in nearby Myanmar and Lao PDR. The article really says nothing new. DOI: https://doi.org/10.1101/2020.05.31.116061
MAY 2020 It was the incompetence of China, and nothing else, that did this mass world-wide killing, Trump tweeted.
MAY 2020 Anthony Fauci tells National Geographic that everything about the stepwise evolution over time strongly indicates that [this virus] evolved in nature and then jumped species.
MAY 16, 2020 Sadly, were not much further than we were weeks and weeks ago, Pompeo said when asked how the investigation is going. The Chinese Communist Party has relentlessly deceived and denied information to the west. We have repeatedly asked to have teams go in and assist them to identify where the virus originated. We know it began in Wuhan, but we dont know from where or from whom, and those are important things. Your listeners need to know. We still need to know that so we can break the code on this thing. One of the key facts for scientists and epidemiologists to build out vaccines and therapeutics and to identify how this was ultimately delivered to the world. You have to know where patient zero began and how patient zero became infected. Yet, the Chinese Communist Party has at every turn attempted to manage the World Health Organization to manage the information flow, to punish doctors who wanted to talk about this publicly, to undermine the central understandings of transparency that every country has a responsibility to deliver. As a result of that, we still have so many unanswered questions. We, the United States, the world, have so many unanswered questions that are incredibly important so we can address these issues going forward to keep the American people safe. This isnt political. This isnt aimed at achieving a political victory. Its aimed squarely at getting the right health outcomes and saving lives across the world. MAY 19, 2020 "In early January, for example, China ordered samples of the virus to be destroyed, depriving the world of critical information. Even now, China continues to undermine the International Health Regulations by refusing to share accurate and timely data, viral samples and isolates, and by withholding vital information about the virus and its origins. And, to this day, China continues to deny international access to their scientists and relevant facilities, all while casting blame widely and recklessly and censoring its own experts...The World Health Organization has failed to publicly call on China to allow for an independent investigation into the origins of the virus, despite the recent endorsement for doing so by its own Emergency Committee. The World Health Organization's failure to do so has prompted World Health Organization member states to adopt the "COVID-19 Response" Resolution at this year's World Health Assembly, which echoes the call by the United States and so many others for an impartial, independent, and comprehensive review of how the World Health Organization handled the crisis. The resolution also
calls for an investigation into the origins of the virus, which is necessary for the world to understand how best to counter the disease." MAY 21, 2020 Days after the news conference in April, the National Institutes of Health emailed Peter Daszak, the head of EcoHealth Alliance. They questioned his work with the Wuhan Institute, and after an exchange of emails, he was informed that the renewal of his grant for more than $3 million was canceled. May 27, 2020 at 8:13 PM Randy W Schekman1 wrote: "Dear Peter, I am part of the Rich Roberts group and helped to line-up more Laureates to join the petition to Azar and Collins. We dont expect a response from them but we wish to make a constructive contribution to your essential work and have resolved to help find private funds to offset your loss. Our first success is with a foundation that makes anonymous contributions to various causes including in support of biomedical science. I am pleased to report that this group will provide the EcoHealth Alliance a grant of $500,000 to at least partially offset the NIH funds that were withdrawn from your program. Since they wish to remain anonymous, I will be happy to serve as the intermediary in transfer of funds to your program. We can communicate about how to proceed. Best wishes, Randy Schekman HHMI Investigator Dept. of Mol. and Cell Biology Li Ka Shing 2 Center UC Berkeley Berkeley, CA 94720-3370 (510) 642-5686 Stephanie Canon Assistant to Randy Schekman 484 Li Ka Shing Center Berkeley, CA 94720-3370 (510) 642-0594 stephaniecanon@berkeley.edu Re: A contribution to your work May 27, 2020 9:04:00 PM EDT Aleksei Chmura
1. Randy W. Schekman The Nobel Prize in Physiology or Medicine 2013 Prize motivation: "for their discoveries of machinery regulating vesicle traffic, a major transport system in our cells."
2. Li Ka Shing is considered to be a top pro-Beijing figure. Also in 2005, Li donated US$40 million to the University of California, Berkeley, citing that he was impressed with the university's accomplishments in the biosciences. In recognition of Li's donation, the university has named the campus' new biosciences facility the Li Ka Shing Center for Biomedical and Health Sciences, which opened in October 2011 https://en.wikipedia.org/wiki/Li_Ka-shing
One of Li Ka Shing's Hutchison Optel Telecom Technology Co., Ltd.; had ties to the Chinese Military. MAY 28, 2020 After the breakout Shi Zheng Li disappeared then made an appearance on TV to show she was alive and not liquidated by the CCP.
MAY 29, 2020 TRUMP: "The Chinese government has continually violated its promises to us and so many other nations these plain facts cannot be overlooked or swept aside. The world is now suffering as a result of the malfeasance of the Chinese government. China's cover-up of the Wuhan virus allowed the disease to spread all over the world instigating a global pandemic that has cost more than 100,000 in American lives and over a million lives worldwide. Chinese officials ignored their reporting obligations to the World Health Organization and pressured the World Health Organization to mislead the world when the virus was first discovered by Chinese authorities. Countless lives have been taken and profound economic hardship has been inflicted all around the globe. They strongly recommended against me doing the early ban from China but I did it anyway it was proven to be 100% correct. China has total control over the World Health Organization despite only paying 40 million dollars per year compared to what the United States has been paying which is approximately 450 million dollars a year. We have detailed the reforms that it must make and engage with them directly but they have refused to act because they have failed to make the requested and greatly needed reforms. We will be today terminating our relationship with the World Health Organization and redirecting those funds to other worldwide and serving urgent global public health needs. The world needs answers from China on the virus. We must have transparency. Why is it that China shut off infected people from Wuhan to all other parts of China. It went nowhere else. It didn't go to Beijing it went nowhere else but they allowed them to freely travel throughout the world including Europe and the United States. The death and destruction caused by this is incalculable. We must have answers not only for us but for the rest of the world. This pandemic has underscored the crucial importance of building up America's economic independence reshoring our critical supply chains and protecting America's scientific and technological advances. For years the government of China has conducted illicit espionage to steal our industrial secrets of which there are many today I will issue a proclamation to better secure our nation's vital university research and to suspend the entry of certain foreign nationals from China who we have identified as potential security risks." https://www.rev.com/blog/transcripts/donald-trump-press-conference-transcript-on-china-hong-kong-and-the-who
JULY 2020 Drs. Aleksei Chmura and Peter Daszak EcoHealth Alliance, Inc. 460 W 34th St Suite 1701 New York, NY 10001 Re: NIH Grant R01AI110964 Dear Drs. Chmura and Daszak:
"In follow-up to my previous letter of April 24, 2020, I am writing to notify you that the National Institute of Allergy and Infectious Diseases (NIAID), an Institute within the National Institutes of Health (NIH), under the Department of Health and Human Services (HHS), has withdrawn its termination of grant R01AI110964, which supports the project Understanding the Risk of Bat Coronavirus Emergence. Accordingly, the grant is reinstated. However, as you are aware, the NIH has received reports that the Wuhan Institute of Virology (WIV), a subrecipient of EcoHealth Alliance under R01AI110964, has been conducting research at its facilities in China that pose serious bio-safety concerns and, as a result, create health and welfare threats to the public in China and other countries, including the United States."
Grant award R01AI110964 is subject to biosafety requirements set forth in the NIH Grants Policy Statement (e.g., NIH GPS, Section 4.1.24 Public Health Security) and the Notice of Award (e.g., requiring that Research funded under this grant must adhere to the [CDC/NIH Biosafety in Microbiological and Biomedical Laboratories (BMBL)].). Moreover, NIH grant recipients are expected to provide safe working conditions for their employees and foster work environments conducive to high-quality research. NIH GPS, Section 4. The terms and conditions of the grant award flow down to subawards to subrecipients. 45 C.F.R. § 75.101. As the grantee, EcoHealth Alliance was required to monitor the activities of the subrecipient as necessary to ensure that the subaward is used for authorized purposes, in compliance with Federal statutes, regulations, and the terms and conditions of the subaward . . . 45 C.F.R. § 75.352(d). We have concerns that WIV has not satisfied safety requirements under the award, and that EcoHealth Alliance has not satisfied its obligations to monitor the activities of its subrecipient to ensure compliance. Moreover, as we have informed you through prior Notices of Award, this award is subject to the Transparency Act subaward and executive compensation reporting requirement of 2 C.F.R. Part 170. To date you have not reported any subawards in the Federal Subaward Reporting system.
Therefore, effective the date of this letter, July 8, 2020, NIH is suspending all activities related to R01AI110964, until such time as these concerns have been addressed to NIHs satisfaction. This suspension is taken in accordance with 45 C.F.R. § 75.371, Remedies for Noncompliance, which permits suspension of award activities in cases of non-compliance, and the NIH GPS, Section 8.5.2, which permits NIH to take immediate action to suspend a grant when necessary to protect the public health and welfare. This action is not appealable in accordance with 42 C.F.R. § 50.404 and the NIH GPS Section 8.7, Grant Appeals Procedures. However, EcoHealth Alliance has the opportunity to provide information and documentation demonstrating that WIV and EcoHealth Alliance have satisfied the above-mentioned requirements. Specifically, to address the NIHs concerns, EcoHealth must provide the NIH with the following information and materials, which must be complete and accurate:
1. Provide an aliquot1 of the actual SARS-CoV-2 virus that WIV used to determine the viral sequence.
2. Explain the apparent disappearance of Huang Yanling, a scientist / technician who worked in the WIV lab but whose lab web presence has been deleted.
3. Provide the NIH with WIVs responses to the 2018 U.S. Department of State cables regarding safety concerns.
4. Disclose and explain out-of-ordinary restrictions on laboratory facilities, as suggested, for example, by diminished cell-phone traffic in October 2019, and the evidence that there may have been roadblocks surrounding the facility from October 14-19, 2019.
5. Explain why WIV failed to note that the RaTG13 virus, the bat-derived coronavirus in its collection with the greatest similarity to SARS-CoV-2, was actually isolated from an abandoned mine where three men died in 2012 with an illness remarkably similar to COVID-19, and explain why this was not followed up.
6. Additionally, EcoHealth Alliance must arrange for WIV to submit to an outside inspection team charged to review the lab facilities and lab records, with specific attention to addressing the question of whether WIV staff had SARS-CoV-2 in their possession prior to December 2019. The inspection team should be granted full access to review the processes and safety of procedures of all of the WIV field work (including but not limited to collection of animals and biospecimens in caves, abandoned man-made underground cavities, or outdoor sites). The inspection team could be organized by NIAID, or, if preferred, by the U.S. National Academy of Sciences.
7. Lastly, EcoHealth Alliance must ensure that all of its subawards are fully reported in the Federal Subaward Reporting System During this period of suspension, NIH will continue to review the activities under this award, taking into consideration information provided by EcoHealth Alliance, to further asses compliance by EcoHealth Alliance and WIV, including compliance with other terms and conditions of award that may be implicated. Additionally, during the period of suspension, EcoHealth Alliance may not allow research under this project to be conducted. Further, no funds from grant R01AI110964 may be provided to or expended by EcoHealth Alliance or any subrecipients; all such charges are unallowable. It is EcoHealth Alliances responsibility as the recipient of this grant award to ensure that the terms of this suspension are communicated to and understood by all subrecipients. EcoHealth Alliance must provide adequate oversight to ensure compliance with the terms of the suspension. Any noncompliance of the terms of this suspension must be immediately reported to NIH. Once the original award is reinstated, NIH will take additional steps to restrict all funding in the HHS Payment Management System in the amount of $369,819. EcoHealth Alliance will receive a revised Notice of Award from NIAID indicating the suspension of these research activities and funding restrictions as a specific condition of award. Please note that this action does not preclude NIH from taking additional corrective or enforcement actions pursuant to 45 CFR Part 75, including, but not limited to, terminating the grant award. NIH may also take other remedies that may be legally available if NIH discovers other violations of terms and conditions of award on the part of EcoHealth Alliance or WIV. Sincerely, Michael S Lauer, MD NIH Deputy Director for Extramural Research. http://downloads.vanityfair.com/lab-leak-theory/Daszak_7_8_20_Reactivation_and_Suspension.pdf
1. A portion of a larger whole, especially a sample taken for chemical analysis or other treatment.
AUGUST 2020 The US National Institutes of Health (NIH) has reinstated a research organizations multimillion-dollar grant to study how coronaviruses move from bats to people which it cancelled in a controversial move earlier this year. But as Peter Daszak, the head of the small non-profit organization EcoHealth Alliance in New York City found out in early July, the funding cant be used unless the organization meets what he says are absurd conditions. The demands, detailed in a letter from the NIH, are politically motivated, he says. To carry out its research, EcoHealth Alliance partners with the Wuhan Institute of Virology (WIV) in China, which has been at the centre of unfounded rumours that the COVID-19 pandemic was caused by a coronavirus released from its laboratory. The NIH cancelled EcoHealth Alliances grant in April 2020, days after US President Donald Trump told a reporter that the United States would stop funding work at the WIV. EcoHealth Alliance and Daszak have been working with Shi Zhengli, a virologist at the WIV, for more than 15 years. Since 2014, an NIH grant has funded EcoHealths research in China, which involves collecting faeces and other samples from bats, and blood samples from people at risk of infection from bat-origin viruses. Scientific studies suggest that the SARS-CoV-2 coronavirus most likely originated in bats, and research on the topic could be crucial to identifying other viruses that might cause future pandemics. The WIV is a subrecipient on the grant. The NIH makes seven demands of EcoHealth; these were first reported by the Wall Street Journal this week, and have been viewed by Nature. The agency asks EcoHealth to obtain a vial of the SARS-CoV-2 sample that was used by the WIV to determine the viruss genetic sequence. It also requests that EcoHealth arranges an inspection of the WIV by US federal officials. Daszak calls the demands from the NIH heinous, and worries that the funding freeze is delaying vital work to identify and prevent the next pandemic. The NIH declined to comment, saying that the agency does not disclose internal deliberations on a specific grant. In an e-mail to Nature, Shi calls the NIHs demands outrageous and echoes Daszaks assertions that the agencys demands are unrelated to the WIVs collaboration with EcoHealth. Nature spoke with Daszak about being caught in the political cross-hairs.
How have you been doing since your grant was suspended?
DASZAK: Its just a very difficult and disappointing and worrisome time. Ive lost people that I know to COVID-19. Its a bat-origin coronavirus. We should have been there trying to find out why this thing emerged. And right now Im sat in my house, looking out the window, unable to do this work, because the NIH has told us that its not appropriate. Its extremely frustrating.
What is the aim of the project thats been affected?
DASZAK: We have specific aims: to go into rural regions of China to find bat-origin coronaviruses and determine which ones are high risk, then to work with local communities in rural China in five provinces. We target communities that are at high risk for viral spillover and see whether theyre getting infected and then finally see whether people are getting sick from novel coronaviruses.
How have you responded to the NIHs letter?
DASZAK: Weve written to the NIH and said: we think that this is inappropriate and wrong for the following reasons. Please, allow us to continue this work. But I doubt theyre going to do that right now. The NIH have told us not to work on this project. Obviously, were not going to break any NIH rules. But we have an ongoing collaboration, we have data that weve gathered over 15 years of working in China 5 years under a previous grant from the NIH which havent been published yet. So we need to carry on with that work.
How is your work relevant to the pandemic today?
DASZAK: We estimate that every single day, somebody in China or in southeast Asia gets infected with a new bat coronavirus. Right now, somebody is walking around, and they might be developing the first signs of a cough from the next COVID. We fully intend to be an organization that helps to prevent the next pandemic. That is our mission.
The NIH has asked you to obtain a vial of isolated SARS-CoV-2 from the WIV. Did you work on the novel coronavirus during your project?
DASZAK: The grant isnt used to fund work on SARS-CoV-2. Our organization has not actually published any data on SARS-CoV-2. We work on bat coronaviruses that are out there in the wild and try to predict what the next one is. We dont work on sequencing SARS-CoV-2. Its absurd because its absolutely outside the remit of the work we do.
Your grant has received widespread attention in the past few months. How has that been?
DASZAK: Conspiracy-theory outlets and politically motivated organizations have made Freedom of Information Act requests on our grants and all of our letters and e-mails to the NIH. We dont think its fair that we should have to reveal everything we do. When you submit a grant, you put in all your best ideas. We dont want to hand those over to conspiracy theorists for them to publish and ruin and make a mockery of.
What are you most worried about?
DASZAK: Were not a political organization. Were just trying to do good work to prevent pandemics. Were feeling the pressure of a very aggressive administration in the US picking fights with a country where we happen to be collaborating. And it turns out that they decided that this issue and our work is going to be one of the angles of attack. Thats extremely unfortunate. The winner in all of this is the virus, and not just this virus SARS-CoV-2 but all the other viruses. The GOP ORIGINS REPORT: "In an August 21, 2020, interview with Nature, after the NIH suspended the grants he was using to fund research at the WIV, Daszak claimed The grant isnt
used to fund work on SARS-CoV-2. Our organization has not actually published any data on SARSCoV-2. This is despite the fact that four days later Nature Communications published Origin and
cross-species transmission of bat coronaviruses in China. Daszak, Shi, Hu, and Wang are all listed as authors, with Shi and Daszak both being listed as corresponding authors. The preprint for the
article was uploaded on May 31, 2020, almost three months before Daszaks interview with Nature. The paper includes a phylogenetic analysis suggesting a likely origin for SARS-CoV-2 in
Rhinolophus spp. bats. Daszak, Shi, three EcoHealth Alliance affiliated researchers, and Linfa Wang are credited with designing the study, conducting fieldwork, and establishing collection and
testing protocols.
AUGUST 2020 EcoHealth Alliance Inc Grantee Website New York, New York, United States Purpose To support India's Ministry of Animal Husbandry and Dairying for establishment of National One Health Platform AUGUST 2020 REGION SERVED GLOBAL, ASIA COMMITTED AMOUNT $1,499,414 GRANT TOPIC Agricultural Development DURATION (MONTHS) 37
GRANTEE LOCATION New York, New York, United States
AUGUST 2020 The draft executive order created the "Trump Commission" named Sen. Tom Cotton as the commissioner, while Pompeos senior policy adviser Mary Kissel and his China adviser Miles Yu were to act as co-chair and vice co-chair or executive director. Retired Air Force Brig. Gen. Robert Spalding would lead the sessions on the geo-politics, and a general from Fort Detrick, the US biological weapons defense program in Maryland, would run the virology portion of the inquiry to investigate whether the virus had been manipulated in a lab. In a highly controversial move, Trumps top adviser on the coronavirus, Fauci, would be hauled in to explain why he funded risky coronavirus research in China and the not-for-profit EcoHealth Alliance president Peter Daszak would be grilled on the missing virus database from the Wuhan Institute of Virology, among other topics, such as why he outsourced this dangerous research to China.
JANUARY 2021
FEBRUARY 2021 Biden orders Pompeo investigation of WIV shut down writing it off as Trump conspiracy theory. Daszak: "Questions were raised about the legitimacy of the findings and the project was deemed to be an ineffective use of resources, explained a source familiar with the decision."
CHI COM AGENT OF INFLUENCE DASZAK HEADED W.H.O. TEAM THAT SUGGESTED COVID-19 ORIGINATED IN BARCELONA - ENTERED CHINA VIA FROZEN MEAT
MARCH 29, 2021 "One member of the team of experts, Peter Daszak, a British disease ecologist who runs EcoHealth Alliance, a New York-based pandemic prevention group, pushed back against the criticism of the teams work and of Chinas level of cooperation. He said the lab leak hypothesis was political from the start. Dr. Daszak added that the W.H.O. team was not restricted in its interviews with scientists who were on the ground at the start of the pandemic. He himself has been accused of having a conflict of interest because of his past research on coronaviruses with the Wuhan Institute of Virology, which, he said, was what a disease ecologist should be doing.We were in the right place because we knew that there was a risk of the virus emerging, said Dr. Daszak. We were working there with this exact viral group and it happened. https://www.nytimes.com/2021/03/29/world/asia/china-virus-WHO-report.htm
"It included one scientist from the U.S.: Peter Daszak, a zoologist and president of EcoHealth Alliance, a New York-based nonprofit. Dr. Daszak had experience hunting for the origins of emerging human viruses in animals, including 16 years working with researchers in China. He was on a team that pinpointed bats as the source of the coronavirus behind SARS. Some U.S. officials and scientists were concerned some of his nonprofits work in China posed a conflict of interest. EcoHealth had in past years provided funding to the Wuhan Institute of Virology as part of a grant from the U.S. National Institutes of Health. The WIV is at the center of assertions by the Trump administration that the pandemic virus could have come from a lab, and Dr. Daszak had publicly dismissed the possibility. In applying for a spot on the team, Dr. Daszak said, he described his expertise and provided a conflict-of-interest statement to the W.H.O. including his work with the WIV. Im known to W.H.O., and Im known for my work on this, he told the Journal. I recognized the historical importance of it and the value I would be able to bring to it. The W.H.O.s Mr. Jasarevic said Dr. Daszaks expertise in studying bats and zoonotic diseases was good experience for the team and didnt pose a conflict of interest. Dr. Daszak said he asked WIV researchers on the visit why a database of viruses the institute had publicly posted had been taken offline. Dr. Shi responded that the institute had had to take down the databasean Excel spreadsheetafter about 3,000 hacking attempts, Dr. Daszak said. The decision to rank the lab theory as extremely unlikely was unanimous, they said. I waited until everyone else had passed their opinion and then I passed mine, Dr. Daszak said, because of the sensitivities around my work with the Wuhan lab.READ THE WALL STREET JOURNAL ARTICLE IN FULL
For each of these possible pathways of emergence, the joint team conducted a qualitative risk assessment, considering the available scientific evidence and findings. It also stated the arguments against each possibility. The team assessed the relative likelihood of these pathways and prioritized further studies that would potentially increase knowledge and understanding globally. The joint teams assessment of likelihood of each possible pathway was as follows:
"The W.H.O. team opened the door to a theory embraced by Chinese officials, saying it was possible the virus might have spread to humans through shipments of frozen food, an idea that has gained little traction with scientists outside China. And the experts pledged to investigate reports that the virus might have been present outside China months before the outbreak in Wuhan in late 2019, a longstanding demand of Chinese officials. The virus was circulating in Wuhan several weeks before it appeared at the Hunan Seafood Wholesale Market, where some of the earliest clusters were initially reported, the experts said. Chinese officials heavily promoted the idea that the virus came from abroad at the news conference on Tuesday, arguing that the search for the origin of the virus should focus on places outside China." "A team of experts selected by the World Health Organization to investigate the origins of the virus that caused the Covid-19 pandemic returned last week from Wuhan, site of the worlds first outbreak. The team, having broken the ice with Chinese scientists, plans to produce a joint report on the possible origins of the virus.
The two groups of scientists, from China and the W.H.O., agreed to pursue some ideas that the Chinese government has been promoting, like the possibility that the virus was transported on frozen food. But the W.H.O. team also became frustrated by Chinas refusal to turn over raw data for analysis. Peter Daszak, a member of the W.H.O. team and the president of EcoHealth Alliance in New York, is primarily concerned with the animal origins of the virus. A specialist in animal diseases and their spread to humans, Dr. Daszak has worked with the Wuhan Virology Institute, a collaboration that last year prompted the Trump administration to cancel a grant to his organization. In an interview after his return to New York, he said that the visit had provided some new clues, which all of the scientists, Chinese and international, agreed most likely pointed to an animal origin within China or Southeast Asia. The scientists have largely discounted claims that the virus originated in a lab, saying that possibility was so unlikely that it was not worth further investigation." https://www.nytimes.com/2021/02/14/health/WHO-covid-daszak-china-virus.html "The supply chains to Huanan market included cold-chain products and animal products from 20 countries, including those where samples have been reported as positive for SARS-CoV-2 before the end of 2019 and those where close relatives of SARS-CoV-2 are found. There is evidence that some domesticated wildlife the products of which were sold in the market are susceptible to SARS-CoV, but none of the animal products sampled in the market tested positive in this study. In the early phase of pandemic, due to lack of awareness of the potential role of cold chain in virus introduction and transmission, the cold-chain products were not tested. These findings, however, do raise the possibility of different potential pathways of introduction. Preliminary sampling and testing of other markets in Wuhan and upstream suppliers to the Huanan market taken during 2020 did not reveal evidence of SARS-CoV-2 circulating in animals.
SARS-CoV-2 has been found to persist in conditions found in frozen food, packaging and cold-chain products. Index cases in recent outbreaks in China have been linked to the cold chain; the virus has been found on packages and products from other countries that supply China with cold-chain products, indicating that it can be carried long distances on cold-chain products.
Further analysis will examine spatial and temporal correlations and correct for underlying biases in sampling, and also to trace frozen products back to the Huanan market from suppliers" The Spanish flu, also known as the 1918 influenza pandemic, was an unusually deadly influenza pandemic caused by the H1N1 influenza A virus. Lasting from February 1918 to April 1920, it infected 500 million people about a third of the world's population at the time in four successive waves.
"It remains to be determined where SARS-CoV-2 originated. Although the virus was first identified as the cause of a cluster of cases of severe pneumonia in Wuhan, to date it is uncertain from where the first cases originated. A few studies suggest that cases may have occurred before December 2019, the time when circulation of SARS-CoV-2 was thought to have started in Hubei Province. In a retrospective survey, sewage samples collected on 12 March 2019 in Barcelona, Spain, were positive for SARS-CoV-2 RNA, but other samples collected between January 2018 and December 2019 were all negative. The PCR signals has not been confirmed by sequencing and could be false-positive signals. Collectively, these studies from different countries suggest that SARS-CoV-2 circulation preceded the initial detection of cases by several weeks. Some of the suspected positive samples were detected even earlier than the first case in Wuhan, suggesting that circulation of the virus in other regions had been missed. So far, however, the study findings were not confirmed, methods used were not standardized, and serological assays may suffer from non-specific signals. Nonetheless, it is important to investigate these potential early events." https://www.ub.edu/web/ub/en/menu_eines/noticies/2020/06/042.html "Three recent COVID-19 outbreaks in China have been linked to exposure to imported refrigerated or frozen seafood products.(27-30) An outbreak in Beijing linked to the Xinfadi market was first identified on 11 June 2020 after 56 days without a single known community case of COVID-19 in Beijing. Full genome sequencing and phylogenetic analysis of publicly available genomes suggests that the virus was from the L lineage European branch 1 with specific mutations characteristic to the market outbreak. However, it is not possible to fully infer the source of contamination from this work yet. In October 2020, an outbreak occurred in Qingdao. The index cases for the cluster were two dock workers from the citys port with no history of travel or recognized contact with anyone with confirmed COVID-19; the only epidemiological link which could be established between the cases was exposure to SARS-CoV-2 on the surface of cold-chain packaging. In addition, SARS-CoV-2 viruses were isolated from swabs of the outside surfaces of imported cold-chain packages in Qingdao. Based on these observations, China has launched a programme for systematic screening of packaged frozen imported food. Although re-introduction of a pandemic virus to epidemic-free areas can occur via various transmission routes including imported goods during a pandemic, the similarities between the outbreaks in the Beijing Xinfadi market and Qingdao, leading to the consideration of potential introduction of the virus through frozen products into the Huanan market in late 2019. For research focusing on the origin of SARS-CoV-2, this will need to be aligned with sources of those products. The proportion of cases in stalls with cold-chain goods (5.6%) is significantly higher than those without cold-chain goods (1.7%), and the relative risk of cases in stalls with cold-chain goods is 3.3 times higher than those without cold-chain goods (relative risk = 3.3, 95% CI:1.2-8.6), and the morbidity rate of vendors of cold-chain products is higher than others (3.3% compared with 1.4%), but there is no statistically significant difference. Epidemiological analysis showed that the first three cases in Huanan market all had a history of exposure to cold chain." "Analyses show that 60% (44/73) of the positive samples are related to 21 stalls, 19 of which were located in the western part of the Huanan market, and the remaining two stalls were located in the eastern part. 16 stalls were dealing with cold-chain product." An inventory was made of imported cold-chain products in large and medium-sized cold warehouses in Wuhan from September to December 2019. It has been confirmed that cold-chain products were still in stock during the above period. From 4-6 February 2021, samples were collected and SARS-CoV-2 NAT were performed on a total of 1055 samples of imported cold-chain food products (no domestic-origin cold chain products could be located at that time) including 330 pieces with outer packages, 244 pieces with inner packages and 481 food samples. The results of SARS-CoV-2 NAT were all negative." "It was noted that in one study, the infectivity of SARS-CoV-2 on cold-chain products did not decline after 21 days at 4 °C (refrigerated food) or at -20 °C (frozen food). Even at 21-23 °C, SARS-CoV-2 on cardboard surface remained infective up to 24 hours. After China successfully controlled the COVID-19 epidemic in Wuhan in April 2020, a series of clustered epidemics occurred in various places. According to the experience of prevention and control of these epidemics, especially the successful traceability results of Xinfadi in June, Dalian in July and Qingdao in October 2020, it is confirmed that SARS-CoV-2 can survive and maintain infection activity in cold chain products and packaging for a long time, which provides a scientific basis for the possibility of introduction of SARS-CoV-2 through cold chain products." "1. CoVs that are phylogenetically related to SARS-CoV-2 were identified in different animals from different countries, including bats (Rhinolophus spp) and customs-seized trafficked Malayan pangolins. Sampling and testing of >1,100 bats in Hubei Province, however, has been conducted but none were positive for viruses close to SARS-CoV-2. Sampling of wildlife across China has been conducted but no samples were positive for SARS-CoV-2.
2. The Huanan market had evidence of extensive sale of frozen products, fresh sea and aquatic animals and products, livestock meat, and limited farmed wildlife products. All the product samples retrieved during the outbreak investigation tested negative for the SARS-CoV-2 nucleic acid.
3. SARS-CoV-2 can persist in conditions found in frozen food, packaging and cold-chain products. Index cases in recent outbreaks in China have been linked to the imported cold chain. These indicates a possibility of transmission of SARS-CoV-2 through frozen products. The supply chains to the markets in Wuhan included cold-chain products (including the seafood, aquatic products, vegetables, animal products and farmed wildlife products) from several provinces in China and 20 other countries. Suppliers included countries and regions where SARS-CoV-2 (NAT and serum) tested positive before the outbreak of SARS-CoV-2, countries where cold chain imported products were sourced, provinces where domestic wildlife farms were sourced, and where the relatives of SARS-CoV-2 are found in bats and pangolins. There is evidence that some domesticated wildlife species sold in the Huanan market are susceptible to SARS-CoV-2 or SARS-CoV, but none of the animal products sampled in the market tested positive. Apart from frozen farmed wildlife products, cold-chain products in Huanan market were not tested specifically in early 2020. These findings do, however, raise the possibility for different potential pathways of introduction, stressing the need for careful trace-back of these supply chains and sample testing.
4. Preliminary sampling and testing at other markets in Wuhan and upstream suppliers to the Huanan market taken during 2020 did not reveal evidence of SARS-CoV-2 circulating in animals. Evidence was not found of presence of SARS-CoV-2 among animal products in the Huanan market and upstream suppliers.
5. Environmental sampling in the Huanan market demonstrated widespread contamination of surfaces with SARS-CoV-2, compatible with the virus shedding from infected people in the market at the end of December 2019. However, through extensive testing of animal products in the market, no evidence of animal infections was found. One environmental sample collected on Jan 22, 2020 on a second market tested positive, implying an environmental contamination from the patients in the communities.
6. Of 923 environmental samples in Huanan market 73 were positive; Forty-four of those positive were from the stalls of 21 vendors dealing in the following products: aquatic animals and products (n = 13), cold-chain products (n = 16), poultry meat (n = 6), seafood products (n = 6), livestock meat (n = 5), vegetable products (n = 2) and farmed wildlife meat (n = 1). Sampling and testing of 38 515 livestock and poultry samples and 41 696 wild animal samples from 31 provinces in China during 2018 to 2020 resulted in no positive SARS-CoV-2 antibody or nucleic acid tests. No evidence was found of circulation of SARS-CoV-2 among domestic livestock, poultry and wild animals before and after the SARS-CoV-2 outbreak in China." By searching for, and then counting, certain coronavirus genes in sewage, researchers hoped to determine whether the virus was present in a particular region and how widespread it was:
Although Covid-19 is primarily a respiratory disease, research conducted early in the pandemic revealed that people infected with the coronavirus often shed it in their stool. This finding, combined with the scale and urgency of the crisis, spurred immediate interest in tracking the virus by sampling wastewater.
In a study not yet published in a journal, scientists have reported that the new coronavirus was present in wastewater in Barcelona, Spain in March 2019, a finding that, if confirmed, would show that the pathogen had emerged much earlier than previously thought.But independent experts who reviewed the findings said they doubted the claim. The study was flawed, they said, and other lines of evidence strongly suggest the virus emerged in China late last year.Up until now, the earliest evidence of the virus anywhere in the world has been from December 2019 in China and it was only known to have hit mainland Spain in February 2020. Barcelona is a city that is frequented by Chinese people, in tourism and business, so probably this happened also elsewhere, and probably at the same time, said the lead author, Albert Bosch, a professor in the Department of Microbiology of the University of Barcelona who has been studying viruses in wastewater for more than 40 years. Several experts not involved in the research pointed out problems with the new study, which has not yet been subjected to the critical review by outside experts that occurs before publication in a scientific journal. They suggested that the tests might very well have produced false positives because of contamination or improper storage of the samples.I dont trust the results, said Irene Xagoraraki, an environmental engineer at Michigan State University. In 2017 Spain was only authorized to export frozen meat and offal and deboned ham to China. A new protocol changes everything, allowing China to import a much larger range of products from Spain, including chilled fresh meat and cured meats such as chorizo, salchichón, ham and shoulder.
"Many of the early cases were associated with the Huanan market, but a similar number of cases were associated with other markets and some were not associated with any markets. Transmission within the wider community in December 2019 could account for cases not associated with the Huanan market which, together with the presence of early cases not associated with that market, could suggest that the Huanan market was not the original source of the outbreak. Other milder cases that were not identified, however, could provide the link between the Huanan market and early cases without an apparent link to the market." "The animal and environment working group reviewed existing knowledge on coronaviruses that are phylogenetically related to SARS-CoV-2 identified in different animals, including horseshoe bats (Rhinolophus spp) and pangolins. However, the presence of SARS-CoV-2 has not been detected through sampling and testing of bats or of wildlife across China. More than 80 000 wildlife, livestock and poultry samples were collected from 31 provinces in China and no positive result was identified for SARS-CoV-2 antibody or nucleic acid before and after the SARS-CoV-2 outbreak in China. Through extensive testing of animal products in the Huanan market, no evidence of animal infections was found." W.H.O. - SARS-COV-2 IS INTRODUCED THROUGH A LABORATORY INCIDENT, REFLECTING AN ACCIDENTAL INFECTION OF STAFF FROM LABORATORY ACTIVITIES INVOLVING THE RELEVANT VIRUSES. WE DID NOT CONSIDER THE HYPOTHESIS OF DELIBERATE RELEASE OR DELIBERATE BIOENGINEERING OF SARS-COV-2 FOR RELEASE, THE LATTER HAS BEEN RULED OUT BY OTHER SCIENTISTS FOLLOWING ANALYSES OF THE GENOME. [What other scientists?]
"Although rare, laboratory accidents do happen, and different laboratories around the world are working with bat CoVs. When working in particular with virus cultures, but also with animal inoculations or clinical samples, humans could become infected in laboratories with limited biosafety, poor laboratory management practice, or following negligence. The closest known CoV RaTG13 strain (96.2%) to SARS-CoV-2 detected in bat anal swabs have been sequenced at the Wuhan Institute of Virology. The Wuhan CDC laboratory moved on December 2, 2019 to a new location near the Huanan market. Such moves can be disruptive for the operations of any laboratory." "The closest relatives of SARS-CoV-2 from bats and pangolin are evolutionarily distant from SARS-CoV-2. There has been speculation regarding the presence of human ACE2 receptor binding and a furin-cleavage site in SARS-CoV-2, but both have been found in animal viruses as well, and elements of the furin-cleavage site are present in RmYN02 and the new Thailand bat SARSr-CoV.1 There is no record of viruses closely related to SARS-CoV-2 in any laboratory before December 2019, or genomes that in combination could provide a SARS-CoV-2 genome. Regarding accidental culture, prior to December 2019, there is no evidence of circulation of SARS-CoV-2 among people globally and the surveillance programme in place was limited regarding the number of samples processed and therefore the risk of accidental culturing SARS-CoV-2 in the laboratory is extremely low. The three laboratories in Wuhan working with either CoVs diagnostics and/or CoVs isolation and vaccine development all had high quality biosafety level (BSL3 or 4) facilities that were well-managed, with a staff health monitoring programme with no reporting of COVID-19 compatible respiratory illness during the weeks/months prior to December 2019, and no serological evidence of infection in workers through SARS-CoV-2-specific serology-screening. The Wuhan CDC lab which moved on 2nd December 2019 reported no disruptions or incidents caused by the move. They also reported no storage nor laboratory activities on CoVs or other bat viruses preceding the outbreak." 1. The ACE2 receptor binding and a furin-cleavage site in animals is non-infectious.
In view of the above, a laboratory origin of the pandemic was considered to be extremely unlikely. What would be needed to increase knowledge?
Regular administrative and internal review of high-level biosafety laboratories worldwide. Follow-up of new evidence supplied around possible laboratory leaks. 1. Author information Affiliations EcoHealth Alliance, 460 West 34th Street, New York, 10001, New York, USA Kevin J. Olival, Parviez R. Hosseini, Carlos Zambrana-Torrelio, Noam Ross, Tiffany L. Bogich & Peter Daszak
"Coronavirus Update: Study Finds 781 Coronaviruses In Bats In China." Business Times - US ed. Date FEBRUARY 6, 2020 Persons Peter Daszak, Edward C. Holmes https://covid19.elsevierpure.com/en/clippings/coronavirus-update-study-finds-781-coronaviruses-in-bats-in-china
Nature Magazine: "That the WIV, a laboratory highly regarded for its work on bat coronaviruses, is located in the city where the outbreak first emerged is probably just a coincidence." https://www.nature.com/articles/d41586-020-01541-z 1. Alignment of the full-length genome sequence of the COVID-19 virus and other available genomes of Betacoronavirus showed the closest relationship was with the bat SARS-like coronavirus strain BatCov RaTG13, identity 96%. THE SIGNFICANCE OF THAT 4% DIFFERENCE
2. Transgenic human ACE2 mice and Rhesus monkey intranasally challenged by this virus isolate induced multifocal pneumonia with interstitial hyperplasia. The COVID-19 virus was subsequently detected and isolated in the lung and intestinal tissues of the challenged animals. ACE2 AFFINITY INDICATES DESIGNER VIRUS
3. COVID-19 is a zoonotic virus. From phylogenetics analyses undertaken with available full genome sequences, bats appear to be the reservoir of COVID-19 virus, but the intermediate host (s) has not yet been identified. However, three important areas of work are already underway in China to inform our understanding of the zoonotic origin of this outbreak. These include early investigations of cases with symptom onset in Wuhan throughout December 2019, environmental sampling from the Huanan Wholesale Seafood Market and other area markets, and the collection of detailed records on the source and type of wildlife species sold at the Huanan market and the destination of those animals after the market was closed...Early cases identified in Wuhan are believed to be have acquired infection from a zoonotic source as many reported visiting or working in the Huanan Wholesale Seafood Market. As of 25 February, an animal source has not yet been identified. MORE ON THE WUHNAN MARKET MYTH
4. Airborne spread has not been reported for COVID-19 and it is not believed to be a major driver of transmission based on available evidence; however, it can be envisaged if certain aerosol-generating procedures are conducted in health care facilities. Nationally, wildlife markets were closed and wildlife captive-breeding facilities were cordoned off.1
1. More of the Daszak CHI-COM animal to market to human CCP LINE.
Similarly, there is a long list of proposed studies on the origins of COVID-19, the natural history of the disease, and the viruss transmission dynamics. However, the urgency of responding to cases and saving lives makes it difficult for policy makers to consider and act on such comprehensive lists.1
1. In other words we must postpone any further investigation of the origin of COV-2 for the distant future..
D. Knowledge Gaps Knowledge gaps and key questions to be answered to guide control strategies include:
MARCH 4, 2021 BEIJINGA World Health Organization team investigating the origins of Covid-19 is planning to scrap an interim report on its recent mission to China amid mounting tensions between Beijing and Washington over the investigation and an appeal from one international group of scientists for a new probe. The group of two dozen scientists is calling in an open letter on Thursday for a new international inquiry. They say the W.H.O. team that last month completed a mission to Wuhanthe Chinese city where the first known cases were foundhad insufficient access to adequately investigate possible sources of the new coronavirus, including whether it slipped from a laboratory. Their appeal comes as the U.S.which recently reversed a decision to leave the W.H.O. lobbies for greater transparency in the investigation, saying it is waiting to scrutinize the report on the Wuhan mission, and urging China to release all relevant data, including on the first confirmed infections in December 2019, and potential earlier ones. Beijing, meanwhile, is pressing for similar W.H.O.-led missions to other countries, including the U.S., to investigate whether the virus could have originated outside China and spread to Wuhan via frozen food packaging. W.H.O. chief Tedros Adhanom Ghebreyesus said on Feb. 12 that the team would release an interim report briefly summarizing the Wuhan mission, possibly the following week, with a full report coming weeks later. But that summary report has yet to be published and the W.H.O. team is now scrapping that plan, said Peter Ben Embarek, the food-safety scientist who led the team. The W.H.O. team plans to publish a summary along with the full, final report, he said. That final report will be published in coming weeks and will include key findings, a W.H.O. spokesman said. ALSO SEE https://int.nyt.com/data/documenttools/covid-origins-letter/5c9743168205f926/full.pdf MARCH 21, 202160 Minutes: Daszak sticks to discredited Wuhan Market theory
MAY 14, 2021 More than one year after the breakout these "courageous" microbiologists are finally calling for lab leak investigation.
MAY 19, 2021 Statement on misinformation about NIH support of specific Gain-of-Function research. Based on outbreaks of coronaviruses caused by animal to human transmissions such as in Asia in 2003 that caused Severe Acute Respiratory Syndrome(link is external) (SARS), and in Saudi Arabia in 2012 that caused Middle East Respiratory Syndrome(link is external) (MERS), NIH and the National Institute of Allergy and Infectious Diseases (NIAID) have for many years supported grants to learn more about viruses lurking in bats and other mammals that have the potential to spill over to humans and cause widespread disease. However, neither NIH nor NIAID have ever approved any grant that would have supported gain-of-function research on coronaviruses that would have increased their transmissibility or lethality for humans. NIH strongly supports the need for further investigation by the World Health Organization (WHO) into the origins of the SARS-CoV-2 coronavirus. Working with a cross-regional coalition of 13 countries(link is external), we urge the W.H.O. to begin the second phase of their study without delay. Francis S. Collins, M.D., Ph.D. Director, National Institutes of Health MAY 21, 2021 The Wuhan Institute of Virology disclosed the genomic sequences of eight new viruses that were the second, third, fourth, fifth, sixth, seventh, eight and ninth genetically closest viruses to SARS-CoV-2. Their full sequences were only disclosed on May 21, even though the viruses were obtained from a mine in Mojiang in 2015 the same mine where RaTG13, a virus that shares a 96 per cent sequence identity with Covid-19, was found.
Severe respiratory disease coronavirus-2 (SARS-CoV-2) causes the most devastating disease, COVID-19, of the recent century. One of the unsolved scientific questions around SARS-CoV-2 is the animal origin of this virus. Bats and pangolins are recognized as the most probable reservoir hosts that harbor the highly similar SARS-CoV-2 related viruses (SARSr-CoV-2). Here, we report the identification of a novel lineage of SARSr-CoVs, including RaTG15 and seven other viruses, from bats at the same location where we found RaTG13 in 2015. Although RaTG15 and the related viruses share 97.2% amino acid sequence identities to SARS-CoV-2 in the conserved ORF1b region, but only show less than 77.6% to all known SARSr-CoVs in genome level, thus forms a distinct lineage in the Sarbecovirus phylogenetic tree. We then found that RaTG15 receptor binding domain (RBD) can bind to and use Rhinolophus affinis bat ACE2 (RaACE2) but not human ACE2 as entry receptor, although which contains a short deletion and has different key residues responsible for ACE2 binding. In addition, we show that none of the known viruses in bat SARSr-CoV-2 lineage or the novel lineage discovered so far use human ACE2 efficiently compared to SARSr-CoV-2 from pangolin or some of the SARSr-CoV-1 lineage viruses. Collectively, we suggest more systematic and longitudinal work in bats to prevent future spillover events caused by SARSr-CoVs or to better understand the origin of SARS-CoV-2. https://www.biorxiv.org/content/10.1101/2021.05.21.445091v1 MAY 26, 2021 "Back in early 2020, when COVID-19 emerged, I called for the CDC to get access to China to learn about the virus so we could fight it more effectively. The failure to get our inspectors on the ground in those early months will always hamper any investigation into the origin of COVID-19.
Nevertheless, shortly after I became President, in March, I had my National Security Advisor task the Intelligence Community to prepare a report on their most up-to-date analysis of the origins of COVID-19, including whether it emerged from human contact with an infected animal or from a laboratory accident. I received that report earlier this month, and asked for additional follow-up. As of today, the U.S. Intelligence Community has coalesced around two likely scenarios but has not reached a definitive conclusion on this question. Here is their current position: while two elements in the IC leans toward the former scenario and one leans more toward the latter each with low or moderate confidence the majority of elements do not believe there is sufficient information to assess one to be more likely than the other. I have now asked the Intelligence Community to redouble their efforts to collect and analyze information that could bring us closer to a definitive conclusion, and to report back to me in 90 days. As part of that report, I have asked for areas of further inquiry that may be required, including specific questions for China. I have also asked that this effort include work by our National Labs and other agencies of our government to augment the Intelligence Communitys efforts. And I have asked the Intelligence Community to keep Congress fully apprised of its work. The United States will also keep working with like-minded partners around the world to press China to participate in a full, transparent, evidence-based international investigation and to provide access to all relevant data and evidence." The leaders of the worlds wealthiest large democracies, at the Group of 7 summit, urged China to be part of a new investigation into the origins of the coronavirus. Mr. Biden told reporters that he and other leaders had discussed access to labs in China. This investigation is going to be a cover-up. The implications of a leak at the WIV are too far reaching and trace back to Baric and Daszak and others including Fauci. And no one wants to mess with China. Philip D. Zelikow investigation is also going to be a cover-up since he is a former CIA agent who worked under diplomatic cover.
MAY 2021 David Relman, a prominent microbiologist at Stanford: There are lots of cases where viruses go from bats to humans." But there were a number of reasons why the possibility of a lab leak remained viable. Chinese scientists have reportedly tested fifty thousand samples from three hundred species of wildlife, searching for the missing link, and hadnt yet found one carrying sars-CoV-2. The fact is, no one has found sars-CoV-2 anywhere other than in humans, MAY 30, 2021 The CCP: "As for the so-called undisclosed US intelligence report that some Wuhan researchers were infected by COVID-19 before an epidemic was confirmed, Dutch virologist Marian Koopmans attributed that to regular, seasonal sickness as early as in March. US intelligence agencies do not have superior research capability than the W.H.O., nor did they have more information. What they will get in the end is nothing but political. They will have to make a political decision either to produce a slanderous report that is in line with the US' anti-China campaign, or to step back in front of mainstream opposition from scientists and do their best to save the reputation of the US government that is already stuck in a dilemma. https://www.globaltimes.cn/page/202105/1224896.shtml
Marian Koopmans studied veterinary medicine at Utrecht University. She graduated from her Master's degree 1976, and remained there for her doctoral research. She earned two graduate degrees in veterinary medicine, and was officially registered as a veterinary microbiologist in 1977. She serves on the scientific advisory group of the World Health Organization. She co-authored papers with Fouchier, R.A.M. (Ron) Genetic adaptation of influenza A viruses in domestic animals and their potential role in interspecies transmission: a literature review
O Munoz, M De Nardi, K van der Meulen, K Van Reeth, Marian Koopmans, ...Ecohealth 13 , 171-198 Epidemiological risk factors for animal influenza A viruses overcoming species barriers
KA Harris, GS Freidl, OS Munoz, S von Dobschuetz, M De Nardi, ...Ecohealth 14 (2), 342-360 GENETIC ADAPTATION OF INFLUENZA A VIRUSES IN DOMESTIC ANIMALS AND THEIR POTENTIAL ROLE IN INTERSPECIES TRANSMISSION: A LITERATURE REVIEW (VOL 13, PG 171, 2016) O Munoz, M De Nardi, K Van Der Meulen, K Van Reeth, M Koopmans, ...Ecohealth 13 JUNE 7, 2021 A survey from The Economist/YouGov found 58 percent of Americans said COVID-19 came from a Wuhan lab, up from 49 percent who said the same when the pollsters first asked the question in May of 2020. Meanwhile, 18 percent said it was "definitely or probably false" that the virus came from a Chinese lab, while 24 percent said they didn't know.
JUNE 7, 2021 Wall Street Journal runs article conclusively proving SARS-CoV-19 was result of GoF / GoT experiments in WIV.
August 18, 2021 1. #DRASTIC Mass Decentralised Email Campaign May 12, 2020 Anthony S. Fauci, who runs the part of the National Institutes of Health that awarded EcoHealths grant and later terminated it, could face questions about the matter during testimony in front of a Senate committee. EcoHealths work in China first gained national attention during the pandemic when Trump, at a news conference in mid-April, pledged to pull its grant from NIH. Days later, the presidents personal attorney, Rudolph W. Giuliani, assailed the grant in a tweet. Neither Trump nor Giuliani mentioned the nonprofit organization by name, instead focusing on its partners in Wuhan. Late last month, NIH followed through on Trumps promise and abruptly pulled more than $3 million in funding over five years that was awarded in 2019. NIH gave the nonprofit organization little reason for doing so, writing in a letter that the project didnt align with program goals and agency priorities. But questions about exactly how and where the natural spillover took place, coupled with Chinas refusal so far to allow an independent investigation into the origins of the pandemic, have bred suspicion about whether the institute could be to blame. So has the coincidence that both the bat coronavirus research and the outbreak took place in the city of Wuhan....This idea that it doesnt fit the goals and objectives there must be more to it, EcoHealth Alliance President Peter Daszak said in a May 1 interview. You cant apply for money from the NIH unless its within the goals and objectives of the organization. They review that when you submit a proposal. NIH confirmed in a statement that EcoHealths grant was terminated and acknowledged that the Wuhan Institute of Virology was a sub-awardee.1
In other words Daszak "laundered" the grant for Fauci.
"The Office of the Director (OD) of NIAID determines Institute programs, plans, and policies and provides management, program analysis, and scientific program reporting services to the Institute, as well as scientific leadership, policy guidance, and overall operational and administrative coordination. The OD serves as the chief liaison with the National Institutes of Health (NIH) director, other components of of the U.S. Department of Health and Human Services (HHS), other federal agencies, Congress, professional societies, voluntary health organizations, and other public health groups. It also coordinates the activities of NIAID extramural and intramural divisions." Fauci IS science, so he tells us. FROM Greg Folkers (NIH/NIA ID) TO Anthony Fauchi: Nature ran a story in 2017 about it building a new blosafety level 4 lab and Included molecular biologist Richard Ebright of Rutgers University, Piscataway, expressing concerns about accidental infections, which he noted repeatedly happened NIH-002400 with lab workers handling SARS in Beijing. Ebright, who has a long history of raising red flags about studies with dangerous pathogens, also in 2015 criticized an experiment in which modifications were made to a SARS-like virus circulating in Chinese bats to see whether it had the potential to cause disease in humans. Earlier this week, Ebright questioned the accuracy of Bedford's calculation that there are at least 25 years of evolutionary distance between RaTG13- the virus held in the Wuhan virology institute - and 2019-nCoV, arguing that the mutation rate may have been different as it passed through different hosts before humans. Ebright tells Science lnsider that the 2019-nCoV data are "consistent with entry into the human population as a natural accident." Shi did not reply to emails from Science, but her longtime collaborator, disease ecologist Peter Daszak of the EcoHealth Alliance, dismissed Ebright's conjecture. "Every time there's an emerging disease, a new virus, the same story comes out: This is a spillover or the release of an agent or a bioengineered virus. It's just a shame. It seems humans can't resist controversy and these myths, yet it's staring us right in the face. There's this incredible diversity of viruses in wildlife and we've just scratched the surface. Within that diversity, there will be some that can infect people and within that group will be some that cause illness." Daszak and Shi's group have for 8 years been trapping bats in caves around China to sample their feces and blood for viruses. He says they have sampled more than 10,000 bats and 2000 other species. They have found some 500 novel coronaviruses, about 50 of wh ich fall relatively close to the SARS virus on the family tree, including RaTG13-it was fished out of a bat fecal sample they collected in 2013 from a cave in Moglang in Yunnan province . "We cannot assume that just because this virus from Yunnan has high sequence identity with the new one that that's the origin," Daszak says, noting that only a tiny fraction of coronaviruses that infect bats have been discovered. "I expect that once we've sampled and sampled and sampled across southern China and central China that we're going to find many other viruses and some of them will be closer [to 2019-nCoV]." It's not just a "curious interest" to figure out what sparked the current outbreak, Daszak says. "If we don't find the origin, it could still be a raging infection at a farm somewhere, and once this outbreak dies, there could be a continued spillover that's really hard to stop. But the jury is still out on what the real origins of this are." Information in this e-mail withheld under (b)(7) records or information compiled for law enforcement purposes, but only to the extent that the production of such law enforcement records or information ( A ) could be reasonably be expected to interfere with enforcement proceedings.
"I have always said, and will say today to you, [John Berman], that I still believe the most likely origin is from an animal species to a human, but I keep an absolutely open mind that if there may be other origins of that, there may be another reason, it could have been a lab leak. I believe if you look historically, what happens in the animal-human interface, that in fact the more likelihood is that you're dealing with a jump of species. But I keep an open mind all the time. And that's the reason why I have been public that we should continue to look for the origin. You can misconstrue it however you want -- that email was from a person to me saying 'thank you' for whatever it is he thought I said, and I said that I think the most likely origin is a jumping of species. I still do think it is, at the same time as I'm keeping an open mind that it might be a lab leak." The Crypto-Nazi LaRouche residue, whose info must be carefully vetted because it mixes real information with false information, such as Daszak is a British MI-5 Agent, reported: "After Daszak came to the United States and volunteered for the CDC, he joined and led the Consortium of Conservation Medicine (CCM) beginning in 2004. CCM, and its successor, EcoHealth Alliance, is a post-modern concoction combining eco-fascism and public health. CCM works to prevent the development of new infrastructure projects within impoverished nations to ostensibly stop the spread of disease. This means they prevent development such as energy, water, and sanitation projects, which are the very foundations for the miracle of modern public health. Instead, CCM looks to cultivate a catalogue of potential new viruses while protecting their natural reservoirs. Here is what their own website says: Wild animals like bats and rodents are the natural reservoir for zoonotic pathogens and have carried these pathogens for thousands of years. They are not to blame for these disease emergence eventsit is humans who are responsible for altering the environment and increasing contact and conflict with these important species. By 2010 Daszaks Consortium of Conservation Medicine joined with an operation called the Royal Society of Wildlife Trust, which was initially established by Gerald Durrell as The Wildlife Preservation Trust International in 1963, and with the Dodo bird as its symbol, is still run from the Crowns Channel Island of Jersey. The Royal Society of Wildlife Trust, set up in 1999, is now an organization of forty-six different wildlife trusts operated from the Crowns Channel Islands of Man and Alderney. EIR - LaRouche
Also see: ECO-HEALTH ABOUT
Wildlife Trust is a nonprofit organization that began in 1971 with a focus on wildlife conservation, but over the past 20 years, the organization has expanded its focus to include conservation medicine based on research about the relationships between ecosystems and animal and human health around the world. Throughout the past two years, the organizations board of directors, staff and alliance members have been working on rebranding Wildlife Trust, and today, the new brand was introduced officially via a press release. Wildlife Trust has been renamed as EcoHealth Alliance with a new website and slogan: Local Conservation, Global Health. The goal of the rebranding effort is to more accurately present the organizations dual focus on local wildlife conservation and its ongoing leadership in the area of conservation medicine and the relationships between animal ecosystems and human health. The effort wont stop with a new name, logo and website. EcoHealth Alliance is also hiring new scientists, forming new alliance partnerships, and developing more programs to support the brands dual focus. As the EcoHealth Alliance website explains: EcoHealth Alliance works at the intersection of ecosystem, animal and human health through local conservation programs and develops global health solutions to emerging diseases. Furthermore, EcoHealth Alliance is marketing its new brand focus with messaging that positions the organization as being on, the forefront of informing the public, businesses, and the scientific community about emerging diseases, including potential pandemics.
COMMENT: If it was the organizations goal to shift its brand focus, then one would think that they made a wise strategic move in rebranding to better promote their efforts related to health and medicine. To the average individual, the Wildlife Trust was very much rooted in wildlife conservation with the masses knowing very little about the organizations other priorities. The new branding puts health and medicine center stage with wildlife conservation taking a back seat. It appears that this was the intent of the rebranding effort, and if thats true, then the rebranding should work well.
What do you think? Does the rebranding work? When you look at the new logo and hear the new brand name, do you still think of wildlife conservation or has the entire brand promise shifted with this rebranding effort? Im leaning toward the latter. I wonder if that was truly the organizations intent. www.corporate-eye.com
WILD LIFE TRUSTS is the trading name of the Royal Society of Wildlife Trusts, is an organisation made up of 46 local Wildlife Trusts in the United Kingdom, the Isle of Man and Alderney. The Wildlife Trusts, between them, look after more than 2,300 nature reserves, covering around 98,500 hectares (243,000 acres). As of 2020, the Trusts have a combined membership of over 850,000 members. The Royal Society of Wildlife Trusts (RSWT) is an independent charity, with a membership formed of the 46 individual charitable Trusts. It acts as an umbrella group for the individual Wildlife Trusts, as well as operating a separate Grants Unit which administers a number of funds. Charles, Prince of Wales serves as the patron of the Wildlife Trusts. David Bellamy was president of The Wildlife Trusts for ten years between 1995 and 2005, Foundation Money
Doctor Richard Ebright, the foremost authority on biological warfare in the U.S., has been on to incalculable dangers of conducting Gain-of-Threat experiments. He had provided valuable information on the EcoHealth Alliance:
(1) Three EcoHealth/WIV lab-generated viruses exhibited >10x to >10,000x higher viral load than the starting bat virus in humanized mice. ECO-HEALTH ALLIANCE scam was just another money making operation that got millions in USG grants. The FBI was unable to identify records responsive to my request [#1495656-000 5/4/21]. Daszak' Eco-Health Alliance's operations need to be examined by congress. His bogus programs that didn't predict COVID-19 in fact it produced it!. Daszak's Ecohealth Alliance diverted $600,000 in grants from U.S. National Institutes of Health to the WIV between 2014 and 2019 to outsource gain-of-function / gain-of-threat experiments with bat coronaviruses. https://taggs.hhs.gov/Detail/RecipDetail?arg_RecipId=ZfeVER2uWwKllnTv6X4AxA%3D%3D
Ecohealth Alliance Inc. is a Florida Foreign Non-Profit Corporation filed On December 4, 2009. The company's filing status is listed as Inactive and its File Number is F09000004818. The company's principal address is 460 W. 34th St., New York, NY 10001 and its mailing address is 460 West 34th Street 17th Floor, New York, NY 10001. The company has 5 principals on record. The principals are Peter Daszak from New York NY, Robert Hoquet from New York NY, Nells Lippert from New York NY, Ellen Shedlarz from New York NY, and Carol C Timmis from New York NY.
https://www.bizapedia.com/fl/ecohealth-alliance-inc.html
This statement was written in the New York Times earlier this year by Peter Daszak. Daszak is the longtime president of the EcoHealth Alliance, a New York-based non-profit whose claimed focus is pandemic prevention.
But the EcoHealth Alliance, it turns out, is at the very center of the COVID-19 pandemic in many ways. To depict the pandemic in such militarized terms is, for Daszak, a commonplace.
In an Oct. 7 online talk organized by Columbia University’s School of International and Public Affairs, Daszak presented a slide titled “Donald Rumsfeld’s Prescient Speech:” “There are known knowns; there are things we know that we know. There are known unknowns; that is to say, there are things that we know we don’t know. But there are also unknown unknowns there are things we don’t know we don’t know. (This Rumsfeld quote is in fact from a news conference.) In the subsequent online discussion, Daszak emphasized the parallels between his own crusade and Rumsfeld’s, since, according to Daszak, the “potential for unknown attacks” is “the same for viruses.” Daszak then proceeded with a not terribly subtle pitch for over a billion dollars. This money would support a fledgling virus hunting and surveillance project of his, the Global Virome Project a “doable project” he assured watchers given the cost of the pandemic to governments and various industries. Also on the video was Columbia University professor Jeffrey Sachs.
Sachs is a former special advisor to the UN, the former head of the Millennium Villages Project, and was recently appointed Chair of the newly-formed EAT Lancet Commission on the pandemic. In September 2021, Sachs’ commission named Daszak to head up its committee on the pandemic’s origins. Daszak is also on the WHO’s committee to investigate the pandemic’s origin. He is the only individual on both committees. These leadership positions are not the only reason why Peter Daszak is such a central figure in the COVID-19 pandemic, however. His appointment dismayed many of those who are aware that Daszak’s EcoHealth Alliance funded bat coronavirus research, including virus collection, at the Wuhan Institute for Virology (WIV) and thus could themselves be directly implicated in the outbreak.
For his part, Daszak has repeatedly dismissed the notion that the pandemic could have a lab origin. In fact, a recent FOIA by the transparency group U.S. Right To Know revealed that Peter Daszak
drafted an influential multi-author letter published on February 18 in the Lancet. That letter dismissed lab origin hypotheses as conspiracy theory. Daszak was revealed to have orchestrated
the letter such as to avoid the appearance of a political statement.
Sachs for his part seemed surprised by Daszak’s depiction of Rumsfeld but Daszak reassured him. “It’s an awesome quote! And yes, it’s
Donald Rumsfeld, Jeff, and I know he’s a Republican, but what a genius!
FOLLOWING THE ECOHEALTH ALLIANCE’S MONEY TRAIL TO THE PENTAGON For much of this year, Daszak’s EcoHealth Alliance garnered a great deal of sympathetic media coverage after its $3.7 million five-year NIH grant was prematurely cut when the Trump administration learned that EcoHealth Alliance funded bat coronavirus research at the WIV. The temporary cut was widely depicted in major media as Trump undermining the EcoHealth Alliance’s noble fight against pandemics. The termination was reversed by NIH in late August 2020, and even upped to $7.5 million. But entirely overlooked amid the claims and counter-claims was that far more funding for the EcoHealth Alliance comes from the Pentagon than the NIH. To be strictly fair to the media, Daszak’s EcoHealth Alliance obscures its Pentagon funding. On its website EcoHealth Alliance states that “A copy of the EHA Grant Management Manual is available upon request to the EHA Chief Financial Officer at finance ( at ) ecohealthalliance.org”. But an email to that address and numerous others, including Peter Daszak’s, requesting that Manual, as well as other financial information, was not returned. Neither were repeated voicemails. Only buried under their “Privacy Policy,” under a section titled “EcoHealth Alliance Policy Regarding Conflict of Interest in Research,” does the EcoHealth Alliance concede it is the “recipient of various grant awards from federal agencies including the National Institute of Health, the National Science Foundation, US Fish and Wildlife Service, and the US Agency for International Development and the Department of Defense.” Even this listing is deceptive. It obscures that its two largest funders are the Pentagon and the State Department (USAID); whereas the US Fish and Wildlife Service, which accounts for a minuscule $74,487, comes before either.
Meticulous investigation of US government databases reveals that Pentagon funding for the EcoHealth Alliance from 2013 to 2020, including contracts, grants and
subcontracts, was just under $39 million. Most, $34.6 million, was from the Defense Threat Reduction Agency (DTRA), which is a branch of the DOD which states it is tasked to “counter and deter weapons of mass destruction and improvised threat networks.” Most of the remaining money to EHA was from USAID (State Dept.), comprising at least $64,700,000 (1).
These two sources thus total over $103 million. (See Fig). Another $20 million came from Health and Human Services ($13 million, which includes National Institutes of Health and Centers for Disease Control), National
Science Foundation ($2.6 million), Department of Homeland Security ($2.3 million), Department of Commerce ($1.2 million), Department of Agriculture ($0.6 million), and Department of Interior ($0.3 million). So, total US government funding for EHA to-date stands at $123 million, approximately one third of which comes from the Pentagon directly. The full funding breakdown is available here and is summarized by year, source, and type, in a spreadsheet format. PDF versions of this spreadsheet are available to download. The summary is here and all Federal grants and contracts are here. MORE MILITARY CONNECTIONS The military links of the EcoHealth Alliance are not limited to money and mindset. One noteworthy ‘policy advisor’ to the EcoHealth Alliance is David Franz. Franz is former commander of Fort Detrick, which is the principal US government biowarfare/biodefense facility. David Franz was part of UNSCOM which inspected Iraq for alleged bioweapons
what were constantly referred to as WMDs or Weapons of Mass Destruction by the US government and the media. Franz has been one of those eager to state, at least when discussing alleged Iraqi programs, that “in biology … everything is dual use the people, the facilities and the equipment.” (NPR, May 14, 2003; link no longer available). Just this year Franz wrote a piece with former New York Times journalist Judith Miller, whose stories of Iraqi WMDs did much to misinform the US public regarding the case for the 2003 invasion of Iraq. Their joint article, “A Biosecurity Failure: Americas key lab for fighting infectious disease has become a Pentagon backwater,” urges more funding for Fort Detrick. Miller and Franz are longtime associates. Miller co-wrote the book Germs, released amid
the 2001 false flag anthrax attacks, which repeatedly quotes Franz. Miller at the time received a hoax letter with a harmless white powder, increasing her prominence. Franz continued hyping the existence of Iraqi WMDs even after the invasion of Iraq. While she was still with the Times, Miller quoted him in a story “US Analysts Link Iraq Labs To Germ Arms” on May 21, 2003 pushing the theory that Iraq had mobile biological WMD units. (This theory was debunked by the British scientist Dr. David Kelly, who would die, apparently by suicide, soon thereafter.1
1. Samuel is hinting the Mossad took him out with no evidence to back up this assertion.
Four significant insights emerge from all this. First, although it is called the EcoHealth Alliance, Peter Daszak and his nonprofit work closely with the military. Second, the EcoHealth Alliance attempts to conceal these military connections. Third, through militaristic language and analogies Daszak and his colleagues promote what is often referred to as, and even then somewhat euphemistically,
an ongoing agenda known as “securitization“. In this case it is the securitization of infectious diseases and of global public health. That is, they argue that pandemics constitute a vast and existential threat. They minimize the very real risks associated with their work, and sell it as a billion dollar solution. The fourth insight is that Daszak himself, as the Godfather of the Global Virome Project, stands to benefit from the likely outlay of public funds. Acknowledgments Thanks to James Baratta and Mariamne Everett for researching the funding sources. Footnote
This originally appeared in Independent Science News.
Sam Husseini is a founder of VotePact.org which helps break out of the two party bind. His latest personal writings
are at http://husseini.posthaven.com/ and tweets at http://twitter.com/samhusseini.
https://pubmed.ncbi.nlm.nih.gov/14569023
www.sciencemag.org/cgi/rapidpdf/1085952v1.pdf
2002-2004 THE WIV SCIENTISTS BEGAN COLLECTING BAT VIRUS SINCE THE SARS-1 OUTBREAK
DEVELOPMENT OF MOUSE HEPATITIS VIRUS MHV AND SARS-COV INFECTIOUS CDNA CONSTRUCTS (BARIC)
https://www.researchgate.net/publication/8119695_Development_of_mouse_hepatitis_virus_and_SARS-CoV_infectious_cDNA_constructs
COMPOSITIONS OF CORONAVIRUSES WITH A RECOMBINATION-RESISTANT GENOME (BARIC)
https://patents.justia.com/patent/7618802
REVIEW OF BATS AND SARS
https://wwwnc.cdc.gov/eid/article/12/12/06-0401_article
GIVING A SARS-LIKE VIRUS THE ABILITY TO INFECT HUMANS IS GAIN-OF-THREAT



SHI ZHENGLI: DIFFERENCE IN RECEPTOR USAGE BETWEEN SEVERE ACUTE RESPIRATORY SYNDROME (SARS) CORONAVIRUS AND SARS-LIKE CORONAVIRUS OF BAT ORIGIN
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2258702/
PETER DASZAK: RISK OF VIRAL EMERGENCE FROM BATS (DASZAK)
RON FOUCHIER, EARLY GAIN-OF-THREAT "SCIENTIST CREATED AN AVIAN FLU VIRUS THAT COULD INFECT HUMANS
https://www.sciencemag.org/news/2011/11/scientists-brace-media-storm-around-controversial-flu-studies
WORLD HEALTH ORGANIZATION IS IN FAVOR OF SHARING H5N1 VIRUS GAIN-OF-FUNCTION / GAIN-OF-THREAT RESEARCH
https://www.bmj.com/content/344/bmj.e30 (Published 04 January 2012)
W.H.O. CONCERNED THAT NEW H5N1 INFLUENZA RESEARCH COULD UNDERMINE THE 2011 PANDEMIC INFLUENZA PFREPAREDNESS FRAMEWORK
http://www.who.int/mediacentre/news/statements/2011/pip_framework_20111229/en/index.html
THE PAPER DESCRIBING HOW THE LETHAL H5N1 BIRD FLU COULD BE MADE EASIER TO SPREAD WAS PUBLISHED SIX MONTHS AFTER A SCIENTIFIC ADVISORY BOARD SUGGESTED THAT THE
PAPERS MOST POTENTIALLY DANGEROUS DATA BE CENSORED.
https://www.nytimes.com/2012/06/22/health/h5n1-bird-flu-research-that-stoked-fears-is-published.html
FOUCHIER EXPLAINED HOW EASY IT WAS TO CREATE AN AIRBORNE AVIAN FLU VIRUS THAT COULD INFECT HUMANS USING GAIN-OF-THREAT TECHNOLOGY
"Flu viruses are basically eight pieces of RNA wrapped up in a ball. To create the gain-of-threat mutations, use a DNA template for each piece, called a plasmid. Making a single mutation in the plasmid is easy and it's commonly done in genetics labs. If you insert all eight plasmids into a mammalian cell, they hijack the cell's machinery to create flu virus RNA. Now you can start to assemble a new virus particle in that cell. One infected cell is enough to grow many new virus particles from one to a thousand to a million; viruses are replication machines. And because they mutate so readily during their replication, the new viruses have to be checked to make sure it only has the mutations the lab caused. The virus then goes into the ferrets, passing through them to generate new viruses until, on the 10th generation, it infected ferrets through the air. By analyzing the virus's genes in each generation, one can figure out what exact five mutations lead to H5N1 bird flu being airborne between ferrets."
US-CHINA ECOLOGY AND EVOLUTION OF INFECTIOUS DISEASES COLLABORATIVE WORKSHOP; KUNMING, CHINA
BATS AS ANIMAL RESERVOIRS FOR THE SARS CORONAVIRUS: HYPOTHESIS PROVED AFTER 10 YEARS OF VIRUS HUNTING VIROLOGICA SINICA
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7091109
NIAD AND HIH GRANTS
ONE OF SEVERAL NEAR FATAL ERRORS THAT TRIGGERED THE GoF / GOT BAN INVOLVED H5N1 INFLUENZA VIRUS
https://www.nytimes.com/2014/07/12/science/cdc-closes-anthrax-and-flu-labs-after-accidents.html
https://www.cdc.gov/labs/pdf/Final_Anthrax_Report.pdf OBAMA BANS GAIN-OF-FUNCTION / GAIN-OF-THREAT RESEARCH IN US
https://www.freethink.com/articles/gain-of-function-mutation

DOING DILIGENCE TO ASSESS THE RISKS AND BENEFITS OF LIFE SCIENCES GAIN-OF-FUNCTION RESEARCH
https://obamawhitehouse.archives.gov/blog/2014/10/17/doing-diligence-assess-risks-and-benefits-life-sciences-gain-function-research
GAIN-OF-FUNCTION / GAIN-OF-THREAT OUTSOURCED TO WUHAN INSTITUTE OF VIROLOGY AFTER AMERICA BANS IT AS TOO DANGEROUS
https://asiatimes.com/2020/04/why-us-outsourced-bat-virus-research-to-wuhan
https://www.snopes.com/fact-check/obama-admin-wuhan-lab-grant
UNDERSTANDING THE RISK OF BAT CORONAVIRUS EMERGENCE (DASZAK)
IN VITRO AND IN VIVO CHARACTERIZATION OF SARSR-COV SPILLOVER RISK, COUPLED WITH SPATIAL AND PHYLOGENETIC ANALYSES TO IDENTIFY THE REGIONS AND VIRUSES OF PUBLIC HEALTH CONCERN (DASZAK)
https://www.nih.gov/news-events/news-releases/nih-researchers-identify-key-genomic-features-could-differentiate-sars-cov-2-other-coronaviruses-cause-less-severe-disease
METHODS AND COMPOSITIONS FOR CHIMERIC CORONAVIRUS SPIKE PROTEINS (BARIC)

SPILLOVER AND PANDEMIC PROPERTIES OF ZOONOTIC VIRUSES WITH HIGH HOST PLASTICITY (DASZAK)
SARS-LIKE WIV1-COV POISED FOR HUMAN EMERGENCE / SHI ZHENGLI AND BARIC CO-AUTHORS

OXFORD UNIVERSITY PUBLICATION NATURE "ENGINEERED BAT VIRUS STIRS DEBATE OVER RISKY RESEARCH: LAB-MADE CORONA VIRUS RELATED TO SARS CAN INFECT HUMAN CELLS"
 November 12, 2015 "Declan Butler: An experiment that created a hybrid version of a bat coronavirus one related to the virus that causes SARS (severe acute respiratory syndrome) has triggered renewed debate over whether engineering lab variants of viruses with possible pandemic potential is worth the risks. In an article published in Nature Medicine1 on November 9, 2015, scientists investigated a virus called SHC014, which is found in horseshoe bats in China. The researchers created a chimaeric virus, made up of a surface protein of SHC014 and the backbone of a SARS virus that had been adapted to grow in mice and to mimic human disease. The chimaera infected human airway cells proving that the surface protein of SHC014 has the necessary structure to bind to a key receptor on the cells and to infect them. It also caused disease in mice, but did not kill them. Although almost all coronaviruses isolated from bats have not been able to bind to the key human receptor, SHC014, is not the first that can do so. In 2013, researchers reported this ability for the first time in a different coronavirus isolated from the same bat population. The findings reinforce suspicions that bat coronaviruses capable of directly infecting humans (rather than first needing to evolve in an intermediate animal host) may be more common than previously thought, the researchers say. But other virologists question whether the information gleaned from the experiment justifies the potential risk. Although the extent of any risk is difficult to assess, Simon Wain-Hobson, a virologist at the Pasteur Institute in Paris, points out that the researchers have created a novel virus that grows remarkably well in human cells. If the virus escaped, nobody could predict the trajectory, he says.
November 12, 2015 "Declan Butler: An experiment that created a hybrid version of a bat coronavirus one related to the virus that causes SARS (severe acute respiratory syndrome) has triggered renewed debate over whether engineering lab variants of viruses with possible pandemic potential is worth the risks. In an article published in Nature Medicine1 on November 9, 2015, scientists investigated a virus called SHC014, which is found in horseshoe bats in China. The researchers created a chimaeric virus, made up of a surface protein of SHC014 and the backbone of a SARS virus that had been adapted to grow in mice and to mimic human disease. The chimaera infected human airway cells proving that the surface protein of SHC014 has the necessary structure to bind to a key receptor on the cells and to infect them. It also caused disease in mice, but did not kill them. Although almost all coronaviruses isolated from bats have not been able to bind to the key human receptor, SHC014, is not the first that can do so. In 2013, researchers reported this ability for the first time in a different coronavirus isolated from the same bat population. The findings reinforce suspicions that bat coronaviruses capable of directly infecting humans (rather than first needing to evolve in an intermediate animal host) may be more common than previously thought, the researchers say. But other virologists question whether the information gleaned from the experiment justifies the potential risk. Although the extent of any risk is difficult to assess, Simon Wain-Hobson, a virologist at the Pasteur Institute in Paris, points out that the researchers have created a novel virus that grows remarkably well in human cells. If the virus escaped, nobody could predict the trajectory, he says.
THE ARGUMENT IS ESSENTIALLY A RERUN OF THE DEBATE OVER WHETHER TO ALLOW LAB RESEARCH THAT INCREASES THE VIRULENCE, EASE OF SPREAD OR HOST RANGE OF
DANGEROUS PATHOGENS WHAT IS KNOWN AS GAIN-OF-FUNCTION RESEARCH.
2015 ISOLATION AND CHARACTERIZATION OF A NOVEL BAT CORONA VIRUS CLOSELY RELATED TO THE DIRECT PROGENITOR OF SEVERE ACUTE RESPIRATORY SYNDROME CORONAVIRUS
A REPORT OF THE NATIONAL SCIENCE ADVISORY BOARD FOR BIOSECURITY RECOMMENDATIONS FOR THE EVALUATION AND OVERSIGHT OF PROPOSED GAIN-OF-FUNCTION RESEARCH
THIS REPORT IS CRITICAL OF GOF
NSABB_Final_Report_Recommendations_Evaluation_Oversight_Proposed_Gain_of_Function_Research.pdf
BAT SEVERE ACUTE RESPIRATORY SYNDROME-LIKE CORONAVIRUS WIV1 ENCODES AN EXTRA ACCESSORY PROTEIN, ORFX, INVOLVED IN MODULATION OF THE HOST IMMUNE RESPONSE (BARIC, SHI ZHENGLI)
EFFICIENT REVERSE GENETIC SYSTEMS FOR RAPID GENETIC MANIPULATION OF EMERGENT AND PREEMERGENT INFECTIOUS CORONAVIRUSES



Adam S Cockrell 1, Anne Beall 2, Boyd Yount 1, Ralph Baric 3 4
Affiliations
1 Department of Epidemiology, University of North Carolina-Chapel Hill, Chapel Hill, NC, USA.
2 Departments of Microbiology and Immunology, University of North Carolina-Chapel Hill, Chapel Hill, NC, USA.
3 Department of Epidemiology, University of North Carolina-Chapel Hill, Chapel Hill, NC, USA.
4 Departments of Microbiology and Immunology, University of North Carolina-Chapel Hill, Chapel Hill, NC, USA. rbaric@email.unc.edu.
PMID: 28508214 PMCID: PMC7120940 DOI: 10.1007/978-1-4939-6964-7_5
2017 MIT REPORTED A group called Global Virome Project will be visiting Beijing to discuss the scope of the project, which is sponsored by USAID and other organizations. They plan to have US and China be the leaders of the project. The China host is China CDC and our dear friend George Gao is China POC for this project. The purpose of the project is to identify viruses present in the wild life with potential crossing over to humans, causing human infection and disease. Following the identification of the viruses is the development of vaccines to protect human population. China has huge capacity for vaccine development (I think it has 7 national owned vaccine manufacturing facility and over 30 private vaccine making companies). (b) (5) One of the partners in this project is EcoHealth Alliance. Peter Daszak from EcoHealth Alliance is one of the leaders for the GYP project and he has NIAID grant from ROB looking at the coronaviruses in Bat populations in China in collaboration with Wuhan Institute of Virology. He came to visit me once in the Embassy. This grant has direct connection with the purpose of GVP. The meeting is scheduled for Feb. 6-7 in Beijing and it is conflict with our Japan EID meeting. Thank you and have a nice weekend. Ping Chen, PhD Director of NIAID Office in China Office of Global Research, NIAID, NIH Ping Chen, PhD Director of NIAID Office in China Office of Global Research, NIAID, NIH
A group called Global Virome Project will be visiting Beijing to discuss the scope of the project, which is sponsored by USAID and other organizations. They plan to have US and China be the leaders of the project. The China host is China CDC and our dear friend George Gao is China POC for this project. The purpose of the project is to identify viruses present in the wild life with potential crossing over to humans, causing human infection and disease. Following the identification of the viruses is the development of vaccines to protect human population. China has huge capacity for vaccine development (I think it has 7 national owned vaccine manufacturing facility and over 30 private vaccine making companies). (b) (5) One of the partners in this project is EcoHealth Alliance. Peter Daszak from EcoHealth Alliance is one of the leaders for the GYP project and he has NIAID grant from ROB looking at the coronaviruses in Bat populations in China in collaboration with Wuhan Institute of Virology. He came to visit me once in the Embassy. This grant has direct connection with the purpose of GVP. The meeting is scheduled for Feb. 6-7 in Beijing and it is conflict with our Japan EID meeting. Thank you and have a nice weekend. Ping Chen, PhD Director of NIAID Office in China Office of Global Research, NIAID, NIH Ping Chen, PhD Director of NIAID Office in China Office of Global Research, NIAID, NIH INSIDE THE CHINESE LAB POISED TO STUDY WORLD'S MOST DANGEROUS PATHOGENS MAXIMUM-SECURITY BIOLAB IS PART OF PLAN TO BUILD NETWORK OF BSL-4 FACILITIES ACROSS CHINA

BAT CAVE SOLVES MYSTERY OF DEADLY SARS VIRUS AND SUGGESTS NEW OUTBREAK COULD OCCUR: CHINESE SCIENTISTS FIND ALL THE GENETIC BUILDING BLOCKS OF SARS IN A SINGLE POPULATION OF HORSESHOE BATS.
NOVEMBER 2017 DISCOVERY OF A RICH GENE POOL OF BAT SARS-RELATED CORONAVIRUSES PROVIDES NEW INSIGHTS INTO THE ORIGIN OF SARS CORONAVIRUS

GO BACK
https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1006698
TRUMP LIFTS GAIN-OF-FUNCTION / GAIN-OF-THREAT BAN
https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(18)30006-9/fulltext
BSL-4 OPENS AT WIV JANUARY 19, 2018
NIH LIFTS FUNDING PAUSE ON GAIN-OF-THREAT RESEARCH
THE STATE DEPARTMENT CABLES INDICATE A LACK OF TRAINED TECHNICIANS AT WIV

BAT CORONAVIRUSES IN CHINA

WANG YANYI
WUHAN LAB CHIEF WANG YANYI DISPUTES APRIL 2018 WIV NATURE ARTICLE
https://www.scmp.com/news/china/science/article/3085825/coronavirus-conspiracy-lab-leak-claims-pure-fabrication-wuhan
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6466186 THE WUHAN INSTITUTE OF VIROLOGY VIRUS DATABASE WAS TAKEN OFFLINE
https://www.vanityfair.com/news/2021/06/the-lab-leak-theory-inside-the-fight-to-uncover-covid-19s-origins
DATA BASE DISAPPEARS
LACK OF CELL PHONE ACTIVITY AT WIV
https://www.documentcloud.org/documents/6884792-MACE-E-PAI-COVID-19-ANALYSIS-Redacted.html
ROAD BLOCKS AROUND WIV
FINANCING SHI ZHENGLI'S GAIN-OF-FUNCTION / GAIN-OF-THREAT EXPERIMENTS WAS PART OF THE PREDICT PROGRAM AND THAT IS WHY IT WAS SHUT DOWN

https://www.nytimes.com/2019/10/25/health/predict-usaid-viruses.html
GOP ORIGINS REPORT
PATIENTS ZERO

DASZAK'S EMAIL'S DELETED BETWEEN NOVEMBER 2019 AND JANUARY 2020

FEDERAL OFFICIALS ENDED A MORATORIUM IMPOSED THREE YEARS AGO ON FUNDING RESEARCH THAT ALTERS GERMS TO MAKE THEM MORE LETHAL
https://www.nytimes.com/2017/12/19/health/lethal-viruses-nih.htm
EARLY W.H.O. REPORT
https://www.who.int/csr/don/05-january-2020-pneumonia-of-unkown-cause-china/en/ BARIC: "They created a virus like H5N1 which has a 50 percent mortality rate and they trained it how to be efficiently transmitted in mammals. And so that's scary."

DASZAK WORKED CLOSELY WITH ZHENG-LI SHI WHEN SARS-CoV-19 SURFACED
https://www.nytimes.com/2020/01/28/opinion/coronavirus-china.html 
THE BAT LADY WANTED TO CALL HER VIRUS TARS
New England Journal of Medicine https://www.nejm.org/doi/full/10.1056/NEJMoa2001316 RECTIFICATION OF LAB THAT MAPPED GENOME
WIV WAS CALLING IN OUTSIDERS TO HELP WITH BIOSAFETY
http://english.whiov.cas.cn/Notice2016/202001/t20200115_229449.html 
DASZAK CLAIMS HE WARNED THE WORLD ABOUT COVID
https://www.nytimes.com/2020/02/27/opinion/coronavirus-pandemics.html

https://www.causeiq.com/organizations/scripps-research-institute,330435954"

IT'S NOT FROM THE LAB BECAUSE KRISTIAN ANDERSON COULD HAVE DESIGNED A BETTER VIRUS
FRANCIS 'THE TALKING MULE' COLLINS: "We debated up one side down the other, and ultimately decided if you were a human trying to design a really dangerous coronavirus, you would not design this one."
ZHENG-LI SHI: A PNEUMONIA OUTBREAK ASSOCIATED WITH A NEW CORONAVIRUS OF PROBABLE BAT ORIGIN
https://www.nature.com/articles/s41586-020-2012-7**
NEW BIOSECURITY LAW DECREEDED IN CHINA
On FEBRUARY 14, 2020 to the surprise of NSC officials, President Xi Jinping of China announced a plan to fast-track a new biosecurity law to tighten safety procedures throughout the countrys laboratories.
KRISTIAN ANDERSON STATEMENT IN SUPPORT OF THE SCIENTISTS, PUBLIC HEALTH PROFESSIONALS, AND MEDICAL PROFESSIONALS OF CHINA COMBATTING COVID-19
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30418-9/fulltext CHINESE ATTEMPT TO PROVE THAT THE UNIQUE CORONA VIRUS WAS FOUND IN NATURE
https://academic.oup.com/nsr/article/7/6/1012/5775463
OXFORD UNIVERSITY PUBLICATION NATURE "ENGINEERED BAT VIRUS STIRS DEBATE OVER RISKY RESEARCH: LAB-MADE CORONA VIRUS RELATED TO SARS CAN INFECT HUMAN CELLS"
https://www.insidehighered.com/news/2018/10/03/book-publishers-part-ways-springer-nature-over-concerns-about-censorship-china
THE CHINESE RESEARCHERS AT WIV WERE RECEIVING ASSISTANCE FROM THE GALVESTON NATIONAL LABORATORY AT THE UNIVERSITY OF TEXAS MEDICAL BRANCH AND OTHER U.S. ORGANIZATIONS, BUT THE CHINESE REQUESTED ADDITIONAL HELP
WHY CITE EXEMPTION FIVE?

TRUMP CUTS OFF FUNDS TO ECOHEALTH ALLIANCE
https://www.the-scientist.com/news-opinion/nih-cancels-funding-for-bat-coronavirus-research-project-7486
https://www.sciencemag.org/news/2020/04/nih-s-axing-bat-coronavirus-grant-horrible-precedent-and-might-break-rules-critics-say
THE UNITED STATES DEPARTMENT OF EDUCATION HAS ASKED THE UNIVERSITY OF TEXAS NATIONAL SCIENCE ADVISORY BOARD FOR BIOSECURITY (NSABB) TO PROVIDE DOCUMENTATION OF ITS DEALINGS WITH THE WIV
https://www.wsj.com/articles/u-s-probes-university-of-texas-links-to-chinese-lab-scrutinized-over-coronavirus-11588325401
National Science Advisory Board for Biosecurity (NSABB)
James W. LeDuc, PhD
Director, Galveston National Laboratory
Professor, Department of Microbiology and
Immunology
University of Texas Medical Branch
https://osp.od.nih.gov/biotechnology/national-science-advisory-board-for-biosecurity-nsabbl (DEAD LINK)
WEIFENG SHI CLAIMS ANOMALOUS INSERTIONS CAN HAPPEN NATURALLY

https://www.technologynetworks.com/genomics/news/spotlight-on-coronavirus-evolution-as-sars-cov-2-relative-is-found-in-bats-334649
TWO WUHAN INSTITUTE OF VIROLOGY SCIENTISTS, XING-YI GE AND ZHENGLI-LI SHI, USE REVERSE GENETICS TO GENERATE A CHIMERICAL VIRUS CLOSELY RESEMBLING THE NOVEL CORONAVIRUS COVID-19
ORIGIN AND CROSS-SPECIES TRANSMISSION OF BAT CORONAVIRUSES IN CHINA
POMPEO SAYS VIRUS WAS FROM LAB THEN WALKS IT BACK
https://www.breitbart.com/politics/2020/05/16/exclusive-secretary-pompeo-president-trump-committed-to-holding-chinese-communist-party-accountable-for-coronavirus-pandemic
TRUMP WANTED AN INVESTIGATION OF THE ORIGIN OF THE VIRUS
https://web.archive.org/web/20200601041112/https://www.whitehouse.gov/wp-content/uploads/2020/05/Tedros-Letter.pdf
TRUMP ENDS GRANT TO ECHO HEALTH ALLIANCE
https://www.nytimes.com/2020/05/21/health/wuhan-coronavirus-laboratory.html
ECO-HEALTH ALLIANCE GET ANONYMOUS GRANT FROM A CCP PUPPET

https://www.commerce.gov/news/press-releases/2020/12/commerce-department-will-publish-first-milkoopitary-end-user-list-naming companies connected to Chinese Military

TRUMP PULLS USA OUT OF W.H.O.
NIH GRANT REINSTATED BUT THE MONEY CAN'T BE SUB-AWARDED TO THE WIV
HEADLINES READ: HEINOUS! CORONA VIRUS RESEARCHER SHUT DOWN FOR WUHAN-LAB LINK SLAMS NEW FUNDING RESTRICTIONS PETER DASZAK, PRESIDENT OF THE RESEARCH ORGANIZATION ECOHEALTH ALLIANCE, DESCRIBES HOW HE HAS BEEN CAUGHT IN POLITICAL CROSS-HAIRS OVER HIS PARTNERSHIP WITH A VIROLOGY LAB IN CHINA.
8/21/2020 https://www.nature.com/articles/d41586-020-02473-4
TRUMP WANTS TO FORM A COMMISSION TO LOOK INTO THE WIV LAB LEAK
CHAPTER 24

THE BARCELONA BEEF THEORY W.H.O. LEGITIMATIZED
THE WALL STREET JOURNAL: HOW THE W.H.O.'S HUNT FOR COVID'S ORIGINS STUMBLED IN CHINA
ACCORDING TO W.H.O.'S REPORT DASZAK'S JOINT INTERNATIONAL TEAM EXAMINED FOUR SCENARIOS:

THE W.H.O. TEAM PUSHES THE PREPOSTEROUS THEORY THAT THE VIRUS WAS SPREAD TO HUMANS IN SHIPMENTS OF FROZEN FOODS FROM SPAIN
https://www.forbesindia.com/article/global-news/china-scores-a-public-relations-win-after-who-mission-to-wuhan/66349/1
https://www.nytimes.com/2021/02/09/world/asia/wuhan-china-who-covid.html

THE COVID IN SPAIN WAS NOT FOUND IN THE DRAIN
EVIDENCE FOR THE EARLY OCCURRENCE OF SARS-COV-2 FROM OTHER STUDIES
THE FROZEN FOOD THEORY LACKS ONE THING - THERE WOULD HAVE BEEN COVID CASES IN BARCELONA, SPAIN, BEFORE THE ONES IN HUNAN BROKE OUT
TYPE OF GOODS DEALT BY ENVIRONMENTAL POSITIVE STALLS / RETROSPECTIVE STUDY ON THE COLD CHAIN IN 2019
THE PERSISTENCE OF LIVE SARS-COV-2 IN ENVIRONMENTS RELATED TO THE COLD-CHAIN / EXAMPLES OF INTRODUCTION OF COVID-19 INTO CHINA THROUGH IMPORTED COLD CHAIN PRODUCTS
CONCLUSIONS
THE BARCELONA WASTE WATER SAMPLES WERE TESTED AFTER THE PANDEMIC BEGAN AND ONLY A MARCH 2019 SAMPLE PROVED POSITIVE FOR SARS CoV-19 INDICATED THE SAMPLE WAS CONTAMINATED IN THE LAB
https://www.nytimes.com/2020/06/26/health/coronavirus-spain.html
THE HUNAN MARKET ORIGIN THEORY DEBUNKED BY W.H.O.
W.H.O.- INTRODUCTION THROUGH A LABORATORY INCIDENT
EXPLANATION OF HYPOTHESIS
ARGUMENTS IN FAVOR OF INTRODUCTION THROUGH A LABORATORY INCIDENT
ARGUMENTS AGAINST INTRODUCTION THROUGH A LABORATORY INCIDENT
ASSESSMENT OF LIKELIHOOD
REFERENCES IN W.H.O. REPORT INDICATE ANDERSON INTERFACES WITH ECOHEALTH
China owns Nature Magazine: Debunking The proximal origin of SARS-CoV-2 claiming COVID-19 definitely wasnt from a lab
https://www.insidehighered.com/news/2018/10/03/book-publishers-part-ways-springer-nature-over-concerns-about-censorship-china
HIGHLIGHTS FROM SHORT W.H.O. REPORT
ENTIRE SHORT REPORT
W.H.O. REPORT IS A BIG NOTHING BURGER
Source of infection
USG REJECTS WHO INVESTIGATION
WALL STREET JOURNAL: SCIENTISTS REJECT COVID-COVER-UP ORCHESTRATED BY DASZAK

CHAPTER 25
INVESTIGATE THE ORIGINS OF COVID-19
Jesse D. Bloom 1,2, Yujia Alina Chan 3, Ralph S. Baric 4, Pamela J. Bjorkman 5, Sarah Cobey 6, Benjamin E. Deverman 3,
David N. Fisman 7, Ravindra Gupta 8, Akiko Iwasaki 9,2, Marc Lipsitch 10, Ruslan Medzhitov 9,2, Richard A. Nehe r11,
Rasmus Nielsen 12, Nick Patterson 13, Tim Stearns 14, Erik van Nimwegen 11, Michael Worobey 15, David A. Relman 16,17,*
1. Basic Sciences and Computational Biology, Fred Hutchinson Cancer Research Center, Seattle, WA 98109, USA.
2. Howard Hughes Medical Institute, Chevy Chase, MD 20815, USA.
3. Stanley Center for Psychiatric Research, Broad Institute of Massachusetts Institute of Technology
and Harvard University, Cambridge, MA 02142, USA.
4. Department of Epidemiology and Department of Microbiology & Immunology,
University of North Carolina at Chapel Hill, Chapel Hill, NC 27599, USA.
5. Division of Biology and Biological Engineering, California Institute of Technology, Pasadena, CA 91125, USA.
6. Department of Ecology and Evolution, University of Chicago, Chicago, IL 60637, USA.
7.Dalla Lana School of Public Health, University of Toronto, Toronto, ON M5S 1A8, Canada.
8. Cambridge Institute of Therapeutic Immunology & Infectious Disease, Cambridge, UK.
9. Department of Immunobiology, Yale University School of Medicine, New Haven, CT 06519, USA.
10. Department of Immunology and Infectious Diseases and Center for Communicable Disease Dynamics,
Department of Epidemiology, Harvard T. H. Chan School of Public Health, Boston, MA 02115, USA.
11. Biozentrum, University of Basel and Swiss Institute of Bioinformatics, Basel, Switzerland.
12. Department of Integrative Biology and Department of Statistics, University of California, Berkeley, CA 94720, USA.
13. Department of Human Evolutionary Biology, Harvard University, Cambridge, MA 02138, USA.
14. Department of Biology and Department of Genetics, Stanford University, Stanford, CA 94305, USA.
15. Department of Ecology and Evolutionary Biology, University of Arizona, Tucson, AZ 85721, USA.
16. Department of Medicine and Department of Microbiology & Immunology,
Stanford University School of Medicine, Stanford, CA 94305, USA.
17. Center for International Security and Cooperation, Stanford University, Stanford, CA 94305, USA.
?* Corresponding author. Email: relman@stanford.edu
https://science.sciencemag.org/content/372/6543/694.1
 GO BACK
GO BACK
FRANK COLLINS CLAIMS NEITHER NIH NOR NIAID HAVE EVER APPROVED ANY GRANT THAT WOULD HAVE SUPPORTED GAIN-OF-THREAT RESEARCH ON CORONAVIRUSES
THAT WOULD HAVE INCREASED THEIR TRANSMISSIBILITY OR LETHALITY FOR HUMANS
https://www.nih.gov/about-nih/who-we-are/nih-director/statements/statement-misinformation-about-nih-support-specific-gain-function-research
STATEMENT BY PRESIDENT JOE BIDEN ON THE INVESTIGATION INTO THE ORIGINS OF COVID-19
https://www.whitehouse.gov/briefing-room/statements-releases/2021/05/26/statement-by-president-joe-biden-on-the-investigation-into-the-origins-of-covid-19
newyorker.com/news/annals-of-inquiry/the-sudden-rise-of-the-coronavirus-lab-leak-theory
CCP MOUTHPIECE GLOBAL TIMES: US INTELLIGENCE INVESTIGATION OF CORONAVIRUS ORIGINS EXPOSES ITS FOUR ACHILLES HEELS

https://scholar.google.com/citations?user=cwUzg-kAAAAJ

W.H.O. WILL ANNOUNCE NEW TEAM TO STUDY CORONAVIRUS ORIGINS
This new group can do all the fancy footwork it wants, but Chinas not going to cooperate, David Fidler, a senior fellow for global health at the Council on Foreign Relations.
The new committee, is known as the Scientific Advisory Group for the Origins of Novel Pathogens. After the Sago list was released, proponents of a lab investigation raised concerns including in open letters to the WHO last week calling for some nominees to be removed and suggesting the proposed group did not include those with the skills or interest to guide recommendations on how best to evaluate the lab leak hypothesis. Beijing has vehemently rejected this theory, which it views as politically motivated and fanned by the US, and has made clear it will not allow further missions to China that seek to audit laboratories. CCP
SAGO WHO?https://t.co/HVOQDUshTuCHAPTER 26
TRUMP-FUELED SUSPICION OF WUHAN LAB ENSNARES NEW YORK NONPROFIT RESEARCHING BAT CORONAVIRUSES. WAPO May 12, 2020 By Paul Sonne and Shane Harris
CHAPTER 27
Mr. SCIENCE, ANTHONY FAUCI EMAILS
This clip is something else
pic.twitter.com/KTdhMsOt9W
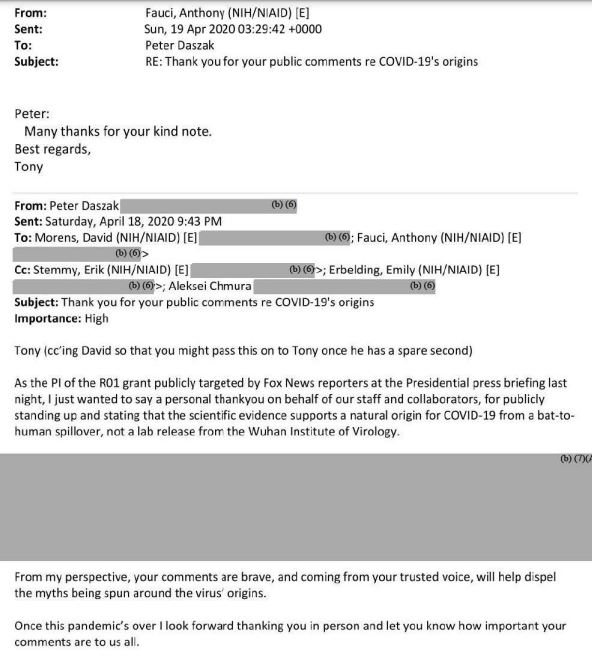

https://www.cnn.com/2021/06/03/health/anthony-fauci-emails/index.htmlCHAPTER 28
THE ECO-HEALTH ALLIANCE
RE-BRANDING PRESS RELEASE
(2) One EcoHealth/WIV lab-generated virus exhibited higher pathogenicity than the starting bat virus in infection studies with humanized mice.CHAPTER 29
THE ECO-HEALTH ALLIANCE GRANTS


https://www.usaspending.gov/search/? hash=29bacd91b448e1783d9e225844f469c7
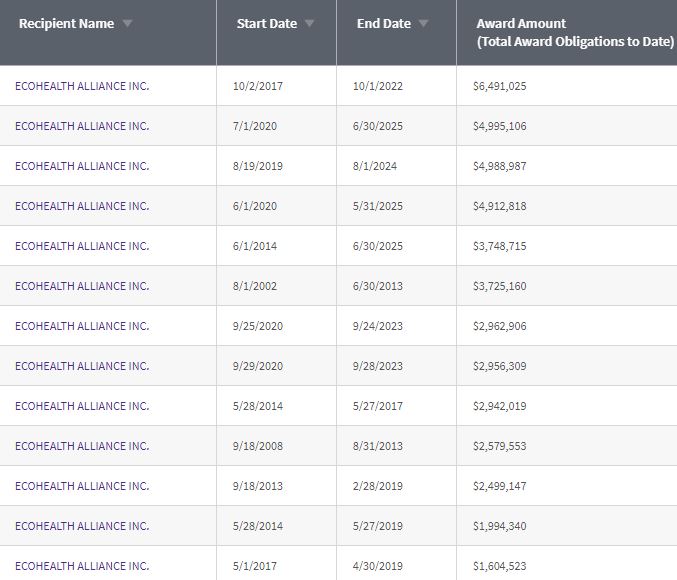
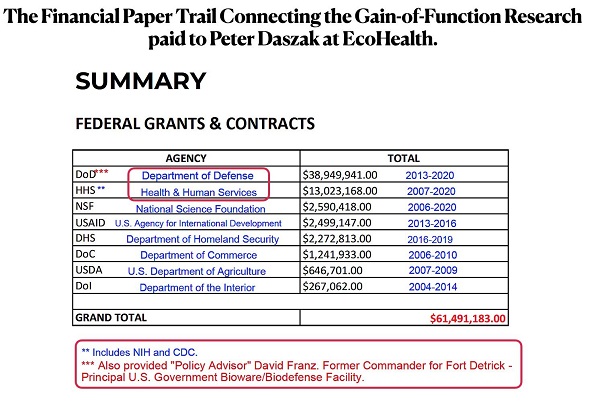
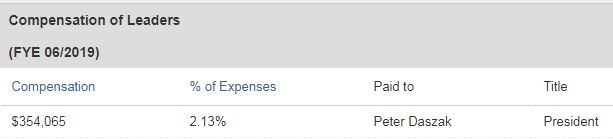
Program Name
Amount Spent
% of Program Expenses
USAID Predict Emerging Pandemic Threat (EPTP)
$12,569,067
83.4%
RVF Virus Investigation
$1,134,848
7.5%
Biological Surveillance
$679,507
4.5%

ECOHEALTH ALLIANCE INC
Address: 1200 LINCOLN AVENUE PROSPECT PARK, NY 10001
County: NEW YORK
Country: United States of America
Type: Supplier Organizations ( Service, Supplies, Material and Equipment )
Class: Private Profit ( Small Business ) Organizations
SAM HUSSEINI: PETER DASZAK'S ECOHEALTH ALLIANCE HAS HIDDEN ALMOST $40 MILLION IN PENTAGON FUNDING AND MILITARIZED PANDEMIC SCIENCE
 Sam Husseini is an Arab of Christian descent. Sam describes himself as a Jordanian-Palestinian writer and "political activist." He is the communications director of the Institute for Public Accuracy, a D.C.-based nonprofit group that promotes anti-Israel "progressive" experts as alternative sources for mainstream media reporters. He formerly worked at the American-Arab Anti-Discrimination Committee and at the media watch group Fairness and Accuracy in Reporting. He is funded by the Open Society Foundation headed by George Soros who collaborated with Nazis during WWII, however his reporting is accurate when it comes to Daszak.
Sam Husseini is an Arab of Christian descent. Sam describes himself as a Jordanian-Palestinian writer and "political activist." He is the communications director of the Institute for Public Accuracy, a D.C.-based nonprofit group that promotes anti-Israel "progressive" experts as alternative sources for mainstream media reporters. He formerly worked at the American-Arab Anti-Discrimination Committee and at the media watch group Fairness and Accuracy in Reporting. He is funded by the Open Society Foundation headed by George Soros who collaborated with Nazis during WWII, however his reporting is accurate when it comes to Daszak.